JEE Exam > JEE Questions > For certain substances such as ammonium chlor...
Start Learning for Free
For certain substances such as ammonium chloride, nitrogen peroxide, phosphorus pentachloride, etc. the measured densities are found to be less than those calculated from their molecular formula. The observed densities decrease towards a limit as the temperature is raised. This is due to the splitting of the molecules into simpler ones. The process is reversible and is called thermal dissociation.
Examples :
With increase in the number of molecules, the volume increases (pressure remaining constant) and, in consequence, the density decreases. As the temperature rises, more and more dissociation takes place, and when practically complete dissociation occursthe density reaches its lowest limit.The extent of issociation, ice., the fraction of the total number of molecules which suffers dissociation is called the degree of dissociation. Gas density measurements can be used to determine the degree of dissociation. Let us take by general case where one molecule of a substance A splits up into n molecule of B on heating; i.e.,
t = 0 a
t = teq a — x nx 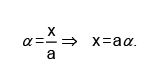
Total no. of moles 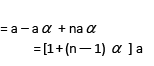
Observed molecular weight or molar mass of the mixture
Q.
x (degree of dissociation) varies with D/d in the above reaction according to :
- a)
- b)
- c)
- d)
Correct answer is option 'B'. Can you explain this answer?
| FREE This question is part of | Download PDF Attempt this Test |
Verified Answer
For certain substances such as ammonium chloride, nitrogen peroxide, p...

|
Explore Courses for JEE exam
|

|
Similar JEE Doubts
For certain substances such as ammonium chloride, nitrogen peroxide, phosphorus pentachloride, etc. the measured densities are found to be less than those calculatedfrom their molecular formula. The observed densities decrease towards a limit as thetemperature is raised. This is due to the splitting of the molecules into simpler ones. The process is reversible and is called thermal dissociation.Examples :With increase in the number of molecules, the volume increases (pressure remaining constant) and, in consequence, the density decreases. As the temperature rises, more and more dissociation takes place, and when practically complete dissociation occursthe density reaches its lowest limit.The extent of issociation, ice., the fraction of the total number of molecules which suffers dissociation is called the degree of dissociation. Gas density measurements can be used to determine the degree of dissociation. Let us take by general case where one molecule of a substance A splits up into n molecule of B on heating; i.e.,t = 0 at = teq a x nx Total no. of moles Observed molecular weight or molar mass of the mixtureQ.x (degree of dissociation) varies with D/d in the above reaction according to :a)b)c)d)Correct answer is option 'B'. Can you explain this answer?
Question Description
For certain substances such as ammonium chloride, nitrogen peroxide, phosphorus pentachloride, etc. the measured densities are found to be less than those calculatedfrom their molecular formula. The observed densities decrease towards a limit as thetemperature is raised. This is due to the splitting of the molecules into simpler ones. The process is reversible and is called thermal dissociation.Examples :With increase in the number of molecules, the volume increases (pressure remaining constant) and, in consequence, the density decreases. As the temperature rises, more and more dissociation takes place, and when practically complete dissociation occursthe density reaches its lowest limit.The extent of issociation, ice., the fraction of the total number of molecules which suffers dissociation is called the degree of dissociation. Gas density measurements can be used to determine the degree of dissociation. Let us take by general case where one molecule of a substance A splits up into n molecule of B on heating; i.e.,t = 0 at = teq a x nx Total no. of moles Observed molecular weight or molar mass of the mixtureQ.x (degree of dissociation) varies with D/d in the above reaction according to :a)b)c)d)Correct answer is option 'B'. Can you explain this answer? for JEE 2024 is part of JEE preparation. The Question and answers have been prepared according to the JEE exam syllabus. Information about For certain substances such as ammonium chloride, nitrogen peroxide, phosphorus pentachloride, etc. the measured densities are found to be less than those calculatedfrom their molecular formula. The observed densities decrease towards a limit as thetemperature is raised. This is due to the splitting of the molecules into simpler ones. The process is reversible and is called thermal dissociation.Examples :With increase in the number of molecules, the volume increases (pressure remaining constant) and, in consequence, the density decreases. As the temperature rises, more and more dissociation takes place, and when practically complete dissociation occursthe density reaches its lowest limit.The extent of issociation, ice., the fraction of the total number of molecules which suffers dissociation is called the degree of dissociation. Gas density measurements can be used to determine the degree of dissociation. Let us take by general case where one molecule of a substance A splits up into n molecule of B on heating; i.e.,t = 0 at = teq a x nx Total no. of moles Observed molecular weight or molar mass of the mixtureQ.x (degree of dissociation) varies