Solved Examples for JEE: Thermodynamics | Chemistry for JEE Main & Advanced PDF Download
Example 1. In which of the paths between initial state i and final state f in the below figure is the work done on the gas the greatest?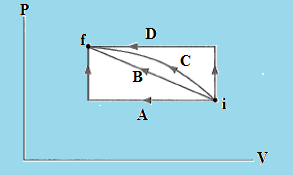
(A) A
(B) B
(C) C
(D) D
Solution. The correct option is (D).
Work is a path function. So work done on the gas depends upon the path. The area under pv diagram gives the work done on the gas between initial state i and final state f. As the area under the path D is greater than the other path, therefore the paths between initial state i and final state f where the work done on the gas is greatest would be D. From the above observation we conclude that, option D is correct.
Example 2. A 1-kg block of ice at 0ºC is placed into a perfectly insulated, sealed container that has 2 kg of water also at 0ºC. The water and ice completely fill the container, but the container is flexible. After some time one can except that
(A) the water will freeze so that the mass of the ice will increase.
(B) the ice will melt so that the mass of the ice will decrease.
(C) both the amount of water and the amount of ice will remain constant.
(D) both the amount of water and the amount of ice will decrease.
Solution: The correct option is (B) the ice will melt so that the mass of the ice will decrease.
In accordance to second law of thermodynamics, if an irreversible process occurs in a closed system, the entropy of that system always increases; it never decreases. Entropy measures the state of disorder. As the ice is more order than water, therefore the ice will melt so that the mass of the ice will decrease. From the above observation we conclude that, option (B) is correct.
Example 3. Which type of ideal gas will have the largest value for Cp – Cv?
(A) Monoatomic
(B) Diatomic
(C) Polyatomic
(D) The value will be the same for all.
Solution:
The correct option is (D).
The specific heat at constant volume (Cv) for monoatomic gas is,
Cv = 3/2 R
The specific heat at constant volume (Cv) for diatomic gas is,
Cv = 5/2 R
The specific heat at constant volume (Cv) for polyatomic gas is,
Cv = 3 R
But we know that, for an ideal gas,
Cp - Cv = R
So,the specific heat at constant pressure (Cp) for monoatomic gas will be,
Cp = Cv + R = 3/2 R + R = 5/2 R
the specific heat at constant pressure (Cp) for diatomic gas will be,
Cp = Cv + R = 5/2 R + R = 7/2 R
the specific heat at constant pressure (Cp) for polyatomic gas will be,
Cp = Cv + R = 3 R + R = 4 R
Therefore,
Cp - Cv for monoatomic gas will be,
Cp - Cv = 5/2 R - 3/2 R = R
Cp - Cv fo diatomic gas will be,
Cp - Cv = 7/2 R - 5/2 R = R
Cp - Cv fo polyatomic gas will be,
Cp - Cv = 4 R - 3 R = R
From the above observation we conclude that, the value of Cp - Cv will be same for all ideal gas. Therefore option (D) is correct.
Example 4. Show that pressure of a fixed amount of an ideal gas is a state function 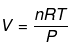
Solution.
=
Example 5. Calculate work done for the expansion of a substance 3m3 to 5m3 against.
(a) Constant pressure = 105 Pa
(b) A veriable pressure = (10 + 5V) Pa
Solution.
(a) W = -PdV = -105(5-3) = -2 ×105 J
(b)
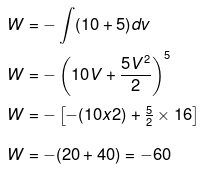
Example 6. Calculate change in internal energy for a gas under going from state-I
(300 K, 2 ×10-2 m3) to state -II (400 K, 4 ×10-2 m3) for one mol. of vanderwaal gas.
(a) If gas is ideal [Cv = 12 J/K/mol]
(b) If gas is real
Cv = 12 J/k/ mol
a = 2 J.m./mol2
Solution.
(a) DU = nCV(T2 - T1)
= 1 × 12 ×100 = 1200 J
(b)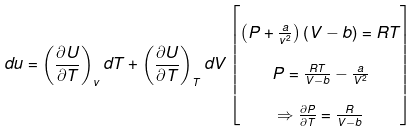
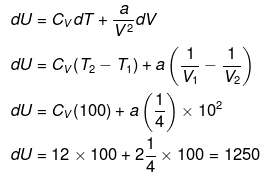
Example 7. One mole of an ideal gas is expanded isothermally at 300 K from 10 atm to 1 atm. Calculate q, w, DU & DH under the following conditions.
(i) Expansion is carried out reversibly.
(ii) Expansion is carried out irreversibly
Solution.
