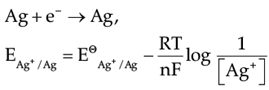Chemistry CUET UG Assertion & Reason Test: Electrochemistry Free Online
MCQ Practice Test & Solutions: Assertion & Reason Test: Electrochemistry (12 Questions)
You can prepare effectively for NEET Chemistry CUET UG Mock Test Series 2026 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Assertion & Reason Test: Electrochemistry". These 12 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 24 minutes
- - Number of Questions: 12
Sign up on EduRev for free to attempt this test and track your preparation progress.
Directions: In the following questions, A statement of Assertion (A) is followed by a statement of Reason (R). Mark the correct choice as.
Assertion (A): Conductivity of an electrolyte increases with decrease in concentration.
Reason (R): Number of ions per unit volume decreases on dilution.
Detailed Solution: Question 1
Directions: In the following questions, A statement of Assertion (A) is followed by a statement of Reason (R). Mark the correct choice as.
Assertion (A): Λm for weak electrolytes shows a sharp increase when the electrolytic solution is diluted.
Reason (R): For weak electrolytes degree of dissociation increases with dilution of solution.
Detailed Solution: Question 2
Directions: In the following questions, A statement of Assertion (A) is followed by a statement of Reason (R). Mark the correct choice as.
Assertion (A): Electrolytic conduction increases with increase in temperature.
Reason (R): Increase in temperature cause the electronic movement more rapid
Detailed Solution: Question 3
Directions: In the following questions, A statement of Assertion (A) is followed by a statement of Reason (R). Mark the correct choice as.
Assertion (A): Molar Conductivity of an ionic solution depends on temperature.
Reason (R): Molar Conductivity of an ionic solution depends on the concentration of electrolytes in the solution.
Detailed Solution: Question 4
Directions: In the following questions, A statement of Assertion (A) is followed by a statement of Reason (R). Mark the correct choice as.
Assertion (A): Ecell should have a positive value for the cell to function.
Reason (R): Ecathode < Eanode.
Detailed Solution: Question 5
Directions: In the following questions, A statement of Assertion (A) is followed by a statement of Reason (R). Mark the correct choice as.
Assertion (A): Cu is less reactive than hydrogen.
Reason (R): Eo Cu2+ / Cu is negative.
Detailed Solution: Question 6
Directions: In the following questions, A statement of Assertion (A) is followed by a statement of Reason (R). Mark the correct choice as.
Assertion (A): Copper sulphate can be stored in a zinc vessel.
Reason (R): Zinc is more reactive than copper.
Detailed Solution: Question 7
Directions: In the following questions, A statement of Assertion (A) is followed by a statement of Reason (R). Mark the correct choice as.
Assertion (A): EAg+/Ag increases with increase in concentration of Ag+ ions.
Reason (R): EAg+/Ag has a positive value.
Detailed Solution: Question 8
Directions: In the following questions, A statement of Assertion (A) is followed by a statement of Reason (R). Mark the correct choice as.
Assertion (A): Electrolysis of NaCl solution gives chlorine at anode instead of O2.
Reason (R): Formation of oxygen at anode requires over voltage.
Detailed Solution: Question 9
Directions: These questions consist of two statements, each printed as Assertion and Reason. While answering these questions, you are required to choose any one of the following four responses.
Assertion: The resistivity for a substance is its resistance when it is one meter long and its area of cross section is one square meter.
Reason: The SI units of resistivity is ohm metre (m).
Detailed Solution: Question 10
Directions: These questions consist of two statements, each printed as Assertion and Reason. While answering these questions, you are required to choose any one of the following four responses.
Assertion : On increasing dilution, the specific conductance keeps on increasing.
Reason : On increasing dilution, degree of ionisation of weak electrolyte increases and molality of ions also increases.
Detailed Solution: Question 11
Directions: These questions consist of two statements, each printed as Assertion and Reason. While answering these questions, you are required to choose any one of the following four responses.
Assertion : Galvanised iron does not rust.
Reason : Zinc has a more negative electrode potential than iron.
Detailed Solution: Question 12
|
4 docs|163 tests
|