Class 10 Science: CBSE Sample Question Paper (2020-21) - 6 | Science Class 10 PDF Download
Class-X
Science Theory
TIME: 3 Hrs.
M.M: 80
General Instructions:
Read the following instructions very carefully and strictly follow them :
1. The question paper comprises four sections A, B, C and D. There are 36 questions in the question paper.
All questions are compulsory.
2. Section-A - Question no. 1 to 20 - all questions and parts thereof are of one mark each. These questions contain multiple-choice questions (MCQs), very short answer questions and assertion - reason type questions. Answers to these should be given in one word or one sentence.
3. Section-B - Question no. 21 to 26 are short answer type questions, carrying 2 marks each. Answers to these questions should be in the range of 30 to 50 words.
4. Section-C - Question no. 27 to 33 are short answer type questions, carrying 3 marks each. Answers to these questions should be in the range of 50 to 80 words.
5. Section-D - Question no. - 34 to 36 are long answer type questions carrying 5 marks each. Answers to these questions should be in the range of 80 to 120 words.
6. There is no overall choice. However, internal choices have been provided in some questions. A student has to attempt only one of the alternatives in such questions.
7. Wherever necessary, neat and properly labelled diagrams should be drawn.
SECTION A
Q.1. Why electrical wires are made up of copper.
Ans. Copper is a very good conductor of electricity.
Q.2. The refractive index of diamond is 2.42. What is the meaning of this statement ? (1 Mark)
Ans. The refractive index of diamond 2.42 suggests that the speed of light in diamond will reduce by a factor 2.42 as compared with its speed in air.
Q.3. What is genetics ? (1 Mark)
Ans. Genetics is the branch of biology that deals with the study of genes and heredity in organisms.
Q.4. Incomplete combustion of coal and petroleum :
(A) increases air pollution.
(B) increases efficiency of machines.
(C) reduces global warming.
(D) produce poisonous gases.
The correct option is :
(a) (A) and (B)
(b) (A) and (D)
(c) (B) and (C)
(d) (C) and (D)
Ans. b
Q.5. The compound obtained on reaction of iron with steam is/are:
(a) Fe2O3
(b) Fe3O4
(c) FeO
(d) Fe2O3 and Fe3O4
Ans. b
Solution.
3Fe(s) + 4H2O(g) → Fe3O4 (s) + 4H2(g)
Q.6. An element ‘X’ reacts with O2 to give a compound with a high melting point. This compound is also soluble in water. The element ‘X’ is likely to be :
(a) iron
(b) calcium
(c) carbon
(d) silicon
Ans. b
Solution.
Calcium reacts with oxygen and form CaO which has high melting point and is water soluble.
Q.7. Name the parts of a bisexual flower that are not directly involved in reproduction. (1 Mark)
Ans. Sepals/calyx
Petals/corolla
Thalamus
OR
State the number of male gametes produced by each pollen grain.
Ans. Each pollen grain produces two male gametes.
Q.8. Where does aerobic respiration occur in a cell ? (1 Mark)
Ans. Aerobic respiration occurs in mitochondria of the cell.
Q.9. Name the physical quantity which is same in all the resistors when they are connected in series. (1 Mark)
Ans. Electric current.
OR
Should the resistance of a voltmeter be low or high? (1 Mark)
Ans. High. In parallel connection, less current passes through high resistance.
Q.10. Study the following ray diagram :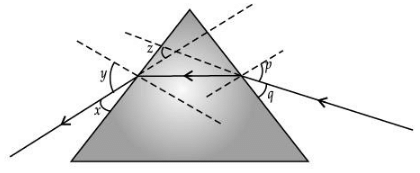
In this diagram, the angle of incidence, the angle of emergence and the angle of deviation respectively have been represented by (1 Mark)
(a) y, p and z
(b) x, q and z
(c) p, y and z
(d) p, z and y
Ans. (c)
Solution.
y, p and z. The angle between the incident ray and the normal is known as the angle of incidence, and the angle between the emergent ray and the normal is known as the angle of emergence. The emergent ray is bent at an angle with the direction of the incident ray. This angle is called the angle of deviation.
