NCERT Exemplar: Matter in Our Surroundings | Science Class 9 PDF Download
Multiple Choice Answer Type Questions
Q.1. Which one of the following sets of phenomena would increase on raising the temperature?
(a) Diffusion, evaporation, compression of gases
(b) Evaporation, compression of gases, solubility
(c) Evaporation, diffusion, expansion of gases
(d) Evaporation, solubility, diffusion, compression of gases
Ans. (c)
Rate of evaporation, diffusion and expansion of gases increase with increase in temperature.
Q.2. Seema visited a Natural Gas Compressing Unit and found that the gas can be liquefied under specific conditions of temperature and pressure. While sharing her experience with friends she got confused. Help her to identify the correct set of conditions.
(a) Low temperature, low pressure
(b) High temperature, low pressure
(c) Low temperature, high pressure
(d) High temperature, high pressure
Ans. (c)
Gases can be compressed under low temperature and high pressure. Under these conditions the particles of gases come closer and liquefy.
Q.3. The property to flow is unique to fluids. Which one of the following statements is correct?
(a) Only gases behave like fluids.
(b) Gases and solids behave like fluids.
(c) Gases and liquids behave like fluids.
(d) Only liquids are fluids.
Ans. (c)
Gases and liquids flow due to less intermolecular forces in the molecules. Gases and liquids take the shape of the container in which they are put.
Q.4. During summer, water kept in an earthen pot becomes cool because of the phenomenon of
(a) Diffusion
(b) Transpiration
(c) Osmosis
(d) Evaporation.
Ans. (d)
Earthen pot has small pores through which water keeps evaporating and evaporation causes cooling.
Q.5. A few substances are arranged in the increasing order of ‘forces of attraction’ between their particles. Which one of the following represents a correct arrangement?
(a) Water, air, wind
(b) Air, sugar, oil
(c) Oxygen, water, sugar
(d) Salt, juice, air
Ans. (c)
Forces of attraction between the particles increase in the order of: Gases < Liquids < solids
Hence, the correct arrangement is oxygen, water, sugar.
Q.6. On converting 25°C, 38°C and 66°C to Kelvin scale, the correct sequence of temperature will be
(a) 298 K, 311 K and 339 K
(b) 298 K, 300 K and 338 K
(c) 273 K, 278 K and 543 K
(d) 298 K, 310 K and 338 K
Ans. (a)
K = 25°C + 273
Hence 25°C = 273 + 25 = 298 K
38°C = 273 + 38 = 311 K
66°C = 273 + 66 = 339 K
Q.7. Choose the correct statement of the following.
(a) Conversion of solid into vapours without passing through the liquid state is called vapourisation.
(b) Conversion of vapours into solid without passing through the liquid state is called sublimation.
(c) Conversion of vapours into solid without passing through the liquid state is called freezing.
(d) Conversion of solid into liquid is called sublimation.
Ans. (b)
Conversion of vapours into solid without passing through the liquid state is called sublimation.
Q.8. The boiling points of diethyl ether, acetone and n-butyl alcohol are 35°C, 56°C and 118°C respectively. Which one of the following correctly represents their boiling points in Kelvin scale?
(a) 306 K, 329 K, 391 K
(b) 308 K, 329 K, 392 K
(c) 308 K, 329 K, 391 K
(d) 329 K, 392 K, 308 K
Ans. (c)
35°C = 273 + 35 = 308 K
56°C = 273 + 56 = 329 K
118°C = 273 + 118 = 391 K
Q.9. Which condition out of the following will increase the evaporation of water?
(a) Increase in temperature of water
(b) Decrease in temperature of water
(c) Less exposed surface area of water
(d) Adding common salt to water
Ans. (a)
Rate of evaporation increases with increase in temperature of water.
Q.10. In which of the following conditions, the distance between the molecules of hydrogen gas would increase?
(i) Increasing pressure on hydrogen contained in a closed container.
(ii) Some hydrogen gas leaking out of the container.
(iii) Increasing the volume of the container of hydrogen gas.
(iv) Adding more hydrogen gas to the container without increasing the volume of the container.
