Water Requirements: Quality Standard | Civil Engineering SSC JE (Technical) - Civil Engineering (CE) PDF Download
| Table of contents |

|
| Introduction |

|
| Raw water source |

|
| Water quality |

|
| Water Quality Benchmarks as per IS 10500 : 2012 |

|
| Water Quality Requirements |

|
Introduction
Water quality is paramount for human health, environmental sustainability, and economic prosperity. It's shaped by numerous factors, including natural processes and human activities. Monitoring key parameters such as turbidity, pH, and microbial contaminants is essential for assessing water quality and guiding effective management strategies. Understanding these parameters is crucial for safeguarding water resources and ensuring access to clean and safe water for all.
In India, for water quality parameters we use the IS Code 10500:2012 as a standard for water quality. We will take a look at those standards later on.
Raw water source
 The various sources of water can be classified into two categories:
The various sources of water can be classified into two categories:
- Surface sources:
(a) Ponds and lakes
(b) Streams and rivers
(c) Storage reservoirs
(d) Oceans - Sub-surface sources:
(a) Springs
(b) Infiltration wells
(c) Wells
Water quality

The raw or treated water is analyzed by testing its physical, chemical, and bacteriological
characteristics.
Water= H2O + Dissolved gas+ Minerals+ Impurities
Useful minerals: Iron, calcium, magnesium, fluorine etc.
Harmful minerals: Arsenic, cadmium, cyanide, lead, chromium etc. Salts make water useless.
Bacteria causing diseases: Cholera, typhoid, paratyphoid, dysentery, hepatitis.
Physical characteristics of water
(i) Turbidity: It is due to suspended particles. It depends upon the fineness and concentration of particles. It can be measured as the interference to the passage of light through the given water.
1. Turbidity rod: It is to be dipped till the needle just disappears. That reading of rod gives the turbidity in mg/l. The calibration is based on suspension of silica.
2. Turbidity meters:
(a) Jackson turbidity meter: The depth of column of water required to cause the image of a candle flame at the bottom to diffuse into a uniform glow.
Units: JTU
(b) Baylis turbidity meter: The turbidity is measured by a technique to measure level of light scattered by the particles at right angles to the incident light beam. The scattered light is proportional to the particle concentration in the sample.
Units: NTU
Lake water: Turbidity < 25 units
Drinking water: Turbidity 10 to 25 units
(ii) Color: Dissolved organic matter from decaying vegetation or some inorganic materials may impart color to the water. It can be measured by comparing the color of water samples with other standard glass tubes (Nessler tubes) containing solutions of different standard color intensities. The standard unit of color is that which is produced by 1mg of platinum cobalt dissolved in 1 litre of distilled water.
