Chemistry Basics – Atoms, Molecules, Elements, Compounds, and Mixtures
Fundamentals of Chemistry
Chemistry is the scientific study of matter: its composition, structure, properties and the changes it undergoes. Matter is anything that has mass and occupies space; at the microscopic level it is composed of atoms and molecules.
Atoms
An atom is the smallest unit of an element that retains the chemical properties of that element. Atoms are extremely small and are made up of subatomic particles arranged in a characteristic structure.
- Structure: An atom has a central nucleus containing protons and neutrons, surrounded by electrons that occupy quantised energy levels or shells.
- Nucleus: The compact central core contains most of the atom's mass and is positively charged because of protons.
- Electrons: Electrons are negatively charged particles that occupy orbitals or shells around the nucleus and determine chemical behaviour.
- Subatomic particles: The principal subatomic particles are protons, neutrons and electrons; collectively they determine mass, charge and chemical properties.
Subatomic Particles
- Protons: Positively charged particles in the nucleus. Each proton carries a charge of +1 (in electronic charge units) and has a mass close to 1 atomic mass unit (amu). The number of protons equals the atomic number (Z) of an element.
- Electrons: Negatively charged particles with charge -1. An electron's mass is very small: approximately 9.11×10-31 kg. Electrons occupy energy levels and determine bonding and chemical reactivity.
- Neutrons: Electrically neutral particles in the nucleus with mass ≈1 amu. Neutrons contribute to atomic mass and nuclear stability.
- Historical discoveries: J. J. Thomson discovered the electron (1897); Ernest Rutherford's work led to discovery of the proton (Rutherford, proton identified in 1918); James Chadwick discovered the neutron (1932).
Nucleus
- Nucleons: Protons and neutrons together are called nucleons.
- Mass number (A): Total number of nucleons (A = Z + N, where N is number of neutrons).
- Isotopes: Atoms of the same element (same Z) with different numbers of neutrons (different A). Examples: carbon-12 and carbon-14.
- Ions: Atoms or groups of atoms that have lost or gained electrons and therefore carry net electrical charge: cations (positive) and anions (negative).
- Nuclear binding energy: Energy that holds the nucleus together; it affects nuclear stability and is related to mass defect.
Molecules
A molecule is an electrically neutral group of two or more atoms held together by chemical bonds. Molecules may consist of atoms of the same element or different elements.
- Examples of molecules composed of one element: O2 (oxygen), N2 (nitrogen).
- Examples of molecules composed of different elements: water (H2O), carbon dioxide (CO2), proteins (large molecules built from amino-acid units).
- Molecular formula: Shows the actual number of each atom in a molecule (for example H2O).
- Empirical formula: Simplest whole-number ratio of atoms in a compound (for example CH2 for some hydrocarbons).
Elements
A chemical element is a pure substance made of only one kind of atom, distinguished by its atomic number. Each element has characteristic chemical properties and is represented by a chemical symbol (for example C for carbon, O for oxygen).
- Examples of elements: Carbon, Oxygen, Silicon, Arsenic, Aluminium, Iron, Copper, Gold, Mercury.
- Hydrogen and helium are the most abundant elements in the universe by number of atoms.
- Iron is the most abundant element by mass in the Earth as a whole (large proportion in the core).
- Oxygen is the most abundant element in the Earth's crust by mass.
The eight most abundant elements in Earth's crust (by mass)
- 46.6% Oxygen (O)
- 27.7% Silicon (Si)
- 8.1% Aluminium (Al)
- 5.0% Iron (Fe)
- 3.6% Calcium (Ca)
- 2.8% Sodium (Na)
- 2.6% Potassium (K)
- 2.1% Magnesium (Mg)
Periodic Table
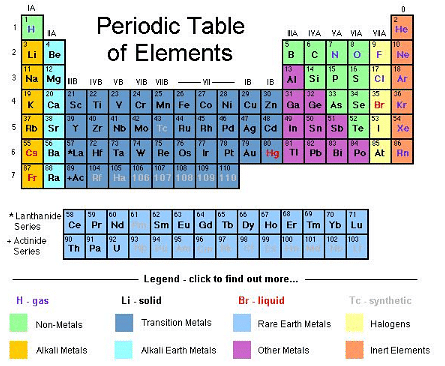
The periodic table organises elements in order of increasing atomic number and groups together elements with similar chemical properties. It reveals periodic trends in properties such as atomic size, ionisation energy and electronegativity.