Q.11. Various steps in a food chain represent : (1 Mark)
(a) Food web
(b) Trophic level
(c) Ecosystem
(d) Biomagnification
Ans. (b) Trophic level
OR
Which of the following statement is incorrect ?
(a) All green plants and blue green algae are producers.
(b) Green plants get their food from organic compounds.
(c) Producers prepare their own food from inorganic compounds.
(d) Plants convert solar energy into chemical energy.
Ans. (b) Green plants get their food from organic compounds.
Explanation : Producers capture the solar energy and convert it into chemical energy. All green plants and certain blue-green algae, which can produce food by photosynthesis come under this category and are called producers.
Q.12. Directions : For question number 12, two statements are given- one labelled Assertion (A) and the other labelled Reason (R). Select the correct answer to these questions from the codes (a), (b), (c) and (d) as given below : (1 Mark)
Assertion (A) : Surgical methods are most effective methods of contraception.
Reason (R) : Surgical method blocks gametes transport and hence prevent fertilization.
Options~
(a) Both A and R are true and R is correct explanation of the assertion.
(b) Both A and R are true but R is not the correct explanation of the assertion.
(c) A is true but R is false.
(d) A is false but R is true
Ans. (a) Both A and R are true and R is correct explanation of the assertion.
Explanation : Surgical method like vasectomy in male and tubectomy in female prevent pregnancy. These methods block gamete transport and hence prevent fertilization. They are very effective but reversibility is very poor.
Q.13. Choose the incorrect statement from the following regarding magnetic lines of field ? (1 Mark)
(a) The direction of magnetic field at a point is taken to be the direction in which the north pole of a magnetic compass needle points.
(b) Magnetic field lines are closed curves.
(c) If magnetic field lines are parallel and equidistant, they represent zero field strength.
(d) Relative strength of magnetic field is shown by the degree of closeness of the field lines.
Ans. (c)
Solution.
If magnetic field lines are parallel and equidistant, they represent zero field strength.
Magnetic field lines appear parallel when they are far from the magnet. But this does not mean that field strength is zero. No field line would be present where field strength becomes zero.
Q.14. Which of the following property of a proton can change while it moves freely in a magnetic field ? (There may be more than one correct answer.) (1 Mark)
(a) Mass
(b) Speed
(c) Velocity
(d) Momentum
Ans. (c) and (d)
Solution.
When a proton enters a magnetic field, it starts moving on a circular path. Because of its movement along a circular path it attains angular momentum. We know that momentum is a product of mass and velocity. Therefore velocity and mass of a proton change when it enters a magnetic field.
Q.15. In an ecosystem, 10% of energy available for transfer from one trophic level to the next is in the form of : (1 Mark)
(a) heat energy
(b) chemical energy
(c) mechanical energy
(d) light energy
Ans. b
Q.16. Soil fertility determined by its ability to: (1 Mark)
(a) Decay organic matter
(b) Hold organic matter
(c) Hold water
(d) Support life
Ans. d
Q.17. For question numbers 17, two statements are given – one labelled Assertion (A) and the other labelled Reason (R).
Select the correct answer to these questions from the codes (a), (b), (c) and (d) as given below:
Assertion (A) : A solar cooker cooks the meal due to green house effect.
Reason (R) : The plane mirror is responsible for producing the green house effect. (1 Mark)
Options~
(a) Both A and R are true and R is correct explanation of the Assertion.
(b) Both A and R are true but R is not the correct explanation of the Assertion.
(c) A is true but R is false.
(d) A is false but R is true.
Ans. c
Solution.
A solar cooker cooks the meal due to greenhouse effect. The glass sheet that covers the box is responsible for producing the greenhouse effect. Plane mirror maximize the amount of light entering the solar cooker.
