Entropy
Clausius theorem and Clausius inequality
Two reversible adiabatic paths cannot intersect each other since such an intersection would allow a violation of the Kelvin-Planck statement of the second law.
Any reversible path between the same end states may be substituted by a reversible zigzag path composed of a reversible adiabatic, followed by a reversible isotherm, and then another reversible adiabatic such that the heat transferred during the isothermal leg is the same as that transferred during the original process. From this construction the following result follows.
The cyclic integral of δQ/T for a reversible cycle is equal to zero:
∮ (δQ/T) = 0
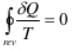
Entropy
The entropy of a system is a thermodynamic property that measures the degree of molecular disorder or randomness in the system. It is a state function and depends on the macroscopic state of the system (for example, pressure, temperature and composition). Entropy is related to the quantity of heat and the possibility of converting heat into work: when entropy is high, less of the heat energy is available to be converted into useful work; when entropy is low, more energy can be converted into work.
Characteristics of entropy
- Entropy increases when heat is supplied to a system, regardless of whether the temperature of the system changes or not.
- Entropy decreases when heat is removed from a system.
- Entropy remains unchanged in all adiabatic reversible processes.
- The increase in entropy due to adding a given amount of heat is smaller at high temperature and larger at low temperature.
Entropy in special processes
For a reversible adiabatic process, we have
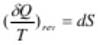
From which
dS = 0 (since dQ = 0)
Therefore, S = constant. Thus a reversible adiabatic process is an isentropic process.
For a reversible isothermal process at temperature T0, the entropy change is
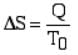
Clausius' inequality (Clausius theorem)
The Clausius theorem (1855) states that for any system exchanging heat with external reservoirs while undergoing a cyclic process (the system returns to its original state), the following inequality holds:
∮ δQ / Tsurr ≤ 0
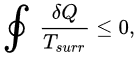
In this expression, δQ is the infinitesimal amount of heat absorbed by the system from a reservoir and Tsurr is the temperature of the external reservoir (surroundings) at that instant.
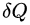
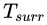
The equality sign applies for a reversible cycle and the inequality is strict for an irreversible cycle.
Entropy principle and isolated systems
Entropy principle:
For an isolated system:
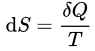
If the process is reversible the total entropy remains constant. For an irreversible process the total entropy increases. Thus the total entropy of an isolated system can never decrease over time; it is constant only when all processes are reversible. An isolated system spontaneously evolves towards thermodynamic equilibrium, the state of maximum entropy.
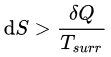
Key points:
- The entropy increase of an isolated system is a measure of the extent of irreversibility of the process.
- At equilibrium, any conceivable change in entropy is zero.
- This statement is equivalent to the second law of thermodynamics.
Applications of entropy principle
- Transfer of heat through a finite temperature difference results in entropy production (irreversibility).
- Mixing of two fluids leads to an entropy increase because the mixed state is more disordered.
Mixing of two fluids:
When two bodies are mixed thermally without work extraction, the final temperature tf of two masses m1, m2 with specific heats c1, c2 initially at temperatures T1, T2 (assuming no phase change and negligible heat loss) is given by energy balance:
m1 c1 T1 + m2 c2 T2 = (m1 c1 + m2 c2) Tf
Therefore
Tf = (m1 c1 T1 + m2 c2 T2) / (m1 c1 + m2 c2)
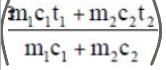
For two identical bodies (m1=m2, c1=c2) brought into thermal contact without work extraction,
Tf = (T1 + T2) / 2
Maximum work is obtainable from two finite identical bodies at temperatures T1 and T2 when one extracts work reversibly using a reversible heat engine between them while maintaining overall entropy balance; the final temperature for maximum work depends on the process and surroundings.
Final temperature of the two bodies (Tf) under different process constraints is shown in the referenced diagrams.
- The final temperature of two bodies initially at T1 and T2 can range from (T1 + T2)/2 when no work is delivered to a lower temperature when maximum work is extracted (see diagram).