- Dmitri Ivanovich Mendeleev: Prepared the first widely recognised periodic table based on atomic masses and used it to predict properties of yet-undiscovered elements.
- Henry G. J. Moseley: Established that atomic number (number of protons) is the proper organising principle; Moseley's work led to the modern arrangement by atomic number.
- The table is organised into groups (vertical columns) and periods (horizontal rows); elements are broadly classified as metals, metalloids and non-metals.
Natural and Synthetic Elements
Natural elements occur naturally on Earth. Synthetic elements are produced artificially, typically in laboratories or reactors; many synthetic elements are unstable and radioactive.
- The first element to be produced artificially and widely recognised as the first synthetic element is technetium (element 43); it was identified and produced in the 1930s (work by Emilio Segrè and Carlo Perrier, 1937).
- Transuranic elements (atomic number > 92) are generally synthetic and produced in nuclear reactors or particle accelerators.
Compounds
A compound is a substance formed when two or more elements are chemically bonded in fixed proportions. Compounds have properties different from those of their constituent elements.
- A compound cannot be separated into its constituent elements by simple physical processes; chemical reactions are required to break chemical bonds.
- Examples: Common salt (NaCl) is formed from sodium and chlorine atoms; sodium carbonate (Na2CO3) contains sodium, carbon and oxygen.
Types of Chemical Bonds
- Ionic bond: Formed by transfer of electrons between atoms producing positive and negative ions (example: NaCl).
- Covalent bond: Formed by sharing of electrons between atoms (example: H2O, O2).
- Metallic bond: A network of positive ions surrounded by a sea of delocalised electrons (characteristic of metals).
- Coordinate (dative covalent) bond: A covalent bond in which both shared electrons are supplied by one atom (example: [NH4]+ from NH3 and H+).
Mixtures
A mixture results when two or more substances are combined physically without chemical bonding. Each component in a mixture retains its own chemical identity and properties.
- Components of a mixture can be separated by physical methods such as filtration, distillation, evaporation, centrifugation or chromatography depending on the nature of the mixture.
- The physical properties of each component in a mixture remain essentially unchanged.
- Examples: a mixture of sand and water; a solution of salt in water; a mixture of sugar and salt.
Types of Mixtures
- Homogeneous mixture (solution): Uniform composition throughout. Example: salt dissolved in water.
- Heterogeneous mixture: Non-uniform composition where components are distinguishable. Example: sand in water.
- Colloids: Mixtures where tiny particles of one substance are dispersed in another but do not settle on standing (example: milk, fog).
- Suspensions: Mixtures in which particles are larger and may settle on standing (example: muddy water).
Common Separation Techniques
- Filtration: Separates insoluble solids from liquids by passing the mixture through a porous medium (filter paper).
- Distillation: Separates components of a liquid mixture based on differences in boiling points (simple and fractional distillation).
- Evaporation: Removes a solvent by heating to leave behind dissolved solids (e.g., obtaining salt from seawater).
- Chromatography: Separates components based on differential partition between stationary and mobile phases (paper, column, thin-layer chromatography).
- Centrifugation: Uses rapid rotation to separate components of different densities (used for suspensions and biological samples).
- Decantation and sieving: Simple physical methods for coarse mixtures like sand and water or mixtures of solids with different particle sizes.
Practical and Conceptual Notes
- Atomic number (Z) uniquely identifies an element and equals the number of protons in its nucleus.
- Mass number (A) is the total number of protons and neutrons; isotopes differ in A but have same Z.
- Chemical behaviour of an atom is largely determined by its valence electrons - the electrons in the outermost shell.
- Periodic trends such as atomic radius, ionisation energy and electronegativity arise from the arrangement of electrons and nuclear charge and provide predictive power for chemical reactivity.
- Understanding the difference between physical combinations (mixtures) and chemical combinations (compounds) is fundamental for predicting what changes are reversible by physical methods and which require chemical reactions.
These fundamentals-atoms, subatomic particles, molecules, elements, compounds and mixtures-provide the basis for chemical thinking and are essential for further study in chemistry, materials science, environmental science and related technologies.
FAQs on Chemistry Basics – Atoms, Molecules, Elements, Compounds, and Mixtures
| 1. What are atoms and why are they important in chemistry? |  |
| 2. How do molecules differ from atoms? |  |
| 3. What is the significance of the periodic table in understanding elements? |  |
| 4. What are compounds and how do they differ from mixtures? |  |
| 5. Can you explain the difference between homogeneous and heterogeneous mixtures? |  |