Q.18. For question numbers 18, two statements are given- one labelled Assertion (A) and the other labelled Reason (R).
Select the correct answer to these questions from the codes (a), (b), (c) and (d) as given below :
Assertion : A geneticist crossed a pea plant having violet flowers with a pea plant with white flowers, he got all violet flowers in first generation.
Reason : White colour gene is not passed on to next generation (1 Mark)
Options~
(a) Both A and R are true, and R is correct explanation of the assertion.
(b) Both A and R are true, but R is not the correct explanation of the assertion.
(c) A is true, but R is false.
(d) A is false, but R is true
Ans. b
Q.19. When sodium hydrogen carbonate is added to ethanoic acid a gas evolves. Consider the following statements about the gas evolved. (1 Mark)
(A) It turns lime water milky.
(B) It is evolved with a brisk effervescence.
(C) It has a smell of burning sulphur.
(D) It is also a by-product of respiration.
The correct statements are :
(a) (A) and (B) only
(b) (B) and (D) only
(c) (A), (C) and (D)
(d) (A), (B) and (D)
Ans. d
Solution.
The gas evolved is carbon dioxide with brisk effervescence. It turns lime water milky. It is also a by-product of respiration.
Q.20. Directions : Q. No 20 contain five sub-parts each. You are expected to answer any four subparts in these question. (1 Mark each)
Read the following and answer any four questions.
The activities of man had adverse effects on all forms of living organisms in the biosphere.
Unlimited exploitation of nature by man disturbed the delicate ecological balance between the living and non-living components of the biosphere. The unfavorable conditions created by man himself threatened the survival not only of him but also of the entire living organisms on the mother earth. One of your classmates is an active member of ‘Eco club’ of your school which is creating environmental awareness amongst the school students, spreading the same in the society and also working hard for preventing environmental degradation of the surroundings.
(a) It is necessary to conserve our environment. Which of these is the reason to conserve our environment? (1 Mark)
(i) To save water, air and soil from pollution.
(ii) To maintain ecological balance.
(iii) To save ozone layer
(iv) All of these
Ans. (iv) All of these
(b) The green and blue dustbin signifies for: (1 Mark)
(i) Biodegradable waste and non-biodegradable waste respectively.
(ii) Non-biodegradable waste and biodegradable waste respectively.
(iii) Eco-friendly waste
(iv) Domestic waste only
Ans. (i) Green dustbins is for biodegradable waste and blue dustbins is for non-biodegradable waste.
(c) Why are plastics difficult to recycle? (1 Mark)
(i) Because it is a very hard material
(ii) Because of the different sizes of plastic
(iii) Because it is very adhesive in its nature
(iv) Because of different types of polymer resins
Ans. (iv) Because of different types of polymer resins.
(d) In the following groups of materials, which group(s) contains only non-biodegradable items? (1 Mark)
(I) Wood, paper, leather
(II) Polythene, detergent, PVC
(III) Plastic, detergent, grass
(IV) Plastic, bakelite, DDT
Options~
(i) (III)
(ii) (IV)
(iii) (I) and (III)
(iv) (II) and (IV)
Ans. (iv)
Solution.
Substances which are not broken down by the bacteria or saprophytes are called non-biodegradable substances. Under certain conditions, the non-biodegradable substances can persist for longer time and can also harm the various components of our ecosystem.
(e) Which of these non-biodegradable wastes generated daily in kitchen can be recycled? (1 Mark)
(i) Polythene bags
(ii) Plastic containers
(iii) Vegetable waste
(iv) Both (i) and (ii)
Ans. (iii) Polythene bags and plastic containers.
SECTION B
Q.21. Why is Tyndall effect shown by colloidal particles? State four instances of observing the Tyndall effect. (2 Mark)
Ans. Tyndall effect is shown by colloidal particles because the colloidal particles size are roughly equal to the wavelength of the light.