(a) (i) and (iii)
(b) (i) and (iv)
(c) (ii) and (iii)
(d) (ii) and (iv)
Ans. (c)
(ii) If some hydrogen gas is leaked from the container the remaining gas will occupy the whole space and the distance between the molecules will increase.
(iii) If the volume of the container is increased, same number of molecules will occupy that space. Hence, the distance between the molecules will increase.
Short Answer Type Questions
Q.11. A sample of water under study was found to boil at 102°C at normal temperature and pressure. Is the water pure? Will this water freeze at 0°C?
Ans. The water is not pure, as pure water boils at 100°C at normal temperature and pressure (1 atm). Due to impurities, it will likely freeze at a temperature below 0°C.
Q.12. A student heats a beaker containing ice and water. He measures the temperature of the content of the beaker as a function of time. Which of the following (figure) would correctly represent the result?
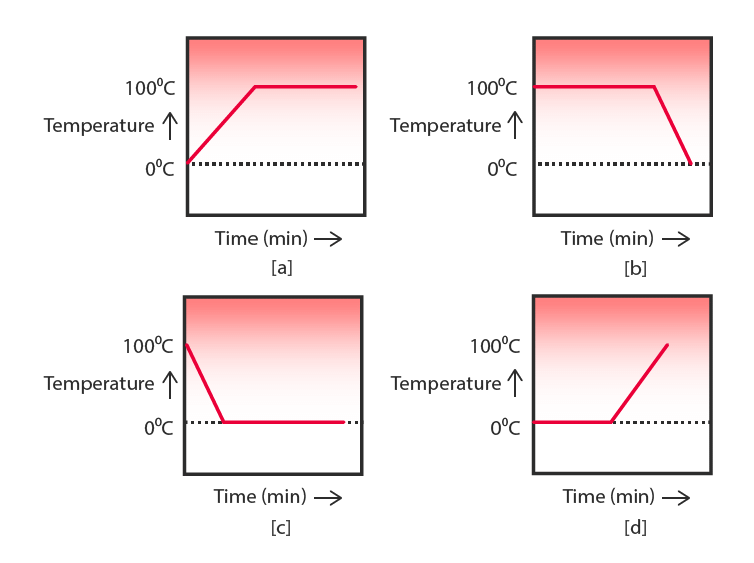 Ans. Ice and water both are present at 0°C. The temperature will remain constant till the whole ice changes into water because ice will melt by taking latent heat of fusion. On further heating, the temperature of water will increase. So, the correct option is (d).
Ans. Ice and water both are present at 0°C. The temperature will remain constant till the whole ice changes into water because ice will melt by taking latent heat of fusion. On further heating, the temperature of water will increase. So, the correct option is (d).
Q.13. Fill in the blanks:
(a) Evaporation of a liquid at room temperature leads to a——— effect.
(b) At room temperature the forces of attraction between the particles of solid substances are———than those which exist in the gaseous state.
(c) The arrangement of particles is less ordered in the ——— state. However, there is no order in the ——— state.
(d) ——— is the change of solid state directly to vapour state without going through the ———state.
(e) The phenomenon of change of a liquid into the gaseous state at any temperature below its boiling point is called———.
Ans.
(a) Cooling
(b) Stronger
(c) Liquid, gaseous
(d) Sublimation, liquid
(e) Evaporation
Q.14. Match the physical quantities given in column A to their SI units given in column B:
| Column (A) | Column (B) |
| (a) Pressure | (i) cubic metre |
| (b) Temperature | (ii) kilogram |
| (c) Density | (iii) pascal |
| (d) Mass | (iv) kelvin |
| (e) Volume | (v) kilogram per cubic metre |
Ans. (a) - (iii), (b) - (iv), (c) - (v), (d) - (ii),(e) - (i)
Q.15. The non-SI and SI units of some physical quantities are given in column A and column B respectively. Match the units belonging to the same physical quantity:
| Column (A) | Column (B) |
| (a) degree celsius | (i) kilogram |
| (b) centimetre | (ii) pascal |
| (c) gram per centimetre cube | (iii) metre |
| (d) bar | (iv) kelvin |
| (e) milligram | (v) kilogram per metre cube |
Ans. (a) - (iv), (b) - (iii), (c) - (v), (d) - (ii), (e) - (i)
Q.16. ‘Osmosis is a special kind of diffusion’. Comment.
