Class 9 Exam > Class 9 Notes > Science Class 9 > Mind Map: Structure of Atoms
Mind Map: Structure of Atoms | Science Class 9 PDF Download
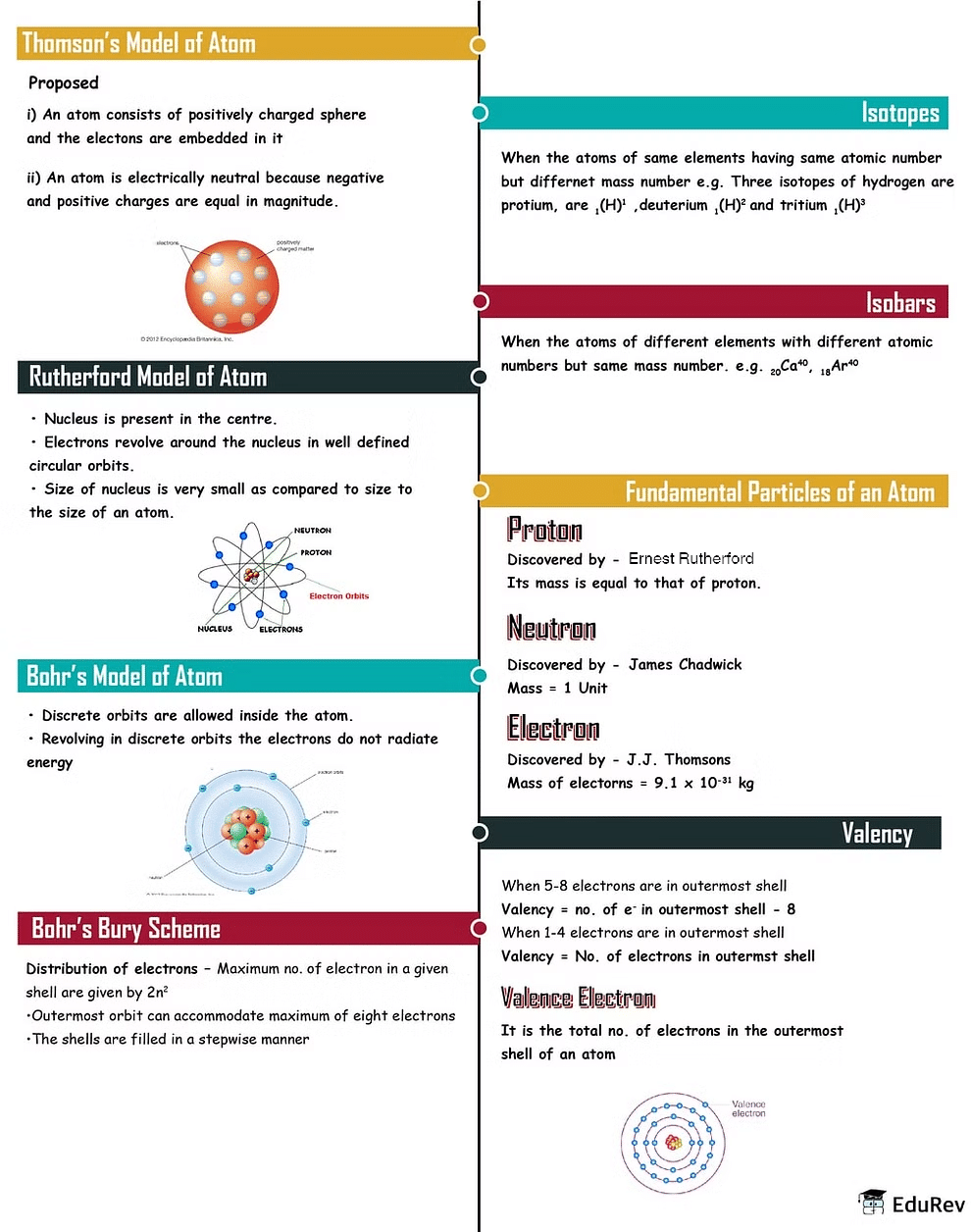
The document Mind Map: Structure of Atoms | Science Class 9 is a part of the Class 9 Course Science Class 9.
All you need of Class 9 at this link: Class 9
|
84 videos|478 docs|60 tests
|
FAQs on Mind Map: Structure of Atoms - Science Class 9
| 1. What is the structure of an atom? |  |
Ans. An atom consists of a nucleus, which contains protons and neutrons, surrounded by electrons in shells or energy levels.
| 2. How do protons, neutrons, and electrons contribute to the structure of an atom? |  |
Ans. Protons and neutrons are located in the nucleus of an atom, with protons carrying a positive charge and neutrons carrying no charge. Electrons, which carry a negative charge, orbit the nucleus in energy levels.
| 3. What is the significance of the nucleus in the structure of an atom? |  |
Ans. The nucleus is crucial in determining the identity of an atom, as it contains the protons that define the element and the neutrons that contribute to the atom's stability.
| 4. How do electrons move within the structure of an atom? |  |
Ans. Electrons move in specific energy levels or shells around the nucleus, with each shell capable of holding a specific number of electrons.
| 5. How does the structure of an atom impact its chemical properties? |  |
Ans. The number of protons, neutrons, and electrons in an atom determines its chemical behavior, influencing factors such as reactivity, stability, bonding, and overall properties of the element.
Related Searches
