Four instances of observing tyndall effect are:
(i) In fog
(ii) When light passes through canopy in forest.
(iii) Blue colour of the sky
(iv) When light passes through the milk
Q.22. Draw a labelled diagram to show
(i) reddish appearance of the sun at the sunrise or the sunset and
(ii) white appearance of the sun at noon when it is overhead. (2 Mark)
Ans. (i) Reddish appearance of the sun at the sunrise and sunset :
At sun-rise and sun-set most of the blue light and shorter wavelengths are scattered away by the particles in the atmosphere as the light from the sun near the horizon passes through thick layers of air and larger distance. The light that reaches us is of longer wavelength (red colour) giving a reddish appearance.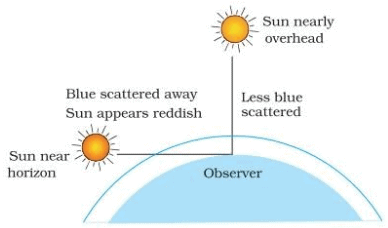 (ii) White appearance of the sun at noon when it is overhead: At noon, the sun is nearly overhead. The sunlight has to pass through much smaller portion of Earth's atmosphere. The scattering is much less and the Sun looks white.
(ii) White appearance of the sun at noon when it is overhead: At noon, the sun is nearly overhead. The sunlight has to pass through much smaller portion of Earth's atmosphere. The scattering is much less and the Sun looks white.
Q.23. A V-I graph for a nichrome wire is given below. What do you infer from this graph? Draw a labelled circuit diagram to obtain such a graph. (2 Mark)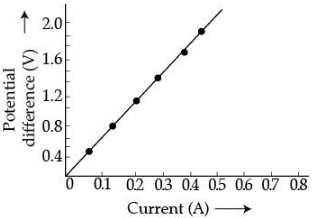 Ans.
Ans.
Graph between V and I is a straight line. So, this infers that the flow of current (I) in the conductor is directly proportional to the potential diffidence (V) established across it. This is ohm's law.
Resistance of the wire can be calculated as: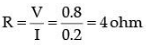
This means nichrome wire has a constant value of the resistance 4 ohm.
Circuit diagram: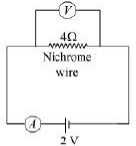
Q.24. Write the mathematical expression for Joule’s law of heating. (2 Mark)
Ans. Mathematical expression of Joule's law of heating is :
H = I2Rt
Where, H = Produced Heat
I = Current flowing through the device
t = Time taken
R = Resistance of the appliance
Q.25. List in tabular form two differences between binary fission and multiple fission. (2 marks)
Ans. Difference between binary fission and multiple fission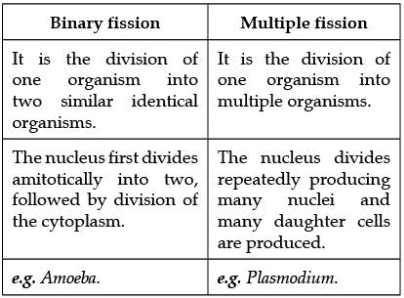
Q.26. What happens when a mature Spirogyra filament attains considerable length. (2 Mark)
Ans. Spirogyra reproduces asexually by fragmentation. In this, the body breaks up into two or more small pieces of fragments upon maturation. These fragments grow into new Spirogyra.
SECTION C
Q.27. Differentiate between a glass slab and a glass prism. What happens when a narrow beam of (i) a monochromatic light, and (ii) white light passes through (a) glass slab and (b) glass prism? (3 Mark)
Ans.
Glass slab | Glass prism |
It is rectangular in shape. | It has two triangular sides, two inclined rectangular sides and one rectangular base. |
In this, the direction of incident ray and emergent ray of the light are parallel to each other. | In this, the direction of incident ray and emergent ray of the light are not parallel to each other. |
When a narrow beam of monochromatic light passes through:
(a) Glass slab : It has deviated from the actual path but the direction of the incident ray and the emergent ray are parallel to each other.
