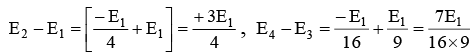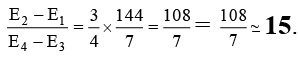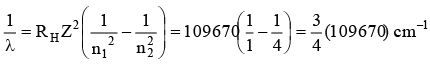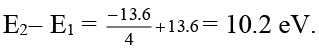Integer Answer Type Questions for JEE: Structure of Atom
Q.1. The ratio of (E2 - E1) to (E4 - E3) for the hydrogen atom is approximately equal to
Ans. 15
Q.2. Assuming Rydberg's constant (RH) to be 109670 cm-1, the longest wavelength line in the Lyman series of the hydrogen spectrum is
Ans. 1215.8 Å
λ = 1215.8 × 10-8 cm i.e.,
λ = 1215.8 Å
Q.3. The first excitation energy for hydrogen atom would be
Ans. 10.2 eV
Q.4. If electrons are excited to 4th shell in He+ ion, the number of wavelengths emitted by electrons during de-excitation are
Ans. 6
By nc2formula ie. Total number of transitions = n(n - 1)/2
Q.5. In Mn2+ ion, the number of unpaired electrons is
Ans. 5
Q.6. The radius of the first Bohr orbit of hydrogen atom (n = 1) is approximately 0.530 Å. The radius of the first excited state (n = 2) of hydrogen atom is
Ans. 2.12 Å
rn = r1 × n2 [for hydrogen atom]
rn = 0.53 × n2 Å [n = 2]
= 0.53 × 4
rn =2.12 Å.
Q.7. An element with atomic number 20 will be placed in the period of the periodic table
Ans. 4
Atomic number 20 → 1s22s22p63s23p64s2
Q.8. How many maximum quantum number can be same for two electrons in an atom.
Ans. 3
Maximum three quantum number can be same but fourth must be different.
Q.9. Find out the number of waves made by a Bohr electron in one complete revolution in its 3rd orbit.
Ans. 3
No of waves = principal quantum no.
n = 3
Q.10. How many unpaired electrons will be in 4f orbital of Gadolinium (64)?
Ans. 8