Anode (canal) rays are streams of positively charged ions produced in a discharge tube and emitted from the anode. They provided key evidence for the presence of positive particles in atoms and helped in understanding the existence of protons.
Atomic Structure - Chemistry
Structure of an Atom
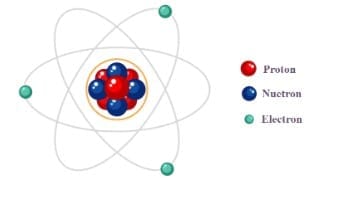
Electron
- The electron is a fundamental sub-atomic particle carrying a single unit negative charge (-1 e) and very small mass compared with nucleons.
- It was discovered by J. J. Thomson in 1897 by experiments with cathode rays. Electrons occupy the region outside the nucleus in discrete shells or energy levels; their arrangement determines chemical behaviour.
Proton
- The proton is a positively charged nucleon with charge +1 e and a mass approximately 1836 times that of an electron.
- The idea of a positively charged constituent in the nucleus came from early ion experiments, Eugen Goldstein's canal-ray (anode-ray) observations in 1886 showed positively charged rays, which later helped in understanding positive nuclear constituents.
- The proton was definitively identified as a nuclear particle through experiments by Ernest Rutherford in the early 20th century.
Neutron
- The neutron is a neutral nucleon (no electric charge) with mass similar to the proton.
- It was discovered by James Chadwick in 1932. Neutrons contribute to the mass of the nucleus and play a key role in nuclear stability.
The atom has two major structural regions:
(i) The central nucleus - a very small, dense region containing protons and neutrons (collectively called nucleons). The nucleus has a positive charge due to protons.
(ii) The external region - electrons move in the space around the nucleus in shells or energy levels. The extent of this region determines the atomic size.
A neutral atom has equal numbers of protons and electrons, so their opposite charges cancel out and the net charge becomes zero.
Cathode Rays
Cathode rays are streams of electrons emitted from the cathode in discharge tubes. They provided key evidence for the existence of electrons and for charge-to-mass measurements (e/m).
- Cathode rays travel in straight lines and cast shadows of solid objects placed in their path, showing particle-like behaviour.
- They can produce mechanical effects (e.g., cause a paddle wheel in a discharge tube to turn), indicating they consist of material particles (electrons).
- Cathode rays carry negative electric charge.
- Cathode rays are deflected by electric and magnetic fields, consistent with their charge and mass.
- They produce heating effects on impact with matter.
- They ionize gases they pass through.
- They cause fluorescence on glass walls and on certain phosphors (e.g., zinc sulphide).
- They can darken photographic plates.
- They have some penetrating power through thin materials.
Anode (Canal) Rays
- Anode rays (also called canal rays or positive rays) travel in straight lines but typically have much lower speed than cathode rays in the same tube.
- They consist of positively charged material particles (positive ions).
- Anode rays are deflected by electric and magnetic fields according to their positive charge and mass.
- The nature (mass and charge) of anode ray particles depends on the gas in the discharge tube; different gases produce different positive ions, so the e/m ratio for anode rays is not constant across gases.
- Early observations of these rays were made by Eugen Goldstein (1886).
Properties of the Nucleus
- The nucleus is composed of protons and neutrons, together called nucleons.
- Nucleons are bound together by the strong nuclear force, making the nucleus extremely small compared with the overall atom but containing nearly all the atom's mass.
- Electrons occupy the space outside the nucleus and determine chemical properties.
- Because atoms are electrically neutral, the number of protons in the nucleus equals the number of electrons outside; this common number is the atomic number (symbol Z).
Atomic Number (Z)
- The atomic number (Z) is defined as the total number of protons present in the nucleus of an atom.
- In a neutral atom, Z is also equal to the number of electrons.
- Notation:
Z = number of protons = number of electrons (for neutral atoms)
Mass Number (A)
- The mass number (A) of an atom is the total number of nucleons (protons + neutrons) in its nucleus.
- Mass number, A = number of protons (Z) + number of neutrons (n).
- Mass number is an integer and is written in the conventional notation as AX Z (for example, 14C6 has A = 14 and Z = 6).
Valency and Valence Shell
- The outermost occupied shell of an atom is called the valence shell.
- Electrons in the valence shell are called valence electrons and play the decisive role in chemical reactions and bonding.
- Valency is the combining capacity of an atom and is determined by the number of electrons an atom can gain, lose or share to achieve a stable electronic configuration (often the nearest noble-gas configuration).
- During chemical combination, only valence electrons are involved: they may be transferred or shared between atoms.
- Valency can often be inferred from the number of valence electrons: for main-group elements, valency ≈ number of electrons needed to reach a full outer shell (usually 8 for many elements, 2 for H and He).
| Alkali Metal | Atomic Number | Electronic Configuration | Number of Valence Electrons |
|---|---|---|---|
| Lithium (Li) | 3 | 2, 1 | 1 |
| Sodium (Na) | 11 | 2, 8, 1 | 1 |
| Potassium (K) | 19 | 2, 8, 8, 1 | 1 |
Significance of Valence Electrons
- Valence electrons determine how an element reacts chemically. They are the electrons involved in bond formation.
- Elements with the same number of valence electrons exhibit similar chemical properties (for example, alkali metals all have one valence electron and react similarly).
- The principal quantum number of the valence shell indicates the period (row) of the element in the Periodic Table.
- Elements with 1,2 and 3 valence electrons are generally metals (exceptions include hydrogen and helium). Elements with 4-7 valence electrons are typically non-metals.
Isotopes
- Isotopes are atoms of the same element (same Z) that have different mass numbers (different numbers of neutrons).
