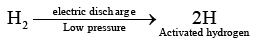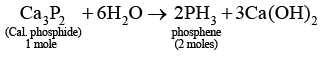JEE Advanced (Single Correct Type): Hydrogen | Chapter-wise Tests for JEE Main & Advanced PDF Download
Q.1. Which of the following is formed by the action of water on sodium peroxide?
(a) H2
(b) N2
(c) O2
(d) CO2
Correct Answer is option (c)
Na2O2 + H2O → 2NaOH + 1/2O2
Q.2. The melting points of most of the solid substances increase with an increase of pressure acting on them. However, ice melts at a temperature lower than its usual melting point when the pressure is increased. This is because
(a) pressure generates heat
(b) the chemical bonds break under pressure
(c) ice is less denser than water
(d) ice is not a true solid
Correct Answer is option (c)
On increasing pressure, coordination number of ions in solids increases thus thereby increasing their melting point but in ice pressure does not increase the coordination number of oxygen & melting point does not increase but decrease under pressure.
Q.3. Activated hydrogen is obtained by
(a) electrolysis of heavy water
(b) reaction of water with heavy metals
(c) thermal decomposition of water
(d) passing silent electric discharge through hydrogen gas at low pressure
Correct Answer is option (d)
Hence, (D) is correct answer.
Q.4. The metal which gives hydrogen on treatment with acid as well as sodium hydroxide is
(a) Fe
(b) Zn
(c) Cu
(d) none of these
Correct Answer is option (b)
Zn + H2SO4 → ZnSO4 + Na2ZnO + H2
Zn + 2NaOH → Na2ZnO2 + H2
Hence (B)
Q.5. Polyphosphates are used as water softening agents because they
(a) Form soluble complexes with anionic species
(b) Precipitate anionic species
(c) Forms soluble complexes with cationic species
(d) Precipitate cationic species
Correct Answer is option (c)
Polyphosphates (sodium hexametaphosphates, sodium tripolyphosphate or STPP) from soluble complexes with Ca+2, Mg+2 present in hard water.
Q.6. When the same amount of zinc is treated separately with excess of sulphuric acid and excess of sodium hydroxide, the ratio of volumes of hydrogen evolved is
(a) 1: 1
(b) 1: 2
(c) 2: 1
(d) 9: 4
Correct Answer is option (a)
Zn + H2SO4 → ZnSO4 + H2
Zn + 2NaOH → Na2ZnO2 + H2
The ratio of volumes of H2 evolved in both the cases is 1: 1.
Q.7. The critical temperature of water is higher than that of O2 because H2O molecule has
(a) Fewer electrons than oxygen
(b) Two covalent bonds
(c) V-shape
(d) Dipole moment
Correct Answer is option (d)
Critical temperature of water is more than O2 due to its dipole moment (Dipole moment of water = 1.84 D; Dipole moment of O2 = zero D) .
Q.8. The reaction: Ag2O + H2O2 → 2Ag + H2O + O2 takes place in
(a) basic medium
(b) bleaching agent
(c) neutral medium
(d) both in acidic and basic medium
Correct Answer is option (d)
H2O2 on oxidation gives O2 only in basic medium.
Q.9. One mole of calcium phosphide on reaction with excess water gives
(a) One mole of phosphine
(b) Two moles of phosphoric acid
(c) Two moles of phosphine
(d) One mole of phosphorus pentaoxide
Correct Answer is option (c)
Q.10. When zeolite, which is hydrated sodium aluminium silicate, is treated with hard water the sodium ions are exchanged with
(a) H+ ions
(b) Ca2+ ions
(c) Mg2+ ions
(d) Both Ca2+ and Mg2+
Correct Answer is option (d)
Zeolite when treated with hard water exchange Cu+2 and Mg+2 ions (present in hard water) with Na+ ions.
Q.11. Pick the odd one out
(a) Sodium borohydride reacts very slowly with cold water.
(b) Sodium borohydride reacts very violently with cold water to produce H 2 .
(c) Solubility of sodium borohydride in water at 25°C is 10.05 g/mL.
(d) Melting point of sodium borohydride is 500°C.
Correct Answer is option (b)
Reaction of NaBH4 with cold water is very slow. All other statements except (b) are correct.
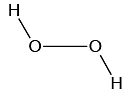
Q.12. Which of the following is the true structure of H2O2?
(a) 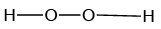
(b) 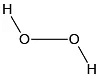
(c) 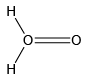
(d) 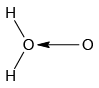
Correct Answer is option (b)
Oxygen atom is sp3 hybridized having two large pair of electrons. Therefore the structure of hydrogen peroxide is
Q.13. Hydrogen will not reduce
(a) Heated cupric oxide
(b) Heated ferric oxide
(c) Heated stannic oxide
(d) Heated aluminium
Correct Answer is option (d)
H will not reduce heated Al2O3 .
Q.14. A hydride of nitrogen which is acidic is
(a) NH3
(b) N2H4
(c) N2H2
(d) N3H
Correct Answer is option (d)
N3H (hydrazoic acid) is the acidic hydride of nitrogen
Q.15. HCl is added to following oxides. Which one would give H2O2
(a) MnO2
(b) PbO2
(c) BaO
(d) None of these
Correct Answer is option (d)
MnO2 , PbO2 and BaO will not give H2O2 with HCl.MnO2 and PbO2 will give Cl2 and BaO will react with HCl to give BaCl2 and water.
Q.16. Saline hydrides react explosively with water, such fires can be extinguished by
(a) Water
(b) Carbon dioxide
(c) Sand
(d) None of these
Correct Answer is option (c)
Fire due to action of water on saline hydrides cannot be extinguished with water or CO2. These hydrides can reduce CO2 at high temperature to produce O2.
Q.17. Which of the following can not be oxidized by H2O2 ?
(a) KI + HCl
(b) O3
(c) PbS
(d) Na 2SO3
Correct Answer is option (c)
O3 is more powerful oxidizing agent than H2O2 . So H2O2 reduces O3 to O2.
O2 + H2O2 → H2O + 2O2
Q.18. D2O is preferred to H2O , as a moderator, in nuclear reactors because
(a) D2O slows down fast neutrons better
(b) D2O has high specific heat
(c) D2O is cheaper
(d) None of these
Correct Answer is option (d)
H2O absorbs neutrons more than D2O and this decreases the number of neutrons for the fission process.
Q.19. Heavy water is a compound of
(a) hydrogen& heavier isotope of oxygen
(b) heavier isotope of hydrogen & heavier isotope of oxygen
(c) oxygen& heavier isotope of hydrogen
(d) none of the above.
Correct Answer is option (d)
D2O is heavy water.
Q.20. When electric current is passed through an ionic hydride in the molten state
(a) hydrogen is liberated at the anode
(b) hydrogen is liberated at the cathode
(c) no reaction takes place
(d) hydride ion migrates towards cathode
Correct Answer is option (d)
Ionic hydride contains H- ion which liberates H2 at the anode.
|
481 docs|964 tests
|