(b) Glass of prism : It has deviated from the actual path but the direction of the incident ray and the emergent ray are not parallel to each other.
When a narrow beam of white light passes through :
(a) Glass slab, it does not split into its constituent colour. The direction of incident ray and emergent ray of light are parallel to each other.
(b) Glass prism, it splits into its constituent seven colour. The direction of incident ray and emergent ray of light are not parallel to each other.
Q.28. What is ‘rusting’? Describe with a labelled diagram an activity to investigate the conditions under which iron rusts. (3 Mark)
Ans. Rusting: The process of acquiring a coating of a brown flaky substance called rust on iron when it is exposed to moist air for a long time.
Activity :
- Take three test tubes and label them as A, B and C.
- In each tube, place clean iron.
- Pour some water in test tube A and cork it.
- Pour distilled water in test tube B, add about 1 ml of oil and cork it.
- Put some anhydrous calcium chloride in test tube C and cork it.
- Leave the test tube for few days.
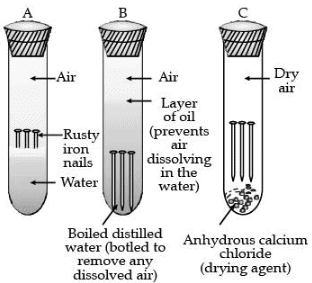
Observation: Iron nail rusts in test tube A but they do not rust in test tubes B and C.
Explanation:
- It is because, in the test tube A, the nails are exposed to both air and water.
- In test tube B, the nails are exposed to only water because the oil prevents the air from dissolving in water.
- In test tube C, the nails are exposed to dry air because CaCl2 will absorb the moisture from the air.
Conclusion : Air and water both are essential conditions for rusting.
Q.29. (a) State the relation correlating the electric current flowing in a conductor and the voltage applied across it. Also draw a graph to show this relationship.
(b) Find the resistance of a conductor if the electric current flowing through it is 0.35 A when the potential difference across it is 1.4 V. (3 Mark)
Ans. (a) The flow of current (I) in the conductor is directly proportional to the potential diffidence (V) established across it provided the physical conditions remain same.
Or V = IR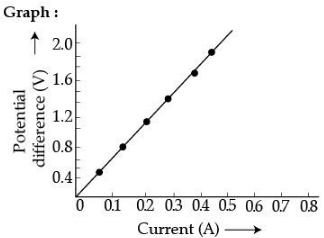
(b) Given : Potential Difference (V)= 1.4 V
Current (I) = 0.35
A As per formula, V = IR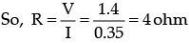
Q.30. (a) Draw the structures for (i) ethanol, (ii) ethanoic acid.
(b) Why is the conversion of ethanol to ethanoic acid considered an oxidation reaction? Write the oxidising angent used in the reaction involved. (3 Mark)
Ans. (a) Structure of ethanol :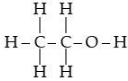 Structure of ethanoic acid :
Structure of ethanoic acid :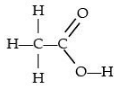
(b) Conversion of ethanol to ethanoic acid is an oxidation reaction because oxygen is added to ethanol to convert it to ethanoic acid.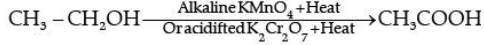
In the above reaction alk. KMnO4/acidified K2Cr2O7 add oxygen to ethanol hence they are called oxidising agent.
Q.31. The near point of the eye of a person is 50 cm. Find the nature and power of the corrective lens required by the person to enable him to see clearly the objects placed at 25 cm. from the eye. (3 Mark)
Ans. Given, Object distance, u = -25 cm
Image distance, v = -50 cm
Focal length, f =?
Using lens formula,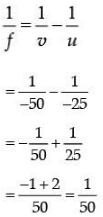
∴ f = 50 cm.
Power of the lens, 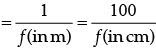
⇒ 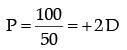
Hence, the corrective lens is convex, because power of lens is positive.