Maximum work obtained with a finite body and a thermal energy reservoir (TER)
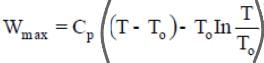
Here T is the temperature of the finite body and T0 is the temperature of the surroundings or thermal energy reservoir (TER).
- Maximum extractable work (available work or exergy decrease) when a system is brought reversibly to equilibrium with a reservoir at T0 is the decrease in availability:
Wmax = (Uinitial - Ufinal) - T0 (Sinitial - Sfinal)
Adiabatic dissipation of work
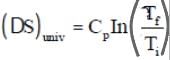
When work done on or by the system is dissipated internally (for example by friction) under adiabatic conditions, mechanical energy is converted into internal energy and entropy is generated. Adiabatic but irreversible processes are not isentropic because of entropy generation inside the system.
Adiabatic process vs isentropic process
- If an isentropic process is reversible, it must be adiabatic.
- If an adiabatic process is reversible, it must be isentropic.
- An adiabatic process may not be isentropic because entropy may increase due to friction or other internal irreversibilities.
- Change in the entropy of a system results from heat transfer and from internal irreversibilities (entropy generation).
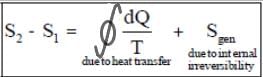
- Entropy is a state (point) function, but entropy generation is path dependent (a process quantity).
Useful relations and identities
| Equation | Holds good for |
|---|---|
dQ = dE + dW | Reversible, irreversible, any system (general energy form) |
dQ = dU + dW | Reversible, irreversible, closed system |
dQ = dU + p dV | Reversible, closed system (mechanical work = p dV) |
dQ = T dS | Reversible process (definition of entropy differential) |
T dS = dU + p dV | Closed system (general relation using first and second law; reversible form) |
T dS = dH - v dP | Closed system (relationship in terms of enthalpy) |
Entropy change when temperature of a solid or liquid changes
For a solid or liquid, the entropy change when temperature changes from T1 to T2 is given by the reversible integral
ΔS = ∫(δQrev / T) = ∫(m c(T) / T) dT
If the specific heat c is approximately constant over the temperature range, this integrates to
ΔS = m c ln(T2/T1)
where c is the specific heat for the solid or liquid (per unit mass) and m is mass. Use temperature in absolute units (kelvin) for entropy calculations.
Notes on entropy, availability and irreversibility
- The entropy generation in a process quantifies irreversibility: Sgen ≥ 0. For a reversible process Sgen = 0; for irreversible processes Sgen > 0.
- Entropy change for a system and its surroundings in any process satisfies: ΔSsystem + ΔSsurr = Sgen.
- When heat δQ flows across a finite temperature difference between a hot body at TH and a cold body at TC, entropy is produced; reversible transfer requires infinitesimal temperature difference and a reversible mechanism.
- Maximum useful work obtainable from a system undergoing a process between initial and final states in contact with a thermal reservoir at T0 is related to decrease in availability (exergy) and the net entropy change.
Worked expressions and simple derivations
Entropy change for a closed system under a reversible process:
ΔS = ∫(δQrev / T)
For constant specific heat c and mass m:
ΔS = ∫T1T2 (m c / T) dT
Therefore,
ΔS = m c ∫T1T2 (1 / T) dT
ΔS = m c [ln T]T1T2
Which gives
ΔS = m c ln(T2/T1)
Summary
Entropy is a state property that quantifies disorder and limits the conversion of heat into work. The Clausius inequality formalises the second law for cycles. For reversible cycles the cyclic integral of δQ/T is zero; for irreversible cycles it is negative. The entropy of an isolated system never decreases. Entropy change is computed from ΔS = ∫ δQrev/T and for solids/liquids with constant specific heat is m c ln(T2/T1). Entropy generation measures irreversibility and reduces the available work (exergy) that can be extracted from a thermodynamic system.
FAQs on Entropy
| 1. What is entropy in mechanical engineering? |  |
| 2. How is entropy related to the efficiency of mechanical systems? |  |
| 3. Can entropy be reduced or eliminated in mechanical systems? |  |
| 4. How is entropy calculated in mechanical engineering? |  |
| 5. What are some practical applications of entropy in mechanical engineering? |  |