Ans. Diffusion in liquids and gases is the movement of particles from low concentration to high concentration. Osmosis is the movement of particles from the lower concentration to higher concentration through the semipermeable membrane. This movement happens due to diffusion. Hence Osmosis is a special kind of diffusion.
Q.17. Classify the following into osmosis/diffusion
(a) Swelling up of raisins on keeping in water.
(b) Spreading of virus on sneezing.
(c) Earthworms dying on coming in contact with common salt.
(d) Shrinking of grapes kept in thick sugar syrup.
(e) Preserving pickles in salt.
(f) Spreading of the smell of cake being baked throughout the house.
(g) Aquatic animals using oxygen dissolved in water during respiration.
Ans.
(a) Osmosis
Reason: Concentration of water will be more than water concentration in raisin. Hence there is a movement of water from higher concentration to lower concentration which will make the raisin swell up.
(b) Diffusion
Reason: Microscopic viruses get diffused in air.
(c) Osmosis
Reason: Because of difference in concentration of water and solute. Water comes out of the earthworm body causing its cell to burst causing its death.
(d) Osmosis
Reason: Thick sugar syrup has more solutes in it than grapes. Hence water moves out of the grapes from higher concentration to lower concentration hence grapes swell.
(e) Osmosis
Reason: Salt helps to remove water from the cells and helps to preserve pickles. Pickling is intended to keep out oxygen. Pickling prevents oxidation of fruits and vegetables through the use of salt. Dry salt forms pickling brine(aqueous sodium chloride) and absorbs excess moisture from fruits and vegetables through the process of osmosis.
(f) Diffusion
Reason: Particles containing smell diffuse into air to spread it throughout the house
(g) Diffusion
Reason: When Aquatic animals breathe dissolved oxygen, oxygen diffuses directly into their blood without any membrane.
Q.18. Water as ice has a cooling effect whereas water as steam may cause severe burns. Explain these observations.
Ans. Water as ice has less energy and water molecules absorb energy from our body to give cooling effect whereas steam has high energy and transfers high energy into our body which transforms as heat to cause burns.
Q.19. Alka was making tea in a kettle. Suddenly, she felt intense heat from the puff of steam gushing out of the spout of the kettle. She wondered whether the temperature of the steam was higher than that of the water boiling in the kettle.
Ans. The temperature of the steam is not higher than that of the boiling water; both are at 100°C at standard atmospheric pressure. However, steam causes severe burns because it releases a large amount of latent heat of vaporization when it condenses on the skin, transferring more energy than boiling water at the same temperature.
Q.20. A glass tumbler containing hot water is kept in the freezer compartment of a refrigerator (temperature < 0°C). If you could measure the temperature of the content of the tumbler, which of the following graphs would correctly represent the change in its temperature as a function of time?
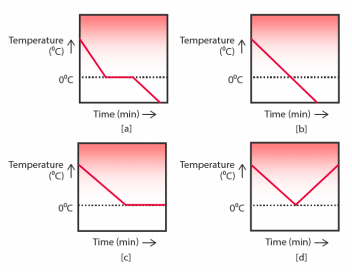 Ans. (a) is the correct graph.
Ans. (a) is the correct graph.
The temperature of hot water decreases to 0°C and remains constant due to latent heat of fusion that is involved in formation of ice and then temperature decreases further.
Q.21. Look at the figures and suggest in which of the vessels A, B, C or D the rate of evaporation will be the highest? Explain.
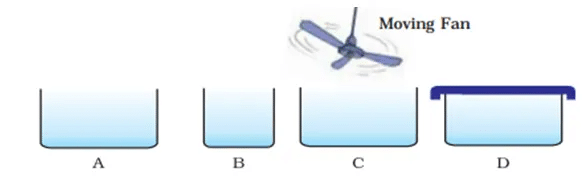 Ans. C is the correct figure. The rate of evaporation will be highest because it has a large open surface area and the wind of moving fan will also increase the rate of evaporation. Greater the surface area and more the wind speed, higher will be the rate of evaporation.
Ans. C is the correct figure. The rate of evaporation will be highest because it has a large open surface area and the wind of moving fan will also increase the rate of evaporation. Greater the surface area and more the wind speed, higher will be the rate of evaporation.
