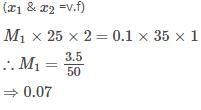Solutions - Free MCQ Practice Test with solutions, JEE Chemistry
MCQ Practice Test & Solutions: P. Bahadur Test: Solutions (25 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "P. Bahadur Test: Solutions". These 25 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 35 minutes
- - Number of Questions: 25
Sign up on EduRev for free to attempt this test and track your preparation progress.
Detailed Solution: Question 1
Detailed Solution: Question 2
Molality is preferred over molarity in handling solutions in chemistry laboratory because
Detailed Solution: Question 3
Aquatic animals are more comfortable in cold water rather that warm water because
Detailed Solution: Question 4
Detailed Solution: Question 5
In the depression of freezing point experiment, it is found that
Detailed Solution: Question 6
If the temperature of an aqueous solution increases, it will cause
Detailed Solution: Question 7
Detailed Solution: Question 8
Detailed Solution: Question 9
Detailed Solution: Question 10
Which among the following is an example of liquid in solid?
Detailed Solution: Question 11
Which among the following is an example of a solid solution in which the solute is a gas?
Detailed Solution: Question 12
The type of intermolecular interaction present in n-Hexane and n-Octane is
Detailed Solution: Question 13
Mathematical expression relating molarity and molality is
Detailed Solution: Question 14
The tanks used by divers are filled with air diluted with
Detailed Solution: Question 15
Ethylene glycol is added to water as antifreeze. It will
Detailed Solution: Question 16
Which of the following solutions of H2SO4 is more concentrated?
Detailed Solution: Question 17
An aqueous solution of methanol and water has vapour pressure
Detailed Solution: Question 18
Detailed Solution: Question 19
A solution showing a large positive deviation from ideal behaviour has
Detailed Solution: Question 20
Detailed Solution: Question 21
In which unit, the concentration of solution remains independent of temperature
Detailed Solution: Question 22
6.3 g oxalic acid is used to make 250 mL aqueous solution .The volume of 0.1N NaOH solution required to neutralize completely 10 mL of this solution is
Detailed Solution: Question 23
The depression in freezing point for 1M urea, 1 M glucose and 1 M NaCl are in the ratio of
Detailed Solution: Question 24
25ml of a solution of barium hydroxide titration with 0.1molar solution of hydro-chloric acid gave a titre value of 35ml.The molarity of barium hydroxide is
Detailed Solution: Question 25
332 videos|699 docs|300 tests |