Diagram Based Questions: Carbon and its compounds
Q1: Answer the following questions based on the diagram given below:
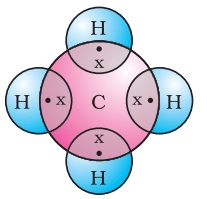 Covalent Bond of Methane
Covalent Bond of Methane(ii) What is the chemical formula of methane?
Ans: The chemical formula of methane is CH4. It contains one carbon atom bonded to four hydrogen atoms.
(ii) How many valence electrons does carbon have in methane?
Ans: Carbon has four valence electrons in methane. These four electrons are shared with four hydrogen atoms to form four single covalent bonds.
(iii) How many hydrogen atoms are bonded with carbon in methane?
Ans: In methane, carbon is bonded to four hydrogen atoms. The hydrogen atoms are arranged tetrahedrally around the central carbon atom.
(iv) What type of bonds are formed between carbon and hydrogen in methane?
Ans: Covalent bonds are formed between carbon and hydrogen in methane. Each C-H bond is a single covalent bond formed by sharing one pair of electrons between carbon and hydrogen.
(v) Why are covalent compounds like methane generally poor conductors of electricity?
Ans: Covalent compounds like methane are poor conductors of electricity because they do not contain free ions or free electrons to carry charge. Electrons in methane are shared and localised in covalent bonds, so there are no mobile charged particles to conduct electric current.
Q2: Answer the following questions based on the diagram given below:
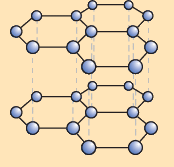 Allotropic Form of Graphite
Allotropic Form of Graphite
(i) What is the basic structure of graphite?
Ans: Graphite is made up of flat layers of carbon atoms arranged in hexagonal rings. Each carbon atom forms three covalent bonds with neighbouring carbon atoms in the same plane, while the remaining electron becomes delocalised within the layer.
(ii) How are the layers of carbon atoms arranged in graphite?
Ans: The layers of carbon atoms in graphite are stacked one above the other, held together by weak forces between the layers. This layered arrangement allows the sheets to slide over one another easily.
(iii) Why do diamond and graphite have different physical properties despite having the same chemical properties?
Ans: Diamond and graphite differ in physical properties because their carbon atoms are bonded in different ways. In diamond, each carbon is bonded to four others in a three-dimensional network, making it extremely hard and an electrical insulator. In graphite, carbon atoms form flat layers with delocalised electrons, so graphite is soft, slippery and conducts electricity within the layers.
(iv) What is the significance of the double bonds between carbon atoms in graphite?
Ans: The presence of alternating single and double bonds (leading to delocalised electrons) in the graphite layers helps satisfy the valency of carbon and provides stability to the planar structure. The delocalised electrons can move freely across each layer, contributing to electrical conductivity.
(v) How does the arrangement of carbon atoms in graphite contribute to its excellent electrical conductivity?
Ans: The hexagonal arrangement of carbon atoms in each graphite layer creates delocalised electrons that are free to move within the layers. These mobile electrons allow electric current to flow easily along the planes, making graphite a good conductor of electricity.
Q3: Answer the following questions based on the diagram given below:
 Formation of Ester
Formation of Ester
(i) What is the process shown in the diagram titled "Formation of Ester"?
Ans: The process shown is esterification - a chemical reaction in which an alcohol reacts with a carboxylic acid to form an ester and water. This is a condensation reaction and is often carried out in the presence of an acid catalyst.
(ii) What are the two main reactants involved in the formation of an ester?
Ans: The two main reactants are an alcohol and a carboxylic acid. For example, ethanol reacting with ethanoic acid gives ethyl ethanoate (an ester) and water.
(iii) What type of bond is formed during the formation of an ester, and what is the byproduct of this reaction?
Ans: A covalent bond called an ester linkage (-COO-) is formed between the acid and the alcohol. The byproduct of the reaction is water (H2O), produced by the combination of -OH from the acid and H from the alcohol's -OH.
(iv) What are some examples of esters that are commonly found in nature or used in everyday life?
Ans: Many esters give pleasant fragrances and flavours. Examples include isoamyl acetate (banana scent), ethyl acetate (used as a solvent and has a fruity smell), and ethyl butanoate (pineapple-like aroma). Esters occur naturally in fruits and flowers and are used in flavourings and perfumes.