- Isotopes have the same number of protons and electrons but different numbers of neutrons.
Examples: Carbon has stable isotopes 12C and 13C and a radioactive isotope 14C used in radiocarbon dating.
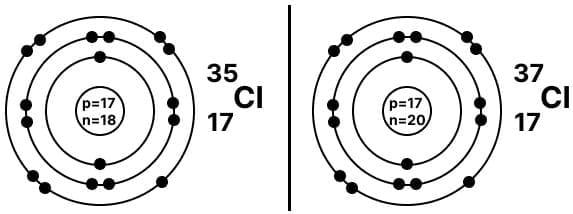 Isotope of Chlorine
Isotope of Chlorine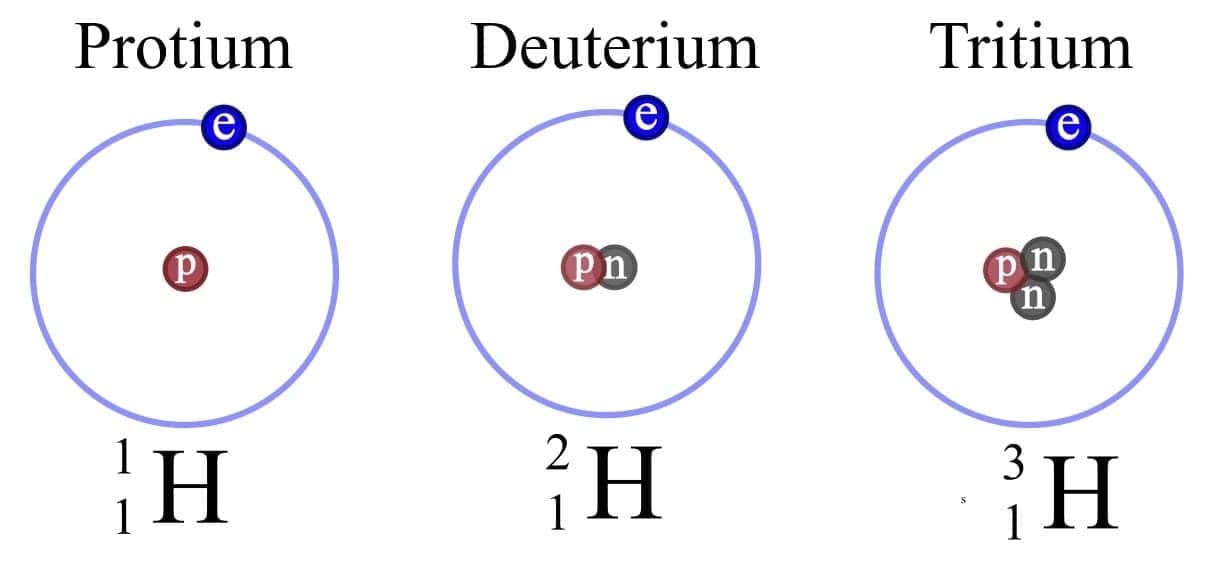 Isotope of Hydrogen
Isotope of Hydrogen Properties of Isotopes
- Isotopes of an element have identical electronic configurations and hence identical chemical properties (because chemistry depends on electrons).
- Their physical properties (mass, density, melting and boiling points) differ slightly due to differences in nuclear mass.
- Certain isotopes are radioactive and have applications in medicine, industry and archaeology (e.g., 99mTc in medical imaging, 14C in dating).
Isobars
- Isobars are atoms of different elements that have the same mass number (A) but different atomic numbers (Z).
- Isobars therefore have different proton (and electron) counts but the same total number of nucleons (protons + neutrons).
| Isobar | Atomic number (Z) | Mass number (A) | Electrons (e) | Protons (p) |
|---|---|---|---|---|
| 40Ar18 | 18 | 40 | 18 | 18 |
| 40K19 | 19 | 40 | 19 | 19 |
| 40Ca20 | 20 | 40 | 20 | 20 |
Properties of Isobars
- Isobars have nearly equal mass numbers but different chemical properties because they are different elements (different Z).
- Physical properties may also differ because electronic structure and nuclear composition differ.
Isotones
- Isotones are atoms of different elements that have the same number of neutrons but different numbers of protons (different Z).
| Isotone | Atomic number (Z) | Mass number (A) | Electrons (e) | Protons (p) | Neutrons (n) |
|---|---|---|---|---|---|
| 14C6 | 6 | 14 | 6 | 6 | 8 (14 - 6) |
| 15N7 | 7 | 15 | 7 | 7 | 8 (15 - 7) |
Isomers
- Isomers are nuclei of the same element with identical atomic number (Z) and mass number (A) but existing in different energy states. They often exhibit different radioactive decay properties and lifetimes.
Example: A metastable isomer such as 99mTc (technetium-99m) has the same Z and A as 99Tc but different nuclear energy. The 'm' indicates a metastable excited state with distinct radioactivity useful in medical diagnostics.
Isosters
- Isosters are molecules or ions of different substances that have the same number of atoms and the same total number of electrons, leading to similarities in some physical or chemical properties.
- Example: CO2 and N2O have the same number of electrons and may show certain analogous properties.
FAQs on Atomic Structure - Chemistry
| 1. What is the difference between electron, proton, and neutron in an atom? |  |
| 2. How do Bohr's model and the quantum mechanical model explain atomic structure differently? |  |
| 3. What exactly are electron shells, subshells, and orbitals, and how are they organised? |  |
| 4. Why do atoms have atomic number and mass number, and what's the connection to isotopes? |  |
| 5. What does valency mean, and how is it related to electron configuration in the outermost shell? |  |