with D/d in the above reaction according to :a)b)c)d)Correct answer is option 'B'. Can you explain this answer? covers all topics & solutions for JEE 2024 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for For certain substances such as ammonium chloride, nitrogen peroxide, phosphorus pentachloride, etc. the measured densities are found to be less than those calculatedfrom their molecular formula. The observed densities decrease towards a limit as thetemperature is raised. This is due to the splitting of the molecules into simpler ones. The process is reversible and is called thermal dissociation.Examples :With increase in the number of molecules, the volume increases (pressure remaining constant) and, in consequence, the density decreases. As the temperature rises, more and more dissociation takes place, and when practically complete dissociation occursthe density reaches its lowest limit.The extent of issociation, ice., the fraction of the total number of molecules which suffers dissociation is called the degree of dissociation. Gas density measurements can be used to determine the degree of dissociation. Let us take by general case where one molecule of a substance A splits up into n molecule of B on heating; i.e.,t = 0 at = teq a x nx Total no. of moles Observed molecular weight or molar mass of the mixtureQ.x (degree of dissociation) varies with D/d in the above reaction according to :a)b)c)d)Correct answer is option 'B'. Can you explain this answer?.
For certain substances such as ammonium chloride, nitrogen peroxide, phosphorus pentachloride, etc. the measured densities are found to be less than those calculatedfrom their molecular formula. The observed densities decrease towards a limit as thetemperature is raised. This is due to the splitting of the molecules into simpler ones. The process is reversible and is called thermal dissociation.Examples :With increase in the number of molecules, the volume increases (pressure remaining constant) and, in consequence, the density decreases. As the temperature rises, more and more dissociation takes place, and when practically complete dissociation occursthe density reaches its lowest limit.The extent of issociation, ice., the fraction of the total number of molecules which suffers dissociation is called the degree of dissociation. Gas density measurements can be used to determine the degree of dissociation. Let us take by general case where one molecule of a substance A splits up into n molecule of B on heating; i.e.,t = 0 at = teq a x nx Total no. of moles Observed molecular weight or molar mass of the mixtureQ.x (degree of dissociation) varies with D/d in the above reaction according to :a)b)c)d)Correct answer is option 'B'. Can you explain this answer? for JEE 2024 is part of JEE preparation. The Question and answers have been prepared according to the JEE exam syllabus. Information about For certain substances such as ammonium chloride, nitrogen peroxide, phosphorus pentachloride, etc. the measured densities are found to be less than those calculatedfrom their molecular formula. The observed densities decrease towards a limit as thetemperature is raised. This is due to the splitting of the molecules into simpler ones. The process is reversible and is called thermal dissociation.Examples :With increase in the number of molecules, the volume increases (pressure remaining constant) and, in consequence, the density decreases. As the temperature rises, more and more dissociation takes place, and when practically complete dissociation occursthe density reaches its lowest limit.The extent of issociation, ice., the fraction of the total number of molecules which suffers dissociation is called the degree of dissociation. Gas density measurements can be used to determine the degree of dissociation. Let us take by general case where one molecule of a substance A splits up into n molecule of B on heating; i.e.,t = 0 at = teq a x nx Total no. of moles Observed molecular weight or molar mass of the mixtureQ.x (degree of dissociation) varies with D/d in the above reaction according to :a)b)c)d)Correct answer is option 'B'. Can you explain this answer? covers all topics & solutions for JEE 2024 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for For certain substances such as ammonium chloride, nitrogen peroxide, phosphorus pentachloride, etc. the measured densities are found to be less than those calculatedfrom their molecular formula. The observed densities decrease towards a limit as thetemperature is raised. This is due to the splitting of the molecules into simpler ones. The process is reversible and is called thermal dissociation.Examples :With increase in the number of molecules, the volume increases (pressure remaining constant) and, in consequence, the density decreases. As the temperature rises, more and more dissociation takes place, and when practically complete dissociation occursthe density reaches its lowest limit.The extent of issociation, ice., the fraction of the total number of molecules which suffers dissociation is called the degree of dissociation. Gas density measurements can be used to determine the degree of dissociation. Let us take by general case where one molecule of a substance A splits up into n molecule of B on heating; i.e.,t = 0 at = teq a x nx Total no. of moles Observed molecular weight or molar mass of the mixtureQ.x (degree of dissociation) varies with D/d in the above reaction according to :a)b)c)d)Correct answer is option 'B'. Can you explain this answer?.