Q.22. (a) Conversion of solid to vapour is called sublimation. Name the term used to denote the conversion of vapour to solid.
(b) Conversion of solid-state to liquid state is called fusion. What is meant by latent heat of fusion?
Ans.
(a) It is called deposition.
(b) The amount of heat energy released or absorbed when a solid changes to liquid at atmospheric pressure at its melting point is known as the latent heat of fusion.
Long Answer Type Questions
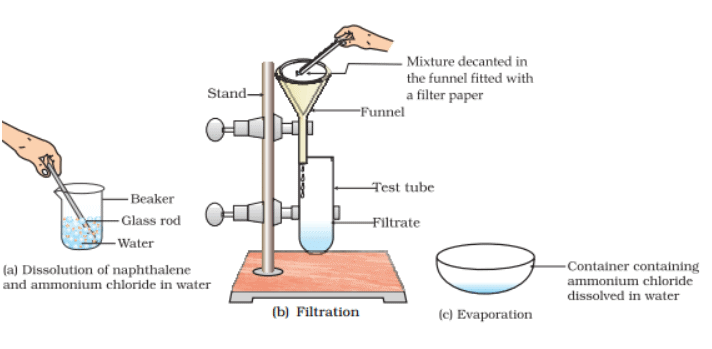
Q.23. You are provided with a mixture of naphthalene and ammonium chloride by your teacher. Suggest an activity to separate them with well labelled diagram.
Ans. Naphthalene is insoluble in water but soluble in benzene (organic solvent). Ammonium chloride dissolves in water but insoluble in benzene. Naphthalene changes into vapours at room temperature whereas, ammonium chloride changes into vapours on heating. Add water to the mixture and shake it vigorously so that ammonium chloride will dissolve. Filter the mixture. Naphthalene is obtained as residue whereas, filtrate contains ammonium chloride dissolved in water. Crystallise it by heating till saturated solution of ammonium chloride is obtained. Cool the hot saturated solution to get crystals of pure ammonium chloride.
Q.24. It is a hot summer day. Priyanshi and Ali are wearing cotton and nylon clothes respectively. Who do you think would be more comfortable and why?
Ans. Cotton is more comfortable because it can absorb water (sweat) more than nylon followed by evaporation which causes cooling. Secondly, cotton clothes have pores through which air can pass through. Therefore, Priyanshi is more comfortable.
Q.25. You want to wear your favourite shirt to a party, but the problem is that it is still wet after a wash. What steps would you take to dry it faster?
Ans. Conditions that can increase the rate of evaporation of water are:
(i) Spread out the shirt so as to increase the surface area so that water can be evaporated faster.
(ii) Increase the wind speed by switching on the fan.
(iii) If wind speed is high outside and sunlight is present, dry it outside. It will dry faster due to higher temperature and higher wind speed. If sunlight is not available, use hot iron to dry it.
Q.26. Comment on the following statements:
(a) Evaporation produces cooling.
(b) Rate of evaporation of an aqueous solution decreases with increase in humidity.
Ans.
(a) In evaporation, surface molecules take heat from surroundings and cause a cooling effect.
(b) If humidity is high, then air already has lot of water vapours, it will not take more water vapours easily. Therefore, the rate of evaporation will be slow.
Q.27. Why does the temperature of a substance remain constant during its melting point or boiling point?
Ans. The temperature of a substance remains constant during melting and boiling points till the completion of melting and boiling because of latent heat of fusion used by the substances. Latent heat of fusion helps to overcome the force of attraction between particles of solid to change into liquid when they melt. Hence temperature remains constant. In the same way during formation of vapours latent heat helps solid substance to convert into gaseous state. Hence temperature of a substance remain constant at boiling point.
|
84 videos|478 docs|60 tests
|
FAQs on NCERT Exemplar: Matter in Our Surroundings - Science Class 9
| 1. What is the physical state of matter in our surroundings? |  |
| 2. How do solids differ from liquids and gases? |  |
| 3. What is the process of conversion of a solid into a liquid called? |  |
| 4. Can gases be compressed easily? |  |
| 5. What happens when a gas is cooled down? |  |






















