Periodic Properties- 3 - Free MCQ Practice Test with solutions, Chemistry
MCQ Practice Test & Solutions: Test: Periodic Properties- 3 (30 Questions)
You can prepare effectively for Chemistry Inorganic Chemistry with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Periodic Properties- 3". These 30 questions have been designed by the experts with the latest curriculum of Chemistry 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 90 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which is not similar characteristic(s) about the electronic configuration of Be, Mg, Ca:
Detailed Solution: Question 1
The most electropositive element possesses the electronic configuration:
Which one of the following elements shows both positive and negative oxidation states:
Which of the following arrangements shows the correct order of decreasing paramagnetism:
Detailed Solution: Question 4
Detailed Solution: Question 5
The elements with the lowest atomic number that has a ground state electronic configuration of (n – 1) d6ns2 is located in the:
The atomic numbers of the metallic and non-metallic elements which are liquid at room temperature respectively are:
In the periodic table, metallic character of the elements shows one of the following trend:
Detailed Solution: Question 8
Which of the following represents the correct order of increasing first ionization enthalpy for Ca,Ba,S,Se andAr?
Detailed Solution: Question 9
Sodium generally does not shown oxidation state of +2, because of its:
Which of the following pairs of molecules have the almost identical bond dissociation energy:
According to modern periodic law the properties of elements repeat at regular intervals when the elements are arranged in order of:
Consider the following four elements, which are represented according to long form of periodic table.
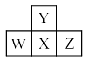
Here W, Y and Z are left, up and right elements with respect to the element ‘X’ and ‘X’ belongs to 16th group and 3rd period. Then according to given informat ion the incorrect statement regarding given elements is:
In the fourth period of the periodic table, how many elements have one or more 4d electrons:
Assuming that elements are formed to complete the seventh period, what would be the atomic number of the alkaline earth metal of the eighth period:
Which of the following represents an excited state of an atom:
Detailed Solution: Question 17
Choose the correct statement regarding transition elements:
Zn and Cd metals do not show variable valency because:
A compound contains three elements A, B and C, if the oxidation number of A = +2, B = +5 and C = –2, the possible formula of the compound is:
Detailed Solution: Question 21
The period number and group number of “Tantalum” (Z = 73) are respectively:
Which of the following pair of elements belongs to the same period?
Detailed Solution: Question 23
Consider the following electronic configuration of an element (P):
[Xe] 4f145d16s2
The correct statement about element ‘P’ is:
Which of the following metal is highest electropositive (metallic) in nature:
Which of the following species must have maximum number of electrons in ‘dxy’ orbital:
Which property decreases from left to right across the periodic table and increases from top to bottom:
(I) Atomic radius
(II) Electronegativity
(III) Ionization energy
(IV) Metallic character
(II) Electronegativity
(III) Ionization energy
(IV) Metallic character
Consider the following information about element P and Q:
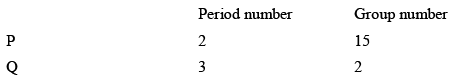
Then formula of the compound formed by P and Q element is:
Detailed Solution: Question 28
Which electronic configuration must represent an atom in an excited state:
Among NaF , NaCl , NaBr and Nal , the NaF has highest melting point because
41 videos|93 docs|41 tests |