Q.32. In 1913, Henry Moseley, a young physicist from England showed that the atomic number (represented as Z) of an element is a more fundamental property than its atomic mass. We know that the atomic mass of an element is due to the mass of protons and neutrons present in the nucleus of its atom. The chemical properties of the elements are related to the number of electrons and also the distribution of electrons in the different shells.
Study the part of Modern Periodic Table given below in which the alphabets represent the symbols of elements and answer the given questions: (3 Mark)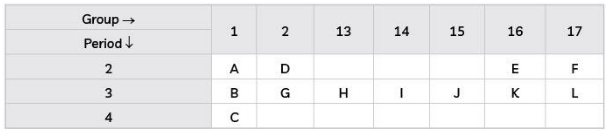
(A) Arrange the elements of 3rd period in order of decreasing metallic character.
(B) Identify the elements of period 2 and group 16 which will form diatomic molecule. Draw the electron dot structure of that element. What type of bond is formed?
Ans. (A) B > G > H > I > J > K > L
Explanation: As we move from Left to right in the periodic table, metallic character decreases and non-metallic increases.
(B) Group-16 Electronic Configuration-2, 6 Period-2
The element 'E' is Oxygen.
Oxygen has 6 valence electrons, so it requires two more electrons to complete its octet. Hence, it will share two of its electrons with two electrons of the other oxygen atom to form a diatomic molecule O2 gas.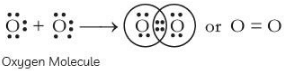
The bond formed is double covalent bond.
Q.33. Answer the following questions:
(A) Why does a current carrying conductor, experiences a force when it is placed in a magnetic field?
(B) A circuit has a Line of 5 A. How many Lamps of rating 40W; 220V can simultaneously glow on this Line safely? (3 Mark)
Ans. (A) When a current-carrying conductor is placed in a magnetic field, the wire experiences a force due to the interaction between the external magnetic field and the magnetic field produced by the moving charges in the wire.
(B) We know that,
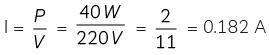
0.182 A or 2/11 A is required by one lamp.
Current rating i.e. maximum current = 5 A
No. of lamps = 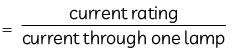
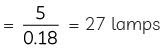
SECTION D
Q.34. (a) What is an electromagnet? List any two uses.
(b) Draw a labelled diagram to show an electromagnet is made.
(c) State the purpose of soft iron core used in making an electromagnet.
(d) List two ways of increasing the strength of an electromagnet if the material of the electromagnet is fixed. (5 Mark)
Ans. (a) Electromagnet : Magnet formed by producing magnetic field inside a solenoid.
Uses of electromagnet :
Inside TVs, sound speakers and radios.
Inside a generator to transform mechanical energy to electrical energy.
(b) Labelled diagram to show how an electromagnet is made :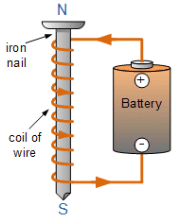
(c) Soft iron rod increases the magnetism of solenoid by a thousand fold. When the solenoid current is switched off, the magnetism is effectively switched off since the soft iron core has low retentivity.
(d) Ways to increase the strength of an electromagnet if the material of the electromagnet is fixed are :
(i) By increasing the amount of current flowing in the solenoid
(ii) By increasing the number of turns in the solenoid.
Q.35. Trace the changes that take place in a flower from gamete formation to fruit formation. (5 Mark)
Ans.
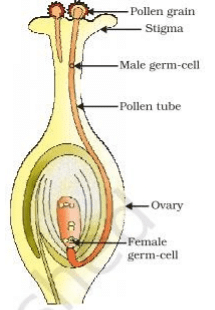
[Diagram drawn and annotated with the following points will also be considered]
(a) Stamen is the male reproductive part and it produces pollen grains.