(v) How does the formation of esters contribute to the world of perfumes and flavors?
Ans: By combining different alcohols and carboxylic acids, chemists can produce a wide variety of esters, each with a distinct scent or flavour. This allows the replication and enhancement of natural aromas in perfumes, food flavourings and fragrances used in everyday products.
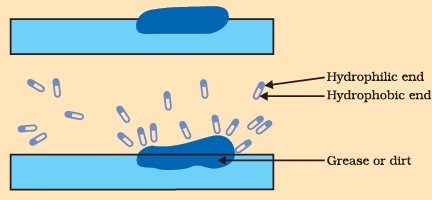
Q4: Answer the following questions based on the diagram given below: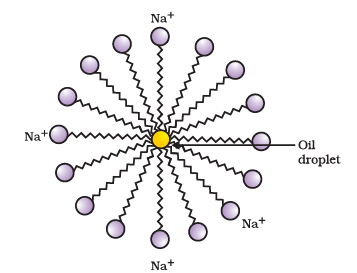 Formation of micelles
Formation of micelles
(i) What is the main reason behind using soap for cleaning?
Ans: The main reason for using soap is that its molecules can form micelles which trap oil and dirt, allowing these to be removed with water.
(ii) How do soap molecules interact with both water and oil?
Ans: Soap molecules are amphiphilic: one end is hydrophilic (water-loving, usually an ionic head) and the other end is hydrophobic (water-repelling, oily tail). The hydrophilic end interacts with water while the hydrophobic end attaches to oil and grease.
(iii) What is a micelle, and how does it form?
Ans: A micelle is a spherical cluster of soap molecules formed in water. The hydrophobic tails turn inward to avoid water and trap oil or grease, while the hydrophilic heads face outward towards the water, keeping the trapped oil suspended.
(iv) Why doesn't oil dissolve in water, and how do micelles help in cleaning oily substances?
Ans: Oil does not dissolve in water because oil molecules are non-polar and water is polar; they do not mix. Micelles help by surrounding oil droplets with their hydrophobic tails, isolating the oil inside the micelle so it can be dispersed and rinsed away in water.
(v) How does soap make it easier to wash clothes clean?
Ans: Soap forms micelles that lift oily dirt and grease away from fibres and keep these particles suspended in water. Washing and rinsing then carry away the micelles with the dirt, leaving the clothes cleaner.
Q5: Answer the following questions based on the diagram given below:
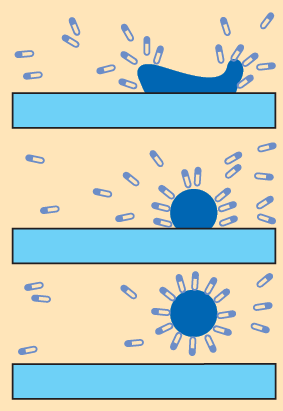
(i) What happens to soap molecules when they are at the surface of water?
Ans: When soap molecules are at the water surface, their hydrophobic tails stick out of the water while their ionic (hydrophilic) heads remain in contact with water. This alignment reduces surface tension and arranges the molecules along the surface.
(ii) How do soap molecules organize themselves when they are in water?
Ans: In bulk water, soap molecules organise into micelles. In a micelle the hydrophobic tails point inward away from the water and the hydrophilic heads face outward, interacting with the surrounding water.
(iii) Why is soap able to clean effectively?
Ans: Soap cleans effectively because its micelles encapsulate oily dirt inside their hydrophobic cores, turning large oil droplets into small, suspended particles that can be washed away with water - a process called emulsification.
(iv) Why do soap micelles stay in solution as a colloid?
Ans: Soap micelles remain dispersed as a colloid because the charged hydrophilic heads on their surface repel one another, preventing micelles from clumping and settling. Their small size and continuous motion in water also keep them suspended.
(v) Why does a soap solution appear cloudy?
Ans: A soap solution appears cloudy because micelles and emulsified oil droplets are large enough to scatter light. This scattering of light by particles in the solution gives it a cloudy appearance.
FAQs on Diagram Based Questions: Carbon and its compounds
| 1. What is the structure of carbon dioxide? |  |
| 2. How is carbon dioxide produced in the atmosphere? |  |
| 3. What are hydrocarbons? |  |
| 4. What is the difference between saturated and unsaturated hydrocarbons? |  |
| 5. How does carbon form multiple compounds? |  |





