Class 8 Science Chapter 11 Question Answers - Chemical Effects of Electric Current
Q1: What happens when an electric current is passed through a cut potato for a considerable time?
Ans: When an electric current is passed through a cut potato for a considerable time, a greenish-blue spot forms around the positive electrode. This is a result of the chemical reactions occurring due to the passage of electric current through the potato.
Q2: What is the magnetic effect of electricity? Explain.
Ans: The magnetic effect of electricity refers to the ability of an electric current to create a magnetic field around it. When current flows through a conductor, it generates a circular magnetic field perpendicular to the direction of the current.
Q3: Why is it easier for the fish to swim in water?
Ans: Fish can easily swim in water because it has a streamlined body which produces less friction. So it easily cuts through water and moves in it very easily.
Q4: What is electroplating? What are its uses?
Ans: Electroplating is a process in which a layer of a desired metal is deposited onto another material using electricity. It is used to improve the appearance of objects, provide corrosion resistance, and enhance durability. For example, chrome plating on car parts or gold plating on jewelry.
Q5: Define good conductors and poor conductors or insulators.
Ans: Good conductors are materials that allow electricity to flow through them easily, while poor conductors or insulators do not conduct electricity effectively. Examples of good conductors include copper, brass, and aluminum, while examples of insulators are rubber, plastic, wood, and air.
Q6: How does a bulb glow in liquid? Explain.
Ans: When the liquid between the two ends of a tester allows electric current to pass through it, the circuit of the tester is completed, and the current flows. This current causes the bulb in the tester to glow, indicating that the liquid is a conductor of electricity.
Q7: Why are the worn out tires discarded?
Ans: Worn-out tires are discarded because their tread is worn off, limiting their effectiveness in providing the required amount of friction.
Q8: How is the conductivity of liquids tested?
Ans: The conductivity of liquids can be tested using a tester. The free ends of the tester are dipped in the liquid. If the bulb in the tester glows, the liquid is a conductor of electricity. If the bulb does not glow, the liquid is an insulator.
Q9: What are infrasonic sounds?
Ans: Sounds which have a frequency lower than 20 Hz are called infrasonic sounds.
Q10: What is an LED? Why is it preferred to other types of bulbs?
Ans: An LED (Light Emitting Diode) is an electric device used in testers and displays. It is preferred to other types of bulbs because it can emit light even when a weak or low current flows through it. LEDs are energy-efficient and have a longer lifespan compared to traditional bulbs.
Q11: Explain the conductivity of water.
Ans: Normal water that we encounter in everyday situations, such as tap water, contains dissolved salts that make it a conductor of electricity. This is why normal water conducts electricity. Pure or distilled water, on the other hand, is free of dissolved salts and does not conduct electricity effectively.
Q12: Current does not flow in a circuit if there is a gap between the two wires. Does it indicate that air is a poor conductor of electricity? Does air never conduct electricity? Explain.
Ans: Yes, a gap between the wires disrupts the circuit, preventing current flow. This indicates that air is a poor conductor of electricity. However, air can conduct electricity under certain conditions, such as during lightning when ionization of air molecules allows the flow of current. Additionally, moist air can conduct electricity better than dry air due to the presence of ions.
Q13: How does the friction depend on the nature of the surface?
Ans: The nature of the surface is one of the factors affecting friction. Rough surfaces have a larger number of irregularities and greater frictional force. When we move on any surface, we apply a force to overcome the interlocking of the surfaces.
Q14: Why is a magnetic compass needed to test the conduction of electric current?
Ans: A magnetic compass is needed to test the conduction of electric current because it can detect even small currents that may not be sufficient to light up a bulb. The magnetic field produced by the current can deflect the needle of the compass, indicating the presence of current.
Q15: Show with the help of a diagram that lemon juice and vinegar are good conductors of electricity.
Ans: When the ends of a tester is dipped in lemon juice or vinegar, the bulb glows. This process indicates that lemon juice and vinegar, both, are good conductors of electricity. Testing Conduction of electricity in lemon juice or vinegar
Testing Conduction of electricity in lemon juice or vinegar
Q16: With the help of a suitable diagram, explain electrolytic refining of copper.
Ans: To purify copper, a thin plate of pure copper and a thick rod of impure copper are used as electrodes in the acidified solution of CuS04. Pure copper is used as cathode and impure copper is used as anode. When electric current is passed through the copper sulphate solution, copper sulphate dissociates into copper and sulphate. The free copper gets drawn to the electrode connected to the negative terminal of the battery and gets deposited on it. From impure copper electrode, an equal amount of copper gets dissolved in the solution. Thus, the loss of copper from solution is restored and the process continues. The impurities are left behind at anode.
Q17: What is electroplating? On which effect of the electric current is it based? Why is it done?
Ans:
- The process of depositing or coating a layer of any desired metal on the surface of other material by means of electricity is called electroplating. It is one of the most common applications of chemical effects of electric current.
- Electroplating is a very useful process. It is widely used in industry for coating metal objects with a thin layer of a different metal. The layer of metal deposited has some desired property, which the metal of the object lacks. For example, chromium plating is done on many objects to make them shiny and attractive.
Q18: On what factors thickness of the electroplated items depend?
Ans: Thickness of electroplated items depend upon:
- The strength of the current passing through the circuit.
- The concentration of the metal ion in the solution.
- The duration of the time the article has been in the solution.
Q19: Show the conductivity of water with the help of an activity.
Ans: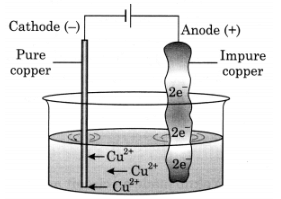 Electrolytic refining of copper
Electrolytic refining of copper
Normal or ordinary water is a good conductor of electricity while distilled water is a bad conductor or insulator. Ordinary water may contain small amount of mineral salts dissolved in it naturally; on the other hand, distilled water is free of salts.
The following activity shows this fact:
About 50 mL of distilled water is taken in a clean and dry beaker. When the tester is dipped into the distilled water, the bulb does not glow which shows that distilled water is a bad conductor of electricity. But when a small amount of common salt is dissolved in distilled water and again tested the bulb glows which shows that distilled water when mixed with salts conduct electricity.
Q20: What are the advantages and disadvantages of electroplating?
Ans: Electroplating is a very useful process. It is widely used in industry for coating metal objects with a thin layer of different metal. The advantages and disadvantages of electroplating are:
Advantages:
- It protects the metals from being corroded.
- It prevents the rusting of metals.
- It makes cheap and dull metals shiny and attractive.
- It can make more reactive metals like iron less reactive.
- Chromium coating on metals give lustre to objects.
Disadvantages:
- Pollutants from electroplating industries are very harmful. Some chemicals are very lethal for both human and animals.
- It is an expensive process.
|
666 docs
|





