Instrument: Tintometer.
The IS value for treated water is 5 to 25 cobalt units.
(iii) Taste and odour: The extent of taste or odour present in a particular sample of water is measured by a term called odour intensity, which is related with the threshold odour or threshold odour number.
Water to be tested is therefore gradually diluted with odour free water, and the mixture at which the detection of odour by human observation is just lost, is determined. The number of times the sample is diluted represents the threshold odour number.
For public supplies, the water should generally free from odour, i.e. the threshold number should be 1 and should never exceed 3.
(iv) Temperature: The increase in temperature decreases palatability, because at elevated temperatures, CO2 and other volatile gases are expelled. The ideal temperature of water for drinking purposes is 5 to 12ºC. Above 25ºC, water is not recommended for drinking.
Chemical characteristics
(i) Solids: The sum of suspended solids and dissolved solids is called total solids. Suspended solids can be found by filtration and total solids can be found by evaporation.
Allowable limit for total solids: 500ppm.
(ii) pH: pH value denotes the acidic or alkaline condition of water. It is expressed on a scale ranging from 0 to 14, which is common logarithm of the reciprocal of the H+ ion concentration. The recommended pH range for drinking water is 6.5 to 8.5.
(iii) Hardness: Hardness in water is that characteristic which prevents the formation of sufficient leather or foam when such hard waters are mixed with soap.The principal hardness causing cations are calcium, magnesium, strontium, ferrous and manganese ions. The major anions associated with these cations are sulphates, carbonates, bicarbonates, chlorides and nitrates.
There are two types of hardness:
1. Temporary hardness: Caused due to presence of carbonates and bicarbonates of calcium and magnesium this can be removed by boiling or by adding lime solution in water. Temporary hardness is also called carbonate hardness.
2. Permanent hardness: Caused due to the presence of sulphates, chlorites and nitrates of calcium and magnesium. They cannot be removed by simple boiling and require special treatment of water softening it is also called as Non-carbonate hardness.
Total hardness= temporary hardness+ permanent hardness
If total hardness < Alkalinity; carbonate hardness = total hardness
Permanent hardness = 0
If total hardness > Alkalinity; carbonate hardness = Alkalinity
Permanent hardness = Total- Alkalinity
