Class 9 Exam > Class 9 Questions > Valency of hydrogen is 1 and that of sulphur ...
Start Learning for Free
Valency of hydrogen is 1 and that of sulphur is 2. What should be the formula of hydrogen sulphide?
- a)HS
- b)H2S2
- c)HS2
- d)H2S
Correct answer is option 'D'. Can you explain this answer?
| FREE This question is part of | Download PDF Attempt this Test |
Verified Answer
Valency of hydrogen is 1 and that of sulphur is 2. What should be the ...
Hydrogen sulfide is the chemical compound with the chemical formula H2S. It is a colorless gas with the characteristic foul odor of rotten eggs. It is very poisonous, corrosive, and flammable.
View all questions of this test
Most Upvoted Answer
Valency of hydrogen is 1 and that of sulphur is 2. What should be the ...
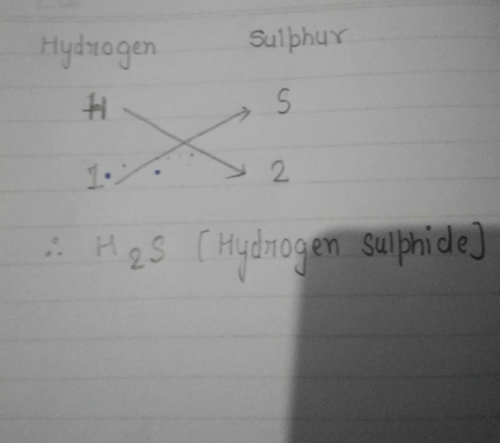
Free Test
FREE
| Start Free Test |
Community Answer
Valency of hydrogen is 1 and that of sulphur is 2. What should be the ...
See- Hydrogen's valency is +1 and sulphur's valency is -2. So we know that we need +2 so that we can make -2 zero. So if we will take two hydrogen then the total valency of hydrogen atoms will be +2 which will cancel -2 of sulphur and make it zero. Therefore d is the correct option - H2S - formula of hydrogen sulphide which contains two atoms of hydrogen and one atom of sulphur.
Attention Class 9 Students!
To make sure you are not studying endlessly, EduRev has designed Class 9 study material, with Structured Courses, Videos, & Test Series. Plus get personalized analysis, doubt solving and improvement plans to achieve a great score in Class 9.

|
Explore Courses for Class 9 exam
|

|
Similar Class 9 Doubts
Valency of hydrogen is 1 and that of sulphur is 2. What should be the formula of hydrogen sulphide?a)HSb)H2S2c)HS2d)H2SCorrect answer is option 'D'. Can you explain this answer?
Question Description
Valency of hydrogen is 1 and that of sulphur is 2. What should be the formula of hydrogen sulphide?a)HSb)H2S2c)HS2d)H2SCorrect answer is option 'D'. Can you explain this answer? for Class 9 2024 is part of Class 9 preparation. The Question and answers have been prepared according to the Class 9 exam syllabus. Information about Valency of hydrogen is 1 and that of sulphur is 2. What should be the formula of hydrogen sulphide?a)HSb)H2S2c)HS2d)H2SCorrect answer is option 'D'. Can you explain this answer? covers all topics & solutions for Class 9 2024 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for Valency of hydrogen is 1 and that of sulphur is 2. What should be the formula of hydrogen sulphide?a)HSb)H2S2c)HS2d)H2SCorrect answer is option 'D'. Can you explain this answer?.
Valency of hydrogen is 1 and that of sulphur is 2. What should be the formula of hydrogen sulphide?a)HSb)H2S2c)HS2d)H2SCorrect answer is option 'D'. Can you explain this answer? for Class 9 2024 is part of Class 9 preparation. The Question and answers have been prepared according to the Class 9 exam syllabus. Information about Valency of hydrogen is 1 and that of sulphur is 2. What should be the formula of hydrogen sulphide?a)HSb)H2S2c)HS2d)H2SCorrect answer is option 'D'. Can you explain this answer? covers all topics & solutions for Class 9 2024 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for Valency of hydrogen is 1 and that of sulphur is 2. What should be the formula of hydrogen sulphide?a)HSb)H2S2c)HS2d)H2SCorrect answer is option 'D'. Can you explain this answer?.
Solutions for Valency of hydrogen is 1 and that of sulphur is 2. What should be the formula of hydrogen sulphide?a)HSb)H2S2c)HS2d)H2SCorrect answer is option 'D'. Can you explain this answer? in English & in Hindi are available as part of our courses for Class 9.
Download more important topics, notes, lectures and mock test series for Class 9 Exam by signing up for free.
Here you can find the meaning of Valency of hydrogen is 1 and that of sulphur is 2. What should be the formula of hydrogen sulphide?a)HSb)H2S2c)HS2d)H2SCorrect answer is option 'D'. Can you explain this answer? defined & explained in the simplest way possible. Besides giving the explanation of
Valency of hydrogen is 1 and that of sulphur is 2. What should be the formula of hydrogen sulphide?a)HSb)H2S2c)HS2d)H2SCorrect answer is option 'D'. Can you explain this answer?, a detailed solution for Valency of hydrogen is 1 and that of sulphur is 2. What should be the formula of hydrogen sulphide?a)HSb)H2S2c)HS2d)H2SCorrect answer is option 'D'. Can you explain this answer? has been provided alongside types of Valency of hydrogen is 1 and that of sulphur is 2. What should be the formula of hydrogen sulphide?a)HSb)H2S2c)HS2d)H2SCorrect answer is option 'D'. Can you explain this answer? theory, EduRev gives you an
ample number of questions to practice Valency of hydrogen is 1 and that of sulphur is 2. What should be the formula of hydrogen sulphide?a)HSb)H2S2c)HS2d)H2SCorrect answer is option 'D'. Can you explain this answer? tests, examples and also practice Class 9 tests.

|
Explore Courses for Class 9 exam
|

|
Suggested Free Tests
Signup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.
























