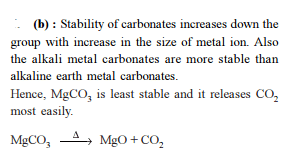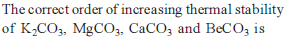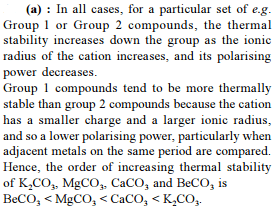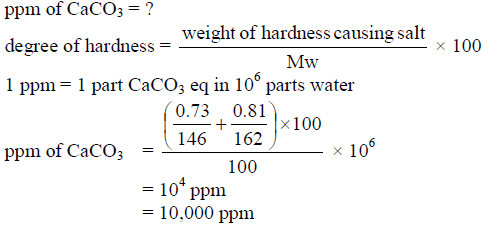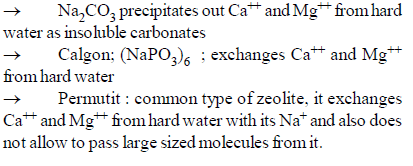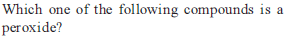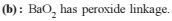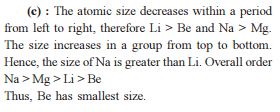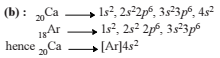s-Block Elements (Alkali and Alkaline Earth Metals) - 2 - Free MCQ Test
MCQ Practice Test & Solutions: s-Block Elements (Alkali and Alkaline Earth Metals) - 2 (30 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "s-Block Elements (Alkali and Alkaline Earth Metals) - 2". These 30 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
An alkaline earth metal (M) gives a salt with chlorine, which is soluble in water at room temperature. It also forms an insoluble sulphate whose mixture with a sulphide of a transition metal is called `lithopone'-a white pigment. Metal M is-
Detailed Solution: Question 1
The hydroxide of alkaline earth metal, which has the lowest value of solubility product (Ksp) at normal temperature (25°C) is-
Detailed Solution: Question 2
Which of the following carbonate of alkali metals has the least thermal stability ?
Detailed Solution: Question 3
The solubility of metal halides depends on their nature, lattice enthalpy and hydration enthalpy of the individual ions. Amongst fluorides of alkali metals, the lowest solubility of LiF in water is due to
Detailed Solution: Question 4
The formula of soda ash is
Detailed Solution: Question 5
The golden yellow colour associated with NaCl to Bunsen flame can be explained on the basis of -
Detailed Solution: Question 6
Detailed Solution: Question 7
The correct order of solubility is -?
Detailed Solution: Question 8
When K2O is added to water, the solution becomes basic in nature because it contains a significant concentration of -
Detailed Solution: Question 9
Detailed Solution: Question 10
Detailed Solution: Question 11
Detailed Solution: Question 12
Detailed Solution: Question 13
Detailed Solution: Question 14
Which is not correctly matched ?
(1) Basic strength of oxides Cs2O< />2O< K2O < Na2O < Li2O(2) Stability of peroxides Na2O2 < K2O2 < Rb2O2 < Cs2O2
(3) Stability of bicarbonates LiHCO3 < NaHCO3 < KHCO3 < RbHCO3 < CsHCO3
(4) Melting point NaF < NaCl < NaBr < NaI
Detailed Solution: Question 15
Detailed Solution: Question 16
The set representing the correct order of first ionization potential is
Detailed Solution: Question 17
Which of the follownig acids will not evolve H2 gas on reaction with alkali metals
Detailed Solution: Question 18
The first four ionization potentials (eV) are given for two elements X and Y. Identify them.
X : 8.296, 25.149, 37.92, 259.298
Y : 5.318, 47.29, 71.65, 98.88
Detailed Solution: Question 19
About alkali metal-liquid NH3 solution which of following statement is not true ?
Detailed Solution: Question 20
Select the correct set of statements
(i) Solubility of alkali metal hydroxides is in order
CsOH > RbOH > KOH > NaOH > LiOH
(ii) Solubility of alkali carbonates is in order
Li2CO3 > Na2CO3 > K2CO3 > Rb2CO3> Cs2O3
(iii) Hydrated radii is in order
Li+ < Na+ < K+ < Rb+ < Cs+
(iv) Stability of peroxides is in order
Na2O2 < K2O2 < Rb2O2 < Cs2O2
The options are :
Detailed Solution: Question 22
Detailed Solution: Question 23
Detailed Solution: Question 24
The alkali metal hydroxides dissolve freely in water with evolution of much heat on account of :
Detailed Solution: Question 25
100 mL of a water sample contains 0.81 g of calcium bicarbonate and 0.73 g of magnesium bicarbonate. The hardness of this water sample expressed in terms of equivalents of CaCO3 is - (molar mass of calcium bicarbonate is 162 g mol–1 and magnesium bicarbonate is 146 g mol–1)
Detailed Solution: Question 26
Which of the following is not a water softener?
Detailed Solution: Question 27
Detailed Solution: Question 28
Detailed Solution: Question 29
Detailed Solution: Question 30
|
335 videos|699 docs|300 tests
|