Shankar IAS Summary: Ocean Acidification
Ocean Acidification

Oceans act as significant reservoirs for atmospheric carbon dioxide (CO2), absorbing roughly one-third of the CO2 produced by human activities. This uptake moderates the rate of atmospheric warming but alters seawater chemistry in a process called ocean acidification. Ocean acidification refers to a long-term decrease in the average pH of ocean surface waters and related changes in carbonate chemistry driven by the absorption of CO2 from the atmosphere.
When CO2 dissolves in seawater it reacts with water to form carbonic acid and products that increase the concentration of free hydrogen ions (H+), thereby lowering pH and reducing the concentration of carbonate ions (CO32-). Reduced carbonate ion availability and lower alkalinity impair organisms that build calcium carbonate structures and change biogeochemical cycles in the ocean.
CO2 effect on ocean acidification
- The ocean absorbs substantial amounts of atmospheric CO2, but the current rate of human emissions is faster than many natural processes can compensate for.
- Carbon dioxide (CO2) transferred from the atmosphere to the ocean is the primary driver of recent ocean acidification.
- Surface ocean waters have become measurably more acidic over the industrial era.
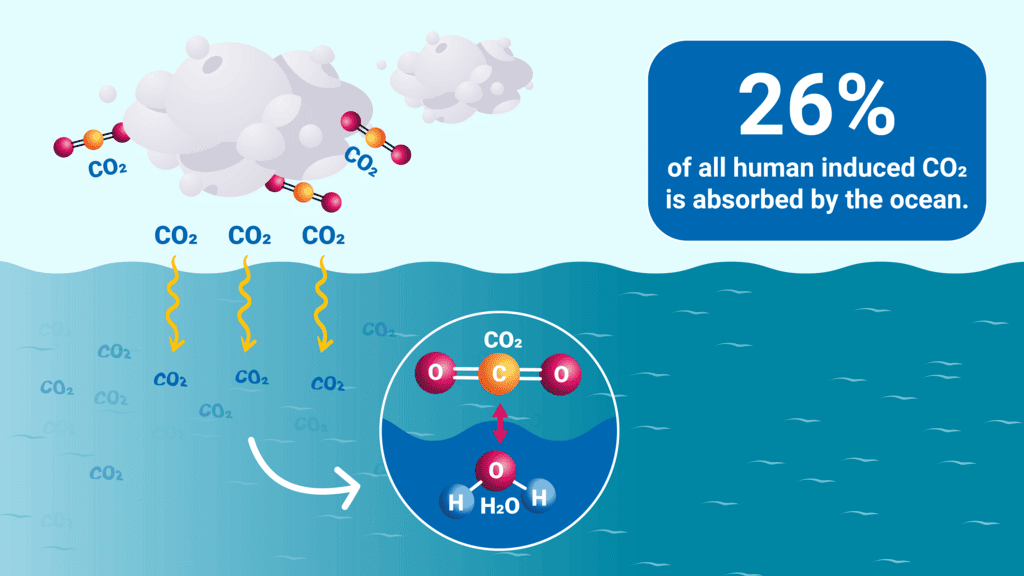 CO2 effect on ocean acidification
CO2 effect on ocean acidification- Since the start of the industrial revolution, the average surface ocean pH has fallen by about 0.1 unit.
- Present-day surface ocean pH remains slightly alkaline (around pH 8.0), but the trend is toward lower pH values.
- The term "ocean acidification" emphasises the directional shift toward greater acidity even though seawater is not becoming a strong acid.
- A decrease of 0.1 pH unit corresponds to approximately a 26% increase in the concentration of free hydrogen ions (H+) in seawater, altering chemical equilibria important to marine life.
Local influences on ocean acidification
Acid Rain

- Effect: Acid rain (precipitation with low pH) can locally change the chemistry of surface waters where it falls, temporarily increasing acidity at coastal interfaces.
- Extent: These effects are typically local or regional; globally, atmospheric CO2 uptake remains the dominant driver of ocean acidification.
Eutrophication

- Cause: Excess nutrients (mainly nitrogen and phosphorus) from agriculture, sewage and runoff fuel phytoplankton blooms in coastal waters.
- Result: When algal blooms collapse, bacterial decomposition consumes oxygen and produces CO2, locally lowering pH and contributing to acidification and hypoxia in coastal zones.
