Kinetic Energy and Molecular Velocities - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Kinetic Energy and Molecular Velocities (10 Questions)
You can prepare effectively for Chemistry Physical Chemistry with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Kinetic Energy and Molecular Velocities". These 10 questions have been designed by the experts with the latest curriculum of Chemistry 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 30 minutes
- - Number of Questions: 10
Sign up on EduRev for free to attempt this test and track your preparation progress.
What is the ratio of urms to ump in oxygen gas at 298k?
Detailed Solution: Question 1
What is the ratio of root mean square speed of 16 grams of Oxygen to 4 grams of hydrogen?
Detailed Solution: Question 2
If the root mean square speed of an argon gas atom at temperature T is equal to the average speed of a helium gas atom at -20 °C, then T will be:
Given:- atomic mass of Ar = 39.9 u
Atomic mass of He = 4.0 u
Detailed Solution: Question 3
What is the ratio of the rms velocities of 2 moles of hydrogen to five moles of helium?
Detailed Solution: Question 4
Calculate the root mean square speed of hydrogen in m/s at 27°C?
Detailed Solution: Question 5
The speed of three particles is recorded as 3 m/s, 4 m/s, and 5 m/s. What is a root mean square speed of these particles?
Detailed Solution: Question 6
Which of the following is greater for identical conditions and the same gas?
Detailed Solution: Question 7
Which among the following options do you think has the highest average speed?
Detailed Solution: Question 8
According to kinetic theory of gases , the root mean square velocity is directly proportional to:
Detailed Solution: Question 9
The root mean square speed of a gas at a certain condition is 1.128 times greater than the most probable speed.
Detailed Solution: Question 10
84 videos|147 docs|67 tests |








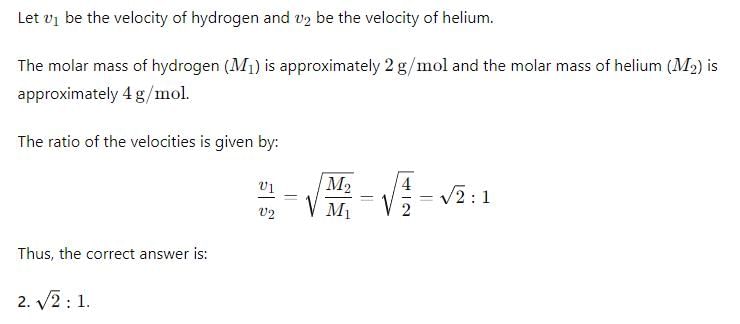
 where R is universal gas constant, T is a temperature in Kelvin and M is the mass in kilograms. From the formula, we understand that the average speed is inversely proportional to the root over the mass. As hydrogen has the least mass among the options it has the highest average speed.
where R is universal gas constant, T is a temperature in Kelvin and M is the mass in kilograms. From the formula, we understand that the average speed is inversely proportional to the root over the mass. As hydrogen has the least mass among the options it has the highest average speed.