Case Based Questions: Structure of the Atom
Case Study I
Read the given passage and answer the questions based on the passage and related studied concepts.
Atom consists of electrons, protons and neutrons. J.J. Thomson proposed that electrons are embedded in a positive charge uniform sphere. Rutherford a-scattering experiment led to discovery of nucleus in the centre of atom which is positively charged and whole mass of atom is concentrated in the nucleus. Neil Bohr proposed that electrons are distributed in different shells in M, N... with discrete energy around the nucleus. If atomic shell is complete, atom will be stable and less reactive. Electrons are negatively charged, protons are positively charged, neutrons are neutral. Valency is the combining capacity of an atom. Atomic number is equal to number of protons in an atom. The mass number of an atom is equal to sum of number of protons and neutrons in the nucleus. Isotopes have same atomic number but different mass numbers. Isobars have same mass number but different atomic numbers. Elements are defined by number of protons (atomic number) they possess.
Q1: Which of the following does not contain neutron?
(a) Al
(b) Na
(c) H
(d) Mg
Ans: (c)
Hydrogen (H) does not contain neutron.
Q2: What is the atomic number of Al3+, if it has 13 protons, 10 electrons, 14 neutrons?
(a) 13
(b) 10
(c) 14
(d) 27
Ans: (a)
The atomic number of an element is determined by the number of protons it contains. In this case:
- The element has 13 protons.
- Therefore, the atomic number is 13.
It is important to note that the number of electrons and neutrons does not affect the atomic number.
Q3: Write electronic configuration of 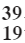 K.
K.
Ans: The electronic configuration of potassium (K) is:
- 2 electrons in the first shell
- 8 electrons in the second shell
- 8 electrons in the third shell
- 1 electron in the fourth shell
Thus, the complete configuration is 2, 8, 8, 1.
Q4: Isotopes differ in which sub-atomic particles?
Ans: Neutrons
Case Study II
Observe the Bar-chart shown for elements with atomic number 1 to 10. Answer the questions based on this graph and related studied concepts.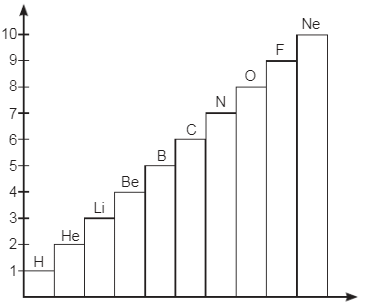 Q1: Name the element whose first shell is complete.
Q1: Name the element whose first shell is complete.
Ans: Helium is the element whose first shell is complete.
- It has two electrons in its outermost shell.
- This configuration makes it stable and non-reactive.
- Other elements typically aim for a full outer shell of eight electrons.
Q2: Name the element in which second shell has twice electrons than 1st shell.
Ans: The element where the second shell has twice the number of electrons compared to the first shell is carbon.
- First shell (K-shell): 2 electrons
- Second shell (L-shell): 4 electrons
This gives the electronic configuration of carbon as C(2, 4).
Q3: How does valence electrons vary from Li to Ne?
Ans: The number of valence electrons varies as you move from Lithium (Li) to Neon (Ne) in the periodic table:
- Li has 1 valence electron.
- Be has 2 valence electrons.
- B has 3 valence electrons.
- C has 4 valence electrons.
- N has 5 valence electrons.
- O has 6 valence electrons.
- F has 7 valence electrons.
- Ne has 8 valence electrons.
In summary, the number of valence electrons increases from Li to Ne, reaching a maximum of 8 in Ne.
Q4: Name the element which has 7 valence electrons.
Ans: Fluorine (F) has 7 valence electrons in its outermost shell.
- It is more stable for fluorine to gain one electron.
- This gives it a valency of 1.
Case Study III
Many scientists contributed in revealing the presence of charged particles in an atom. It was known by 1900 that the atom was indivisible particle but contained at least one sub-atomic particle - the electron identified by J. J. Thomson. Even before the electron was identified, in 1886 E. Goldstein discovered the presence of new radiations in a gas discharge and called them canal rays. These rays were positively charged radiations which ultimately led to the discovery of another sub-atomic particle. This sub-atomic particle had a charge, equal in magnitude but opposite in sign to that of the electron. Its mass was approximately 2000 times as that of the electron. It was given the name of proton. In general, an electron is represented as 'e-' and a proton as 'p+'. The mass of a proton is taken as one unit and its charge as plus one. The mass of an electron is considered to be negligible and its charge is minus one.
It seemed that an atom was composed of protons and electrons, mutually balancing their charges. It also appeared that the protons were in the interior of the atom, for whereas electrons could easily be removed off but not protons.
Read the given passage carefully and give the answer of the following questions:
Q1: Who discovered electron?
(a) E. Goldstein
(b) Bohr
(c) J. J. Thomson
(d) J. Chadwick
Ans: (c)
J. J. Thomson discovered the electron in 1897 using cathode ray tube experiments and showed these rays were negatively charged particles.
Q2: Which of the following has a charge of +1 and a mass of 1 amu?
(a) A neutron
(b) A proton
(c) An electron
(d) A helium nucleus
Ans: (b)
A proton has +1 charge and ≈1 amu mass; a neutron is neutral and an electron is much lighter.
Q3: Mass of proton is:
(a) equal to the mass of hydrogen atom
(b) less than the mass of hydrogen atom
(c) negligible
(d) more than the mass of hydrogen atom
Ans: (a)
The proton's mass is approximately equal to that of a hydrogen atom (~1 amu), ignoring the small electron mass.
Q4: Proton was discovered by:
(a) Thomson
(b) Rutherford
(c) Chadwick
(d) Goldstein
Ans: (d)
E. Goldstein observed positively charged canal rays in discharge tubes, leading to identification of the proton (hydrogen nucleus).
Q5: Which statement is true?
(a) The nucleus of an atom contains only neutrons.
(b) The nucleus of an atom contains only protons and electrons.
(c) Protons and neutrons are sub-atomic particles.
(d) Protons have the same charge as neutrons.
Ans: (c)
Protons, neutrons, and electrons are sub-atomic particles; the nucleus contains protons and neutrons, while electrons are negatively charged.
FAQs on Case Based Questions: Structure of the Atom
| 1. What is the difference between electrons, protons, and neutrons in an atom? |  |
| 2. Why did Rutherford's gold foil experiment change how we understand atomic structure? |  |
| 3. How many shells can electrons occupy, and what's the maximum number in each shell? |  |
| 4. What's the difference between atomic number and mass number? |  |
| 5. How do isotopes of the same element have different mass numbers but the same atomic number? |  |