Chemical reactions that underlie ocean acidification
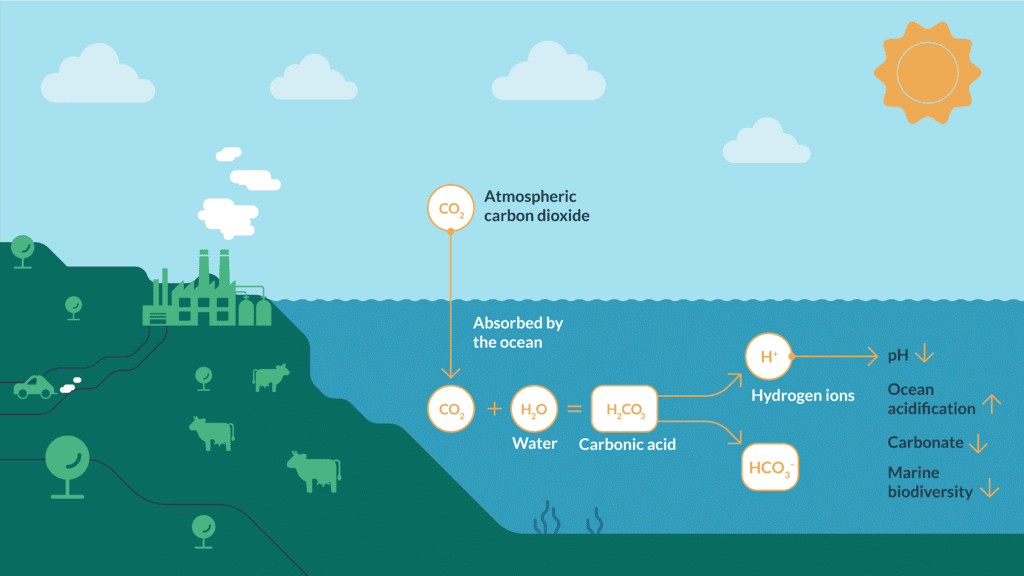
- Formation of carbonic acid and dissociation: CO2 + H2O → H2CO3 → H+ + HCO3-
 Chemical Reaction involved in ocean acidification
Chemical Reaction involved in ocean acidificationThis reaction produces hydrogen ions that increase acidity and generate bicarbonate.
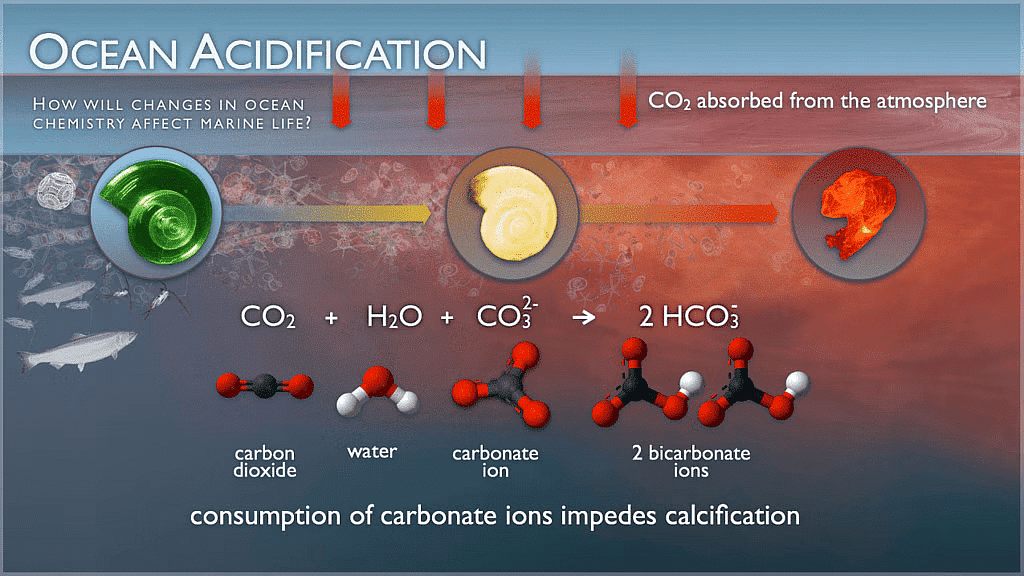
- Interaction with carbonate ions: CO32- + CO2 + H2O → 2 HCO3-
This shifts carbonate into bicarbonate, reducing available carbonate ions needed for calcifying organisms.
Effect of ocean acidification
- Seawater and CO2: Dissolved CO2 forms carbonic acid and related carbon species (bicarbonate HCO3- and carbonate CO32-), changing the buffer chemistry of the ocean.
- Importance of carbonate ions: Carbonate ions (CO32-) are essential for calcifying organisms (corals, molluscs, some plankton, echinoderms) to form shells and skeletons of calcium carbonate (CaCO3).
- Problem with more CO2: Elevated atmospheric CO2 increases the concentration of H+ in seawater, shifting equilibria toward bicarbonate and reducing CO32- availability.
- Consequences for calcification: Reduced carbonate ion concentrations make it more energetically costly for organisms to calcify; some species may form thinner or weaker shells, grow more slowly, or suffer higher mortality.