Isothermal process
(i) For ideal gas ΔU = 0, ΔH = 0
q = - w
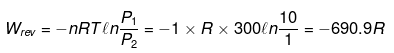
(ii)
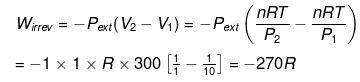
Example 8. Calculate work done for an ideal gas (ln 2 = 0.7)
Solution.
w = -nRT ln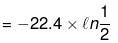
= 22.4 × 0.7 = 15.68
Example 9. Calculate w = ? (ln 2 = 0.7)
Solution.
P - V Relation from plot
y = mx + C
1 = m + C .............(1)
4 = 2m + C .............(2)
⇒ m = 3, C = -2
Hence,
Example 10. One mole an ideal gas is expanded from (10 atm, 10 lit) to (2 atm, 50 litre) isothermally. First against 5 atm then against 2 atm. Calculate work done in each step and compare it with single step work done with 2 atm.
Solution.
(i) P1V1 = P2V2
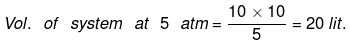 wirrev = - Pext(V2 - V1) = - 5 (20 - 10) = - 50 atm lit.
wirrev = - Pext(V2 - V1) = - 5 (20 - 10) = - 50 atm lit.
(ii) Work done against 2 atm
P1 V1 P2 V2
5 atm 20 lit 2 atm 50 lit
wirrev = - Pext(V2 - V1) = - 2 (50 - 20) = - 60 atm lit.
wtotal = - 50 - 60 = - 110 atm lit.
P1 V1 P2 V2
10 atm 10 lit 2 atm 50 lit
w = - 2 (50 - 10) = - 80 lit
magnitude of work done in more than one step is more than single step work done.
Example 11. For 1 mole of monoatomic gas. Calculate w, DU, DH, q
Solution.
Isochoric process
w = 0
q = dU = Cv (T2 - T1)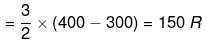
DH = CpΔT = R (400 - 300) = 250 R
Example 12. One mole of an non linear triatomic ideal gas is expanded adiabatically at 300 K from 16 atm to 1 atm.
Calculate Work done under the following conditions.
(i) Expansion is carried out reversibly.
(ii) Expansion is carried out irreversibly
Solution.
q = 0
w = DU = Cv(T2 - T1)
Cv for triatomic non linear gas = 3R
(i) For rev. process. , r = 4/3
ΔU = w = 3R (150 - 300) = - 450 R
(ii) n = 1
- Pext (V2 - V1) = Cv (T2 - T1)
-16T2 T1 = 48 T2 - 48 T1
49 T1 = 64 T2
T2 = 229.69
wirr = Cv(T2 - T1) 3R (229.69 - 300)
= - 210.93 R
Example 13. One mole of an non linear triatomic ideal gas is compressed adiabatically at 300 K from 1 atom to 16 atm.
Calculate Work done under the following conditions.
(i) Expansion is carried out reversibly. (ii) Expansion is carried out irreversibly.
Solution.
q = 0 Adiabatic process
(i)
wrev = DU = Cv(T2 - T1)
w = 3R (600 - 300) = 900 R
(ii)
- (T2 - 16 T1) = 3R (T2 - T1)
- T2 16 T1 = 3T2 - 3T1
4T2 = 19 T1
w = ΔU = 3R (1425 - 300) = 3375 R
Example 14. Calculate work done in process BC for 1 mol of an ideal gas if total 600 cal heat is released by the gas in whole process.
Solution.
For cyclic process
dU = 0
q = - w
PV1 =1× R × 300
PV2 =1× R × 400
⇒ P(V2 - V1) = R (400 - 300)
q = - (wBA + wAC + wCB)
- 600 = - (0 + (-) 2) × (400 - 300) + wCB
wCB = 800 cal
Example 15. Calculate entropy change in each step for an ideal gas (monoatomic)
Solution.
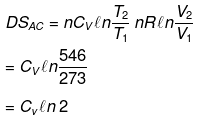
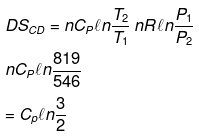
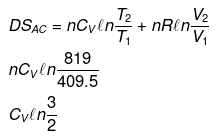
Example 16. One mole of an ideal gas is expanded isothermally at 300 K from 10 atm to 1 atm. Find the values of
ΔSsys, ΔSsurr & ΔStotal under the following conditions.
(i) Expansion is carried out reversibly.
(ii) Expansion is carried out irreversibly
(iii) Expansion is free.
Solution.