(iv) Alkalinity: The alkalinity of water is a measure of its capacity to neutralise acids. It is expressed as mg/L in terms of calcium carbonate. The various forms of alkalinity are (a) hydroxide alkalinity, (b) carbonate alkalinity, (c) hydroxide plus carbonate alkalinity, (d) carbonate plus bicarbonate alkalinity, and (e) bicarbonate alkalinity, which is useful mainly in water softening and boiler feed water processes. Alkalinity is an important parameter in evaluating the optimum coagulant dosage.
(v) Chlorides: Chloride ion may be present in combination with one or more of the cations of calcium, magnesium, iron and sodium. Chlorides of these minerals are present in water because of their high solubility in water. The chloride content of treated water to be supplied to the public should not exceed a value of about 250 ppm. The chloride content of water can be measured by titrating the water with a standard silver nitrate solution using potassium chromate as indicator.
(vi) Nitrogen: The presence of nitrogen in water may occur in one or more of the following reasons:
- Free ammonia: It indicates very first stage of decomposition of organic matter. It should not exceed 0.15mg/l
- Albuminous or Organic Matter: It indicates the quantity of nitrogen present in water before the decomposition of organic molten has started. It should not exceed 0.3mg/l
- Nitrites: Not fully oxidized organic matter in water.
- Nitrates: It indicates fully oxidized organic matter in water (representing old pollution). Nitrates in water is not harmful.
However the presence of too much of nitrates in water may adversely affect the health of infants causing a disease called methemoglobinemia commonly called blue baby disease. Permissible limits of Nitrate is 45mg/L.
Ammonia nitrogen + organic nitrogen = Kjeldahl nitrogen
(vii) Fluoride: 1.5 ppm, excess of this effects human lungs and other respiratory organs.
< 1 mg causes dental cavities
(viii) Dissolved gases:
(a) H2S: bad taste and odour
(b) Methane: Explosive tendency
(c) CO2: Biological activity and corrosion
(d) C2: Deficiency indicates organic matter. 4 mg/ is required for Fish
Bacteriological characteristics
Water polluted by sewage contains one or more species of disease-producing pathogenic bacteria. Pathogenic organisms cause water-borne diseases and many non-pathogenic bacteria such as E.coli, a member of Coliform group, also live in the intestinal track of human beings. Coliform itself is not a harmful group but it has more resistance to adverse conditions than any other group. So if it is ensured to minimize the number of Coliforms, the harmful species will be very less. So, the Coliform group serves as an indicator of contamination of water with sewage and the presence of pathogens.
The methods to estimate the bacterial quality of water are:
