Practice Questions: Metals and Non-metals
Fill in the Blanks
Q1: The ability of metals to be drawn into thin wires is called _______________.
Ans: ductility
Q2: Non-metals generally have _______________ lustre.
Ans: no
Q3: The element that is an essential constituent of chlorophyll is _______________.
Ans: magnesium
Q4: The process of applying a thin layer of zinc on iron to prevent rusting is called _______________.
Ans: galvanization
Q5: _______________ is a soft metal used in making electric wires and cables.
Ans: Copper
Short Answer Questions
Q6: Differentiate between metals and non-metals based on their physical properties.
Ans:
- Metals: Generally have a metallic lustre; are good conductors of heat and electricity; are usually malleable (can be beaten into sheets) and ductile (can be drawn into wires); most are solid at room temperature (except mercury).
- Non-metals: Usually lack lustre (appear dull) when solid; are generally poor conductors of heat and electricity; are often brittle (shatter when struck) if solid; exist as solids, liquids or gases at room temperature (for example, bromine is a liquid, oxygen is a gas).
Q7: Why do ionic compounds generally have high melting and boiling points?
Ans: Ionic compounds are composed of positive and negative ions held together by strong electrostatic forces of attraction (ionic bonds) in a three-dimensional lattice. A large amount of energy is required to overcome these forces and separate the ions, so ionic compounds usually have high melting and boiling points.
Q8: Explain the process of rusting of iron with the help of a chemical reaction.
Ans:
Rusting is the corrosion of iron caused by its reaction with oxygen in the presence of moisture.
Iron is oxidised and hydrated to form hydrated iron(III) oxide (rust).
Chemical representation: 4Fe + 3O2 + 6H2O → 4Fe(OH)3.
Over time, Fe(OH)3 may dehydrate to form Fe2O3·nH2O, the reddish-brown substance commonly called rust, which weakens the metal.
Rusting is accelerated by the presence of salts and acids because they increase the conductivity of the aqueous layer on iron and facilitate electron transfer.
Q9: How does the reactivity of metals change as we move down the reactivity series?
Ans: In the standard reactivity series, metals at the top are the most reactive whereas those at the bottom are the least reactive. Therefore, moving from top to bottom, the reactivity of metals generally decreases. Highly reactive metals easily lose electrons to form positive ions, while less reactive metals do not lose electrons readily.
Q10: Why is sodium kept immersed in kerosene oil?
Ans: Sodium is a very reactive metal that reacts vigorously with air (oxygen) and with water, producing heat and hydrogen gas. Storing sodium under kerosene oil prevents contact with air and moisture, stopping oxidation and dangerous reactions and keeping the metal safe to handle.
Long Answer Questions
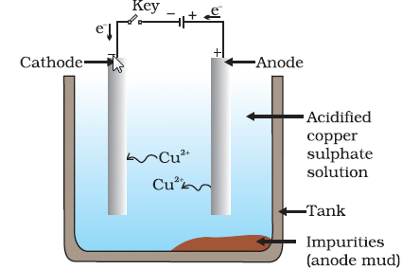
Q11: Explain the process of electrolytic refining of copper.
Ans:
The purpose of electrolytic refining is to obtain pure copper from impure copper.
An electrolytic cell is set up where the impure copper block serves as the anode (positive electrode) and a thin sheet of pure copper serves as the cathode (negative electrode).
The electrolyte is an aqueous solution of copper sulfate, often with a little dilute sulphuric acid to increase conductivity.
When electric current is passed, copper atoms from the anode lose electrons and enter the solution as Cu2+ ions.
Cu2+ ions move through the solution and gain electrons at the cathode, depositing as pure copper on the cathode.
Insoluble impurities (such as gold and silver) fall off and collect as anode mud at the bottom of the cell; soluble impurities remain in the electrolyte and can be removed later.
Over time the cathode becomes thicker with pure copper while the anode becomes thinner, yielding refined copper suitable for electrical and industrial applications.
Q12: Compare the properties of metals and non-metals in terms of their chemical reactivity.
Ans:
- Metals: Usually lose electrons to form positive ions (cations); act as reducing agents because they donate electrons; many metals react with acids to produce hydrogen gas (for example, Zn + 2HCl → ZnCl2 + H2).
- Non-Metals: Tend to gain electrons to form negative ions (anions) or share electrons to form covalent bonds; often act as oxidising agents because they accept electrons; most non-metals do not react with acids to liberate hydrogen gas - they react in other ways (for example, chlorine reacts with metals to form salts).
Q13: Describe the extraction of iron from its ore, haematite, using a blast furnace.
Ans:
- Raw materials: Haematite (Fe2O3), coke (carbon) and limestone (calcium carbonate).
- Function of coke: Burns to produce heat and forms carbon monoxide which acts as the reducing agent.
- Reduction: Carbon monoxide reduces iron(III) oxide to iron: Fe2O3 + 3CO → 2Fe + 3CO2.
- Role of limestone: Decomposes to CaO which reacts with silica and other impurities to form molten slag (calcium silicate). The slag floats on the molten iron and is removed.
- Product: Molten iron (pig iron) collects at the bottom and is tapped off; the molten iron can be further refined to make steel.
Q14: Why do ionic compounds conduct electricity in molten state or aqueous solutions but not in solid state?
Ans:
- In the solid state, ions in ionic compounds are fixed in a rigid lattice and cannot move; therefore they cannot carry electric current.
- In the molten state (when melted) or in aqueous solution, the ions are free to move through the liquid.
- Since electrical conduction requires mobile charged particles, ionic compounds conduct electricity only when their ions are free to move (molten or dissolved), not when they are locked in a solid lattice.
Q15: Discuss the uses of metals and non-metals in daily life with suitable examples.
Ans:
- Iron: Used in construction - beams, rods, sheets, and in making tools and machinery.
- Aluminium: Used for making lightweight utensils, aircraft parts, window frames and packaging because it is light and corrosion-resistant.
- Copper: Used extensively in electrical wiring and cables because it is a very good conductor of electricity.
- Sulfur: Used in the manufacture of matches, sulphuric acid and various chemicals.
- Carbon: In the form of graphite it is used for pencil leads and as a lubricant; carbon in other forms is used in fuels and filters.
- Oxygen and Helium: Oxygen is essential for respiration and medical uses; helium is used to fill balloons because of its low density and inert nature.
FAQs on Practice Questions: Metals and Non-metals
| 1. What are the physical properties that distinguish metals from non-metals? |  |
| 2. How do metals and non-metals react with oxygen? |  |
| 3. Can you give examples of common metals and non-metals used in everyday life? |  |
| 4. What is the significance of the reactivity series in understanding metals and non-metals? |  |
| 5. What are the methods of extraction for metals and how do they differ from non-metals? |  |