- Ecological and economic impact: Impacts on shell-forming organisms cascade through food webs and affect fisheries and ecosystems that provide food, coastal protection and livelihoods.
Mitigation
- Reduce CO2 emissions: The most effective long-term measure is to cut anthropogenic CO2 at source by reducing fossil fuel use.
- Government policies: Support and implement policies that limit greenhouse gas emissions, promote decarbonisation and encourage carbon sequestration where appropriate.
- Limit offshore fossil fuel activities: Reducing activities that can increase CO2 emissions or locally alter marine chemistry (for example, some forms of offshore extraction) may help mitigate local risks.
- Energy efficiency and conservation: Improving energy efficiency across sectors reduces demand and associated CO2 emissions.
- Switch to low-carbon energy: Increasing deployment of renewable energy (wind, solar, hydropower) and other low-emission technologies reduces future CO2 inputs to the ocean.

Saturation horizons
The saturation horizon is the depth in the water column below which calcium carbonate minerals (aragonite and calcite) begin to dissolve because seawater is under-saturated with respect to those minerals.
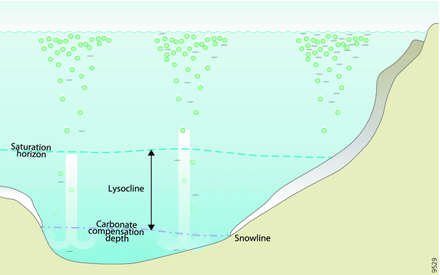 Saturation horizon
Saturation horizon- Deep, cold waters: Cold, deep ocean waters naturally contain less carbonate ion and are often corrosive to CaCO3; shells sinking into these waters may dissolve.
- Surface waters: Warmer, surface waters generally have higher carbonate concentrations and are supersaturated with respect to shell minerals, enabling calcification.
- Organism strategies: Some deeper-water organisms have physiological or chemical mechanisms to protect shells from dissolution where waters are near or below saturation.
- Effect of acidification: As the ocean absorbs more CO2, the saturation horizons for aragonite and calcite shoal (move upward), exposing more organisms and habitats to corrosive conditions.
- Historical change: Compared with pre-industrial times (for example, the 1800s), the saturation horizons for calcite and aragonite have moved closer to the surface, increasing biological vulnerability.
Ocean acidification and the short- and long-term fate of carbon
Long-term fate of carbon
- Natural balance: Over very long timescales (tens to hundreds of thousands of years), geological processes (volcanic outgassing, rock weathering, sediment burial) regulate atmospheric CO2 and ocean carbon.
- Rock weathering: Chemical weathering of rocks consumes CO2 on geologic timescales, but it is too slow to offset century-scale anthropogenic emissions.
Upwelling
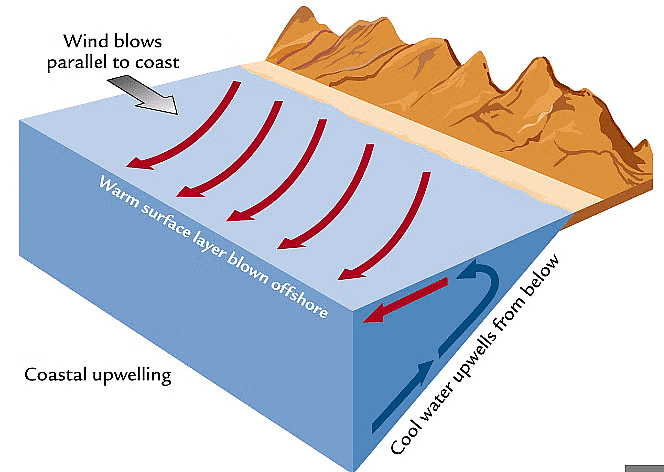 Coastal Upwelling
Coastal Upwelling- Definition: Upwelling brings deep water rich in nutrients and CO2 to the surface, supporting productivity but locally elevating CO2 and reducing pH.
- Effect of acidification: With surface waters becoming less saturated in carbonate, upwelled waters-already high in CO2-may pose greater stress to calcifiers in coastal upwelling regions.