(a) Standard plate count test: In this test, the bacteria are made to grow as colonies, by inoculating a known volume of sample into a solidifiable nutrient medium, which is poured in a Petri dish. After incubating (35º C) for a specified period (24 hours), the colonies of bacteria (as spots) are counted. The bacterial density is expressed as number of colonies per 100 ml of sample.
(b) Most probable number: MPN is a number which represents the bacterial density which is most likely to be present. E.coli is used as indicator of pollution. E.coli ferment lactose with gas formation with 48 hours of incubation at 35ºC. Based on this E.coli density in a sample is estimated by multiple tube fermentation procedure, which consists of the identification of E.coli in different dilution combinations. MPN value is calculated as follows.
Five 10ml (Five dilution combination) tubes of sample is tested for E.coli out of five only one gives positive test for E.coli and all others negative. From the tables MPN value for one positive and four negative results is read which is 2.2 in present case. The MPN value is expressed as 2.2 per 100ml.
(c) Membrane filter technique: In this test a known volume of water sample is filtered through a membrane with opening less than 0.5 microns. The bacteria present in the sample will be retained upon the filter paper. The filter paper is put in contact of a suitable nutrient medium and kept in an incubator for 24 hours at 35°C. The bacteria will grow upon the nutrient medium and visible colonies are counted. Each colony represents one bacterium of the original sample. The bacterial count is expressed as a number of colonies per 100 ml of sample.
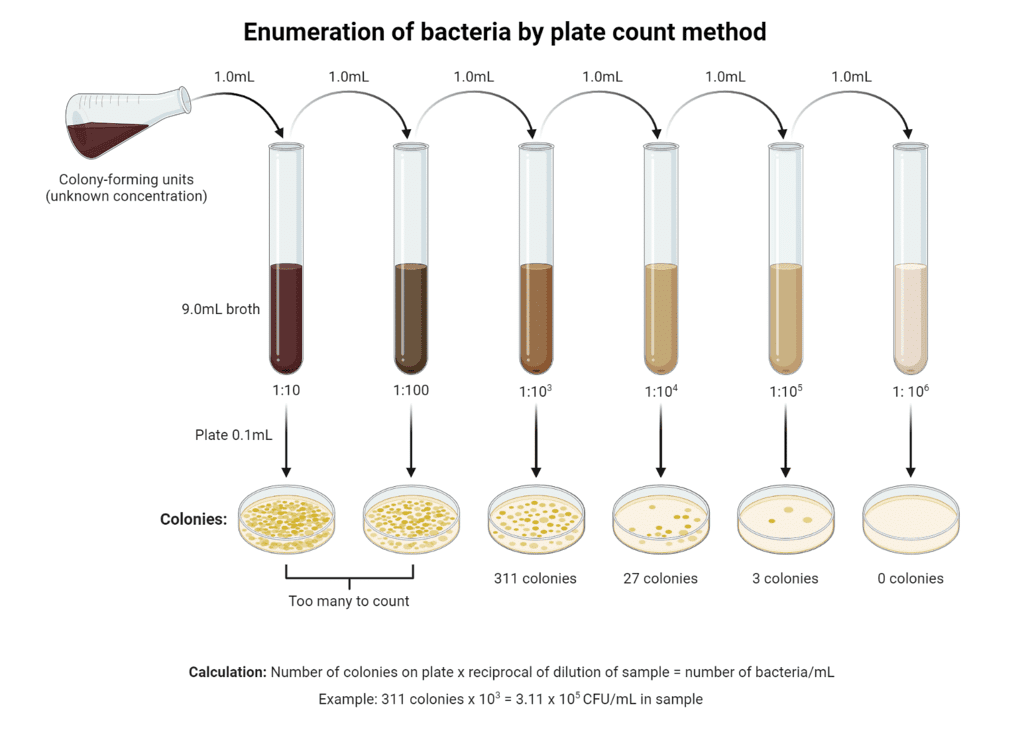 Plate Count Method
Plate Count Method
Radioactive Elements
- Radionuclides: Naturally occurring or anthropogenic radioactive elements like radium, uranium, and radon. Exposure to elevated levels can increase cancer risks and pose health hazards. Monitoring and treatment ensure compliance with regulatory limits.
Emerging Contaminants:
- Pharmaceuticals and Personal Care Products (PPCPs): Include medications, hormones, and cosmetics. PPCPs enter waterways through wastewater discharges and can impact aquatic ecosystems and human health. Monitoring and treatment technologies are evolving to address emerging contaminant challenges.
- Microplastics: Ubiquitous in water bodies due to plastic pollution. Microplastics can adsorb and transport contaminants, affecting aquatic organisms and food webs. Research on microplastic impacts and mitigation strategies is ongoing.
Water Quality Benchmarks as per IS 10500 : 2012
Now that we understand different quality degraders for water we can take a look at different benchmarks as given in Indian Standard code 10500.
Organoleptic and Physical Parameters:
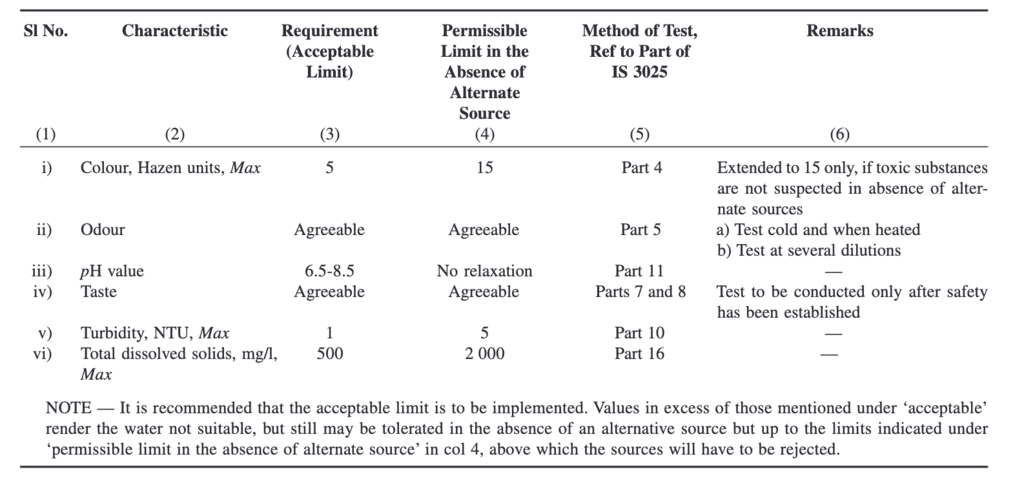
General Parameters Concerning Substances Undesirable in Excessive Amounts
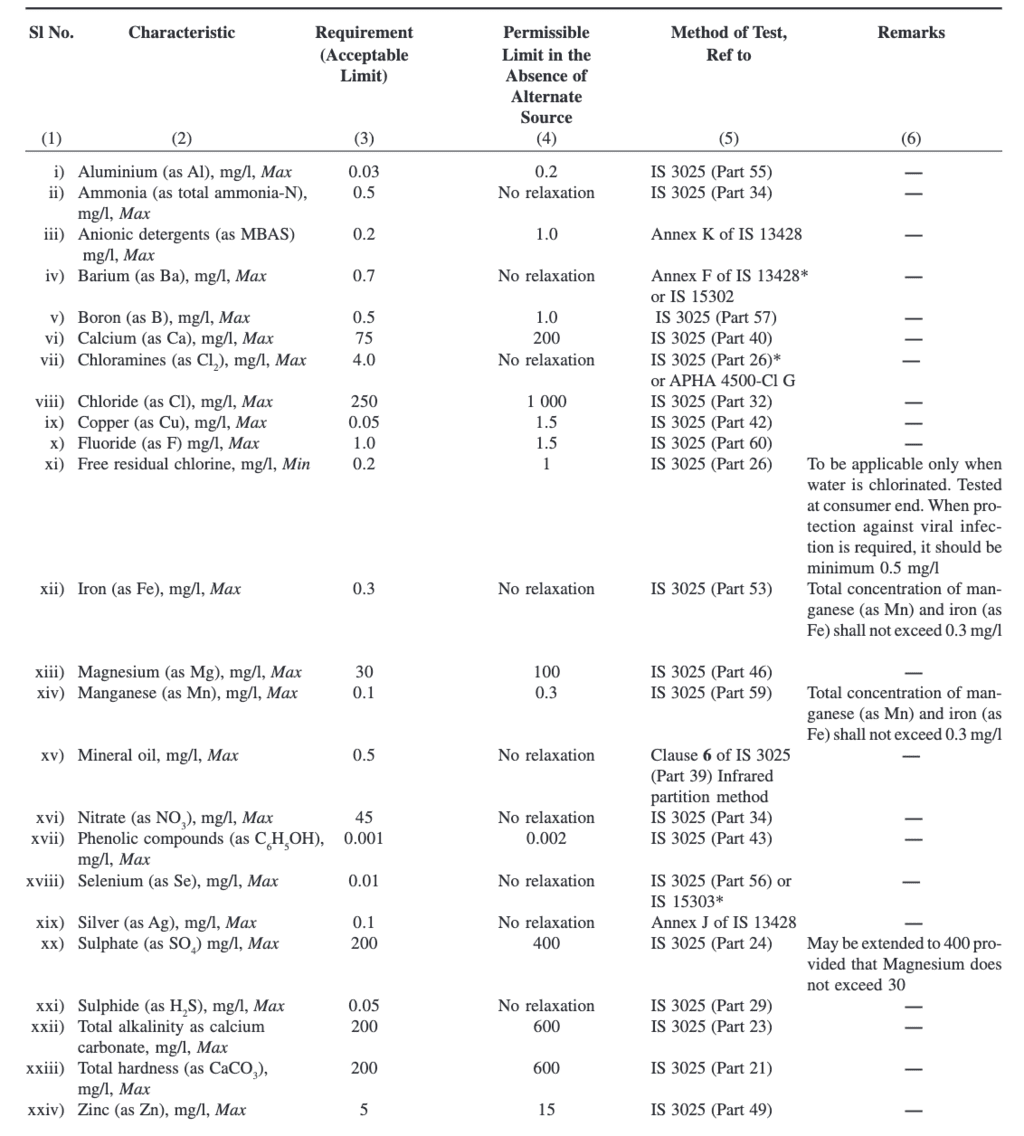
Parameters Concerning Toxic Substances