Solutions for For certain substances such as ammonium chloride, nitrogen peroxide, phosphorus pentachloride, etc. the measured densities are found to be less than those calculatedfrom their molecular formula. The observed densities decrease towards a limit as thetemperature is raised. This is due to the splitting of the molecules into simpler ones. The process is reversible and is called thermal dissociation.Examples :With increase in the number of molecules, the volume increases (pressure remaining constant) and, in consequence, the density decreases. As the temperature rises, more and more dissociation takes place, and when practically complete dissociation occursthe density reaches its lowest limit.The extent of issociation, ice., the fraction of the total number of molecules which suffers dissociation is called the degree of dissociation. Gas density measurements can be used to determine the degree of dissociation. Let us take by general case where one molecule of a substance A splits up into n molecule of B on heating; i.e.,t = 0 at = teq a x nx Total no. of moles Observed molecular weight or molar mass of the mixtureQ.x (degree of dissociation) varies with D/d in the above reaction according to :a)b)c)d)Correct answer is option 'B'. Can you explain this answer? in English & in Hindi are available as part of our courses for JEE.
Download more important topics, notes, lectures and mock test series for JEE Exam by signing up for free.
Here you can find the meaning of For certain substances such as ammonium chloride, nitrogen peroxide, phosphorus pentachloride, etc. the measured densities are found to be less than those calculatedfrom their molecular formula. The observed densities decrease towards a limit as thetemperature is raised. This is due to the splitting of the molecules into simpler ones. The process is reversible and is called thermal dissociation.Examples :With increase in the number of molecules, the volume increases (pressure remaining constant) and, in consequence, the density decreases. As the temperature rises, more and more dissociation takes place, and when practically complete dissociation occursthe density reaches its lowest limit.The extent of issociation, ice., the fraction of the total number of molecules which suffers dissociation is called the degree of dissociation. Gas density measurements can be used to determine the degree of dissociation. Let us take by general case where one molecule of a substance A splits up into n molecule of B on heating; i.e.,t = 0 at = teq a x nx Total no. of moles Observed molecular weight or molar mass of the mixtureQ.x (degree of dissociation) varies with D/d in the above reaction according to :a)b)c)d)Correct answer is option 'B'. Can you explain this answer? defined & explained in the simplest way possible. Besides giving the explanation of
For certain substances such as ammonium chloride, nitrogen peroxide, phosphorus pentachloride, etc. the measured densities are found to be less than those calculatedfrom their molecular formula. The observed densities decrease towards a limit as thetemperature is raised. This is due to the splitting of the molecules into simpler ones. The process is reversible and is called thermal dissociation.Examples :With increase in the number of molecules, the volume increases (pressure remaining constant) and, in consequence, the density decreases. As the temperature rises, more and more dissociation takes place, and when practically complete dissociation occursthe density reaches its lowest limit.The extent of issociation, ice., the fraction of the total number of molecules which suffers dissociation is called the degree of dissociation. Gas density measurements can be used to determine the degree of dissociation. Let us take by general case where one molecule of a substance A splits up into n molecule of B on heating; i.e.,t = 0 at = teq a x nx Total no. of moles Observed molecular weight or molar mass of the mixtureQ.x (degree of dissociation) varies with D/d in the above reaction according to :a)b)c)d)Correct answer is option 'B'. Can you explain this answer?, a