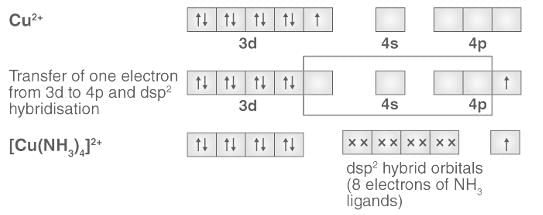
Valence Bond Approach: structure, color & magnetic properties of CN Complexes
MCQ Practice Test & Solutions: Test: Valence Bond Approach: structure, color & magnetic properties of CN Complexes (10 Questions)
You can prepare effectively for Chemistry Inorganic Chemistry with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Valence Bond Approach: structure, color & magnetic properties of CN Complexes ". These 10 questions have been designed by the experts with the latest curriculum of Chemistry 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 10
Sign up on EduRev for free to attempt this test and track your preparation progress.
For the central metal atom in a complex, coordination number is
Detailed Solution: Question 1
The square planar geometry is based on
Detailed Solution: Question 2
The minimum number of monodentate ligand required for the formation of square planar and tetrahedral complex is
Detailed Solution: Question 3
The zero magnetic moment of octahedral K2NiF6 is due to
Detailed Solution: Question 4
Cuprammonium ion has ___________ shape.
Detailed Solution: Question 5
Which orbital would form a more stronger bond if both of them have identical stability?
Detailed Solution: Question 6
The p-orbital is in the shape of a _____________
Detailed Solution: Question 7
According to VBT, the formation of a stable bond requires _____________
Detailed Solution: Question 8
Valence Bond Theory was developed in the year?
Detailed Solution: Question 9
The s-orbital does not show preference to any direction because _____________
Detailed Solution: Question 10
41 videos|93 docs|41 tests |