(i)
(ii)
ΔU = 0 = q + w
qirr = pext)(v2 - v1)
(iii) Free expansion ΔT = 0
w = 0
q = 0
Example 17. One mole of an non linear triatomic ideal gas is expanded adiabatically at 300 K from 16 atm to 1 atm. Find the values of DSsys, DSsurr & DStotal under the following conditions.
(i) Expansion is carried out reversibly.
(ii) Expansion is carried out irreversibly
(iii) Expansion is free.
Solution.
For non-linear tri-atomic ideal gas
Cv = 3R, Cp = 4R
(i)
q = 0
ΔSsurr = - ΔSsys = 0
ΔStotal = 0
(ii) First of all we will have to calculate the temperature of the gas after it has undergoes the said adiabatic reversible expansion we have q = 0
ΔU = q + w
nCv (T2 - T1) = - pext (v2 - v1)
T2 = 229.68 K
= - 1.068 R + 2.77 R
= 1.702 R
ΔStotal =ΔSsys = 1.702 R
(iii) In free adiabatic expansion we have
w = 0
q = 0
ΔT = 0
Example 18. For the reaction
N2 + 2O2 → 2NO2
Given : at 1 atm, 300 K
SN2 = 180 J /mol/K Cp(N2) = 30 J/mol/K
SO2 = 220 J /mol/K Cp(O2) = 30 J/mol/K
SNO2 = 240 J /mol/K Cp(NO2) = 40 J/mol/K
Calculate
(i) ΔS300 K, 1 atm
(ii) ΔS400 K, 1 atm
(iii) ΔS300 K, 5 atm
(iv) ΔS400 K, 5 atm
Solution.
(i) (ΔSr)300 = 2SNO2 - 2SO2 - SN2
= 2 × 240 - 2 × 220 - 180
= -140 J mol-1k-1
(ΔCp)r = 2Cp(NO2)- 2Cp(O2) - Cp(N2)
= 2 × 40 - 2 × 30 - 30
= -10 J mol-1k-1
(ii) (ΔSr)400 = (ΔSr)300 (ΔCp)r ln
= - 140 - 10 ln (4/3)
= - 142.88 J mol-1k-1
(iii) (ΔSr)300k, 5 atm
= (ΔSr)300k, 1 atm ΔngRln
= - 140 (-1)Rln (1/5)
= - 140 R ln 5
=- 140 8.314 ln 5
= - 126.62 J mol-1k-1
(iv) (ΔSr)400k, 5 atm
= (ΔSr)400k, 1 atm - R ln
= 142.88 R ln 5
=-129.5 J mol-1k-1
Example 19. From the T-S diagram of a reversible carnot engine calculate:
(i) efficiency
(ii) workdone per cycle
(iii) Heat taken from the source and rejected to sink
(iv) In order to illuminate 10000 bulbs of 40 watt power each calculate the no. of cycle per second the above must go through.
Solution.
(i)
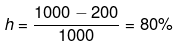
(ii) qrev1-2 = ∫Tds = 1000 x 100 J
q3-4 = ∫Tds = - 200 x 100 J
qnet = 800 × 100 = 80 kJ
W = -80 kJ
(iii) 105 J,
2 × 104 J
(iv) 5
Example 120. Calculate entropy change
H2O(l, 1 atm, 100ºC) → H2O (g, 1 atm, 110ºC)
H2O(l, 1 atm, 100ºC) → H2O (g, 2 atm, 100ºC)
ΔHvap = 40 kJ/mol Cp(l) = 75 J/mol /K Cp(g) = 35 J/mol/K
Solution.
For 1 mol
(i) H2O(l, 1 atm, 100ºC) → H2O (g, 1 atm, 100ºC) → H2O(g, 1 atm, 110ºC)
(A) (B) (C)
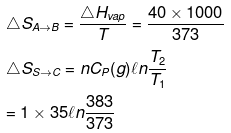
(ii) H2O(l, 1 atm, 100ºC) → H2O (g, 1 atm, 100ºC) → H2O(g, 2 atm, 100ºC)
(A) (B) (C)
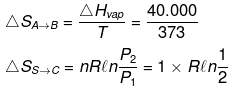
|
361 videos|821 docs|301 tests
|
FAQs on Solved Examples for JEE: Thermodynamics - Chemistry for JEE Main & Advanced
| 1. What is the first law of thermodynamics and how does it apply to closed systems? |  |
| 2. How do you calculate work done in an isothermal process? |  |
| 3. What are the differences between isothermal, adiabatic, and isochoric processes? |  |
| 4. What is the significance of the Carnot cycle in thermodynamics? |  |
| 5. How can you determine the change in enthalpy for a chemical reaction? |  |






















