Compounds of Transition Metals - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Compounds of Transition Metals (20 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Compounds of Transition Metals". These 20 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
In the laboratory, manganese (II) salt is oxidised to permanganate ion in aqueous solution by
Detailed Solution: Question 1
When 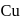 reacts with
reacts with 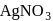 solution, the reaction takes place is
solution, the reaction takes place is
Detailed Solution: Question 2
Detailed Solution: Question 3
Which of the following compounds gives red precipitate with 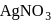 ?
?
Detailed Solution: Question 4
When horn silver ore is dissolved in excess of sodium cyanide solution compound formed is
Detailed Solution: Question 5
Detailed Solution: Question 6
If 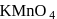 is reduced by oxalic acid in an acidic medium then oxidation number of
is reduced by oxalic acid in an acidic medium then oxidation number of 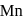 changes from
changes from
Detailed Solution: Question 7
In the preparation of 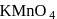 pyrolusite
pyrolusite 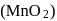 is first converted to potassium manganate
is first converted to potassium manganate 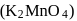 . In this conversion, the oxidation state of manganese changes from
. In this conversion, the oxidation state of manganese changes from
Detailed Solution: Question 8
In an alkaline condition 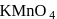 reacts as follows:
reacts as follows:
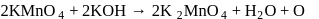
Its equivalent weight is
Detailed Solution: Question 9
In which of the following compounds manganese has oxidation number equal to that of iodine in 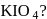
Detailed Solution: Question 10
The colour of 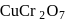 solution in water is green because
solution in water is green because
Detailed Solution: Question 11
A compound of iron exists as a dimer in vapour state. It is hygroscopic in nature and dissolves in water giving brown acidic solution. The compound is
Detailed Solution: Question 12
Detailed Solution: Question 13
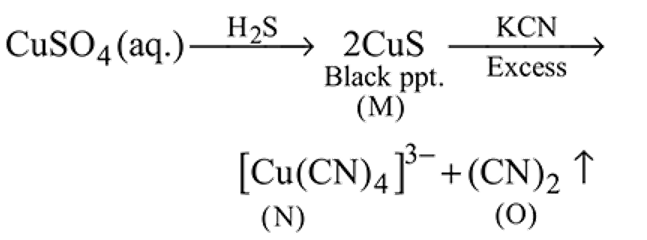
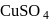 (aq.)
(aq.) 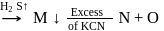
Then final products 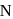 and
and 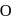 are respectively.
are respectively.
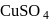 (aq.)
(aq.) 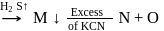
Then final products
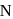 and
and 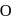 are respectively.
are respectively.Detailed Solution: Question 14
Which of the following statements is false ?
Detailed Solution: Question 15
What is the change in the oxidation state of 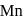 in the reaction of
in the reaction of 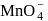 with
with 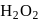 in acidic medium?
in acidic medium?
Detailed Solution: Question 16
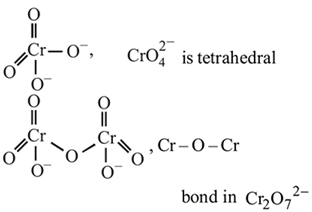
Of Cr 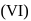 as
as 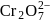 and
and 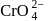 , which is better oxidising agent?
, which is better oxidising agent?
Detailed Solution: Question 17
Detailed Solution: Question 18
Which of the following molecules is colourless?
Detailed Solution: Question 19
The black compound formed during the reaction between sodium thiosulphate and silver nitrate is
Detailed Solution: Question 20
335 videos|699 docs|300 tests |


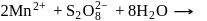
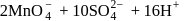
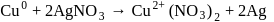
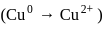

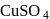 reacts with excees
reacts with excees 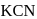 solution to form:
solution to form: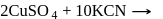
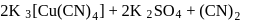
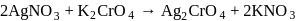
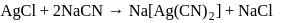
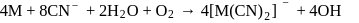
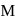 . It is
. It is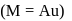 .
.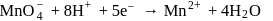
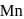 changes form
changes form 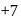 to
to  ).
).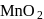 (O.S. of
(O.S. of 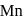 is
is 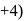 in
in 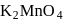 (O.S.of Mn is +6). Hence O.S. changes by 2 .
(O.S.of Mn is +6). Hence O.S. changes by 2 .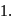 Hence eq. wt. is
Hence eq. wt. is 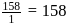
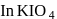 , O.S of I is
, O.S of I is 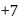 and in
and in 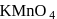 , O.S. of
, O.S. of 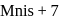
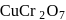 is due to blue colour of
is due to blue colour of 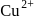 ions and yellow colour of
ions and yellow colour of 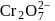 ions
ions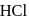 and brown due to
and brown due to 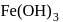
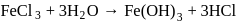
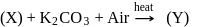
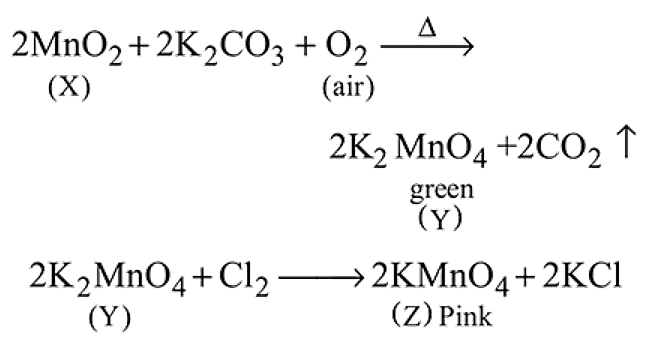
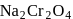 is a secondary standard solution in volumetry as it is hygroscopic in nature.
is a secondary standard solution in volumetry as it is hygroscopic in nature. is greater than
is greater than  because of smaller size of
because of smaller size of 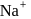 . So,
. So, 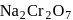 is more soluble than
is more soluble than 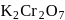
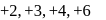 and +7 .
and +7 .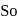 , in acidic medium on reaction of
, in acidic medium on reaction of 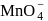 with
with 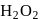 it changes from 7 to 2 .
it changes from 7 to 2 .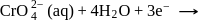
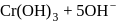 ,
,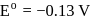
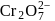 (aq.)
(aq.) 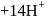 (aq.)
(aq.) 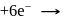
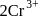 (aq.)
(aq.) 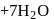 ,
,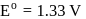
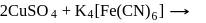
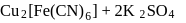
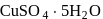 is blue in colour. The ligand (water) molecules causes splitting of
is blue in colour. The ligand (water) molecules causes splitting of 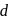 -orbitals. This facilitated
-orbitals. This facilitated 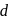 -
- 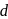 transition and colour.
transition and colour.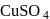 is colourless. In the absence of ligand (water) molecules, splitting of
is colourless. In the absence of ligand (water) molecules, splitting of 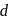 -orbitals is not possible. Hence,
-orbitals is not possible. Hence, 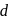 -
- 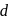 transition is not possible. Hence, option (b) is correct.
transition is not possible. Hence, option (b) is correct.