UPSC Exam > UPSC Tests > Test: Alcohols & Carboxylic Acids - UPSC MCQ
Test: Alcohols & Carboxylic Acids - UPSC MCQ
Test Description
10 Questions MCQ Test - Test: Alcohols & Carboxylic Acids
Test: Alcohols & Carboxylic Acids for UPSC 2025 is part of UPSC preparation. The Test: Alcohols & Carboxylic Acids questions and answers have been prepared
according to the UPSC exam syllabus.The Test: Alcohols & Carboxylic Acids MCQs are made for UPSC 2025 Exam.
Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests for Test: Alcohols & Carboxylic Acids below.
Solutions of Test: Alcohols & Carboxylic Acids questions in English are available as part of our course for UPSC & Test: Alcohols & Carboxylic Acids solutions in
Hindi for UPSC course.
Download more important topics, notes, lectures and mock test series for UPSC Exam by signing up for free. Attempt Test: Alcohols & Carboxylic Acids | 10 questions in 10 minutes | Mock test for UPSC preparation | Free important questions MCQ to study for UPSC Exam | Download free PDF with solutions
Detailed Solution for Test: Alcohols & Carboxylic Acids - Question 1
Test: Alcohols & Carboxylic Acids - Question 2
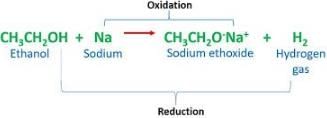
Which gas is released when ethyl alcohol reacts with sodium?
Detailed Solution for Test: Alcohols & Carboxylic Acids - Question 2
Test: Alcohols & Carboxylic Acids - Question 3
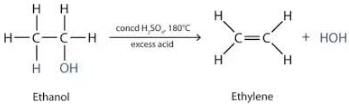
In conversion from ethanol to ethene, concentrated sulphuric acid is used as:
Detailed Solution for Test: Alcohols & Carboxylic Acids - Question 3
Detailed Solution for Test: Alcohols & Carboxylic Acids - Question 4
Detailed Solution for Test: Alcohols & Carboxylic Acids - Question 5
Test: Alcohols & Carboxylic Acids - Question 6
In the reaction, identify the reactant X.
CH3COOH + X CH3COOC2H5 + H2O
Ethanoic
acid
Detailed Solution for Test: Alcohols & Carboxylic Acids - Question 6
Detailed Solution for Test: Alcohols & Carboxylic Acids - Question 7
Test: Alcohols & Carboxylic Acids - Question 8
What is removed from ethanol during its dehydration to ethene?
Detailed Solution for Test: Alcohols & Carboxylic Acids - Question 8
Test: Alcohols & Carboxylic Acids - Question 9
Which of the following will give a pleasant smell of ester when heated with ethyl alcohol and a small quantity of sulphuric acid?
Detailed Solution for Test: Alcohols & Carboxylic Acids - Question 9
Test: Alcohols & Carboxylic Acids - Question 10
Why is ethanoic acid called glacial acetic acid?
Detailed Solution for Test: Alcohols & Carboxylic Acids - Question 10
Information about Test: Alcohols & Carboxylic Acids Page
In this test you can find the Exam questions for Test: Alcohols & Carboxylic Acids solved & explained in the simplest way possible.
Besides giving Questions and answers for Test: Alcohols & Carboxylic Acids, EduRev gives you an ample number of Online tests for practice
Download as PDF