detailed solution for For certain substances such as ammonium chloride, nitrogen peroxide, phosphorus pentachloride, etc. the measured densities are found to be less than those calculatedfrom their molecular formula. The observed densities decrease towards a limit as thetemperature is raised. This is due to the splitting of the molecules into simpler ones. The process is reversible and is called thermal dissociation.Examples :With increase in the number of molecules, the volume increases (pressure remaining constant) and, in consequence, the density decreases. As the temperature rises, more and more dissociation takes place, and when practically complete dissociation occursthe density reaches its lowest limit.The extent of issociation, ice., the fraction of the total number of molecules which suffers dissociation is called the degree of dissociation. Gas density measurements can be used to determine the degree of dissociation. Let us take by general case where one molecule of a substance A splits up into n molecule of B on heating; i.e.,t = 0 at = teq a x nx Total no. of moles Observed molecular weight or molar mass of the mixtureQ.x (degree of dissociation) varies with D/d in the above reaction according to :a)b)c)d)Correct answer is option 'B'. Can you explain this answer? has been provided alongside types of For certain substances such as ammonium chloride, nitrogen peroxide, phosphorus pentachloride, etc. the measured densities are found to be less than those calculatedfrom their molecular formula. The observed densities decrease towards a limit as thetemperature is raised. This is due to the splitting of the molecules into simpler ones. The process is reversible and is called thermal dissociation.Examples :With increase in the number of molecules, the volume increases (pressure remaining constant) and, in consequence, the density decreases. As the temperature rises, more and more dissociation takes place, and when practically complete dissociation occursthe density reaches its lowest limit.The extent of issociation, ice., the fraction of the total number of molecules which suffers dissociation is called the degree of dissociation. Gas density measurements can be used to determine the degree of dissociation. Let us take by general case where one molecule of a substance A splits up into n molecule of B on heating; i.e.,t = 0 at = teq a x nx Total no. of moles Observed molecular weight or molar mass of the mixtureQ.x (degree of dissociation) varies with D/d in the above reaction according to :a)b)c)d)Correct answer is option 'B'. Can you explain this answer? theory, EduRev gives you an
ample number of questions to practice For certain substances such as ammonium chloride, nitrogen peroxide, phosphorus pentachloride, etc. the measured densities are found to be less than those calculatedfrom their molecular formula. The observed densities decrease towards a limit as thetemperature is raised. This is due to the splitting of the molecules into simpler ones. The process is reversible and is called thermal dissociation.Examples :With increase in the number of molecules, the volume increases (pressure remaining constant) and, in consequence, the density decreases. As the temperature rises, more and more dissociation takes place, and when practically complete dissociation occursthe density reaches its lowest limit.The extent of issociation, ice., the fraction of the total number of molecules which suffers dissociation is called the degree of dissociation. Gas density measurements can be used to determine the degree of dissociation. Let us take by general case where one molecule of a substance A splits up into n molecule of B on heating; i.e.,t = 0 at = teq a x nx Total no. of moles Observed molecular weight or molar mass of the mixtureQ.x (degree of dissociation) varies with D/d in the above reaction according to :a)b)c)d)Correct answer is option 'B'. Can you explain this answer? tests, examples and also practice JEE tests.

|
Explore Courses for JEE exam
|

|
Suggested Free Tests
Signup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.