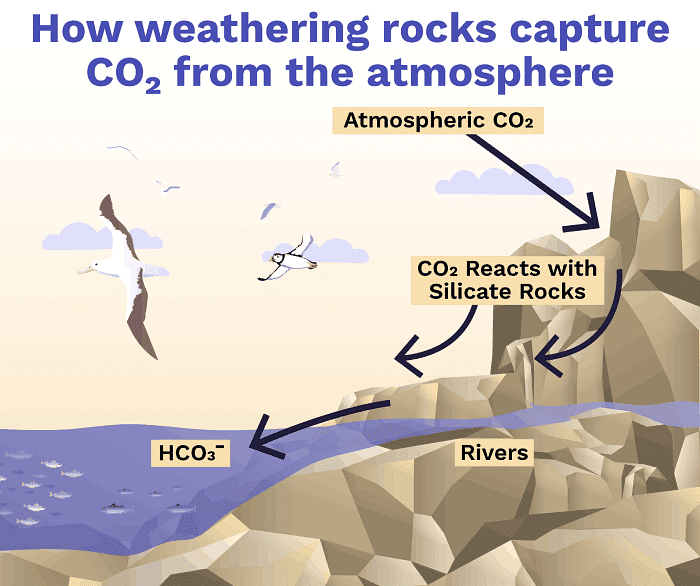 Rock Weathering
Rock Weathering- Implication: Upwelling regions can show early and strong biological responses to acidification due to the combined effect of natural CO2-rich deep water and anthropogenic surface CO2 uptake.
Short-term carbon cycling and sediment interactions
- Internal feedbacks: On timescales of centuries to a few thousand years, ocean carbonate chemistry and sedimentary processes provide partial buffering via dissolution and reprecipitation of CaCO3.
- Ocean layers: The upper ocean tends to be a region where carbonate can remain without dissolving; deeper layers are where dissolution predominates.
- Lysocline: The lysocline is the depth interval where the rate of CaCO3 dissolution strongly increases; below this the carbonate compensation depth (CCD) is where little CaCO3 is preserved in sediments.
- Sinking shells: Shells and tests from marine organisms sink to the seabed. In shallow regions they are more likely to be buried and preserved; in deep regions they often dissolve and do not sequester carbon long term.
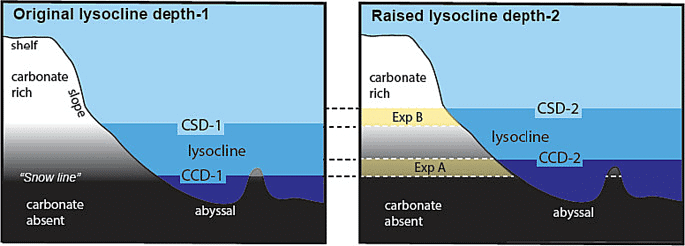 Carbonate Compensation Depth (CCD)
Carbonate Compensation Depth (CCD)- Impact of acidification: Increasing CO2 shoals the lysocline and CCD, exposing more sinking CaCO3 to dissolution. This process can act as a slow negative feedback on ocean pH, but the timescales involved (centuries to millennia) are long compared with the pace of modern emissions.
Effects on marine plants and plankton
 Effect of ocean acidification on marine life
Effect of ocean acidification on marine life- Some species may benefit: Certain photosynthetic organisms, including some phytoplankton and seagrasses, can respond positively to higher CO2 because CO2 is a substrate for photosynthesis.
- Responses vary: Increased CO2 does not uniformly benefit all marine plants; species-specific physiology, nutrient availability, light and acidification stress determine net outcomes.
- Complex community effects: Shifts in phytoplankton composition and productivity may alter food webs, biogeochemical cycles and fisheries in ways that are not always predictable from single-species responses.
- Importance of mitigation: To limit negative effects on marine ecosystems, reducing atmospheric CO2 is essential; local management (nutrient reduction, habitat protection) can also reduce vulnerability in coastal areas.
Key terms and concepts (concise)
- Ocean acidification: Long-term decrease in ocean pH due to uptake of atmospheric CO2.
- pH: A measure of hydrogen ion concentration; small changes in pH correspond to large changes in H+ concentration.
- Carbonate chemistry: The system of CO2, H2CO3, HCO3- and CO32- that buffers seawater and controls calcification.
- Calcification: The biological process of forming calcium carbonate (CaCO3) skeletons and shells.
- Saturation horizon / lysocline / CCD: Depths that mark where calcium carbonate begins to dissolve and where preservation in sediments effectively stops.
- Upwelling: The upward movement of deep, nutrient- and CO2-rich water to the surface, with local effects on pH and biology.
Concluding note
Ocean acidification is a global change in seawater chemistry primarily driven by anthropogenic CO2 emissions. It reduces carbonate availability, stresses calcifying organisms, alters ecosystems and interacts with local stresses such as eutrophication and upwelling. Effective mitigation requires cutting CO2 emissions combined with local measures (nutrient management, habitat protection and fisheries management) to enhance resilience while science continues to monitor, model and understand species- and region-specific responses.
FAQs on Shankar IAS Summary: Ocean Acidification
| 1. What is ocean acidification and how is it caused by CO2? |  |
| 2. What are some local influences on ocean acidification? |  |
| 3. How does ocean acidification affect marine life? |  |
| 4. What are some mitigation strategies for ocean acidification? |  |
| 5. How does ocean acidification impact the fate of carbon in the system? |  |