(b) The ovary contains ovules and each ovule has an egg cell.
(c) The pollen needs to be transferred from the stamen to the stigma.
(d) If this transfer of pollen occurs in the same flower, it is referred to as self-pollination. On the other hand, if the pollen is transferred from one flower to another, it is known as cross-pollination.
(e) After the pollen lands on a suitable stigma, it has to reach the female germ-cells which are in the ovary. For this, a tube grows out of the pollen grain and travels through the style to reach the ovary/Figure
(f) The male germ-cell produced by pollen grain fuses with the female gamete present in the ovule.
(g) This fusion of the germ-cells or fertilisation gives the zygote.
(h) After fertilisation, the zygote divides several times to form an embryo within the ovule.
(i) The ovule develops a tough coat and is gradually converted into a seed. The ovary grows rapidly and ripens to form a fruit.
(j) Meanwhile, the petals, sepals, stamens, style and stigma may shrivel and fall off.
OR
In the given circuit, A, B, C and D are four lamps connected with a battery of 60V. (5 Mark)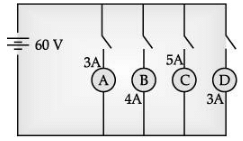
Analyse the circuit to answer the following questions.
(i) What kind of combination are the lamps arranged in (series or parallel) ?
(ii) Explain with reference to your above answer, what are the advantages (any two) of this combination of lamps ?
(iii) Explain with proper calculations which lamp glows the brightest.
(iv) Find out the total resistance of the circuit.
Ans. The lamps are in parallel.
Advantages : If one lamp is faulty, it will not affect the working of the other lamps. They will also be using the full potential of the battery as they are connected in parallel.
The lamp with the highest power will glow the brightest.
P = VI
In this case, all the bulbs have the same voltage. But lamp C has the highest current.
Hence, for lamp C, P = 5 × 60 Watt = 300 W. (the maximum).
The total current in the circuit = 3 + 4 + 5 + 3A = 15A
The voltage = 60V
V = IR and hence R = V/I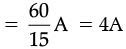
Q.36. (a) Identify the given diagram ? Name the parts 1 to 5.
(b) What is contraception ? List three advantages of adopting contraceptive measures : (5 Mark)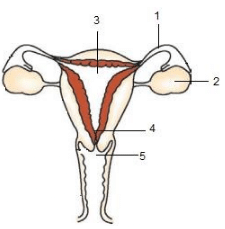 Ans. (a) Female reproductive system
Ans. (a) Female reproductive system
Name of parts :
1: Fallopian tube/Oviduct
2: Ovary
3: Uterus
4: Cervix
5: Vagina
(b) Contraception : Method to avoid pregnancy
Advantages :
(i) Proper gap between two pregnancies
(ii) Avoiding unwanted pregnancy
(iii) Keeping population under control
OR
(i) Prostate glands and seminal vesicle add their secretions so that the sperms are in a fluid (seminal fluid) and it makes their transport easier and also provides nutrition. Testes secrete testosterone which brings about changes in the appearances in the boys at the time of puberty.
(ii) Female foeticides/illegal sex selected abortion of female fetus.
(iii) Interfere in release of egg and eggs are not released.
|
80 videos|569 docs|80 tests
|
FAQs on Class 10 Science: CBSE Sample Question Paper (2020-21) - 6 - Science Class 10
| 1. What is the format of the CBSE Sample Question Paper for Class 10 Science (2020-21)? |  |
| 2. How many marks does the CBSE Sample Question Paper for Class 10 Science (2020-21) carry? |  |
| 3. Are the CBSE Sample Question Papers for Class 10 Science (2020-21) helpful for board exam preparation? |  |
| 4. Can I rely solely on the CBSE Sample Question Papers for Class 10 Science (2020-21) for exam preparation? |  |
| 5. Where can I find the CBSE Sample Question Papers for Class 10 Science (2020-21)? |  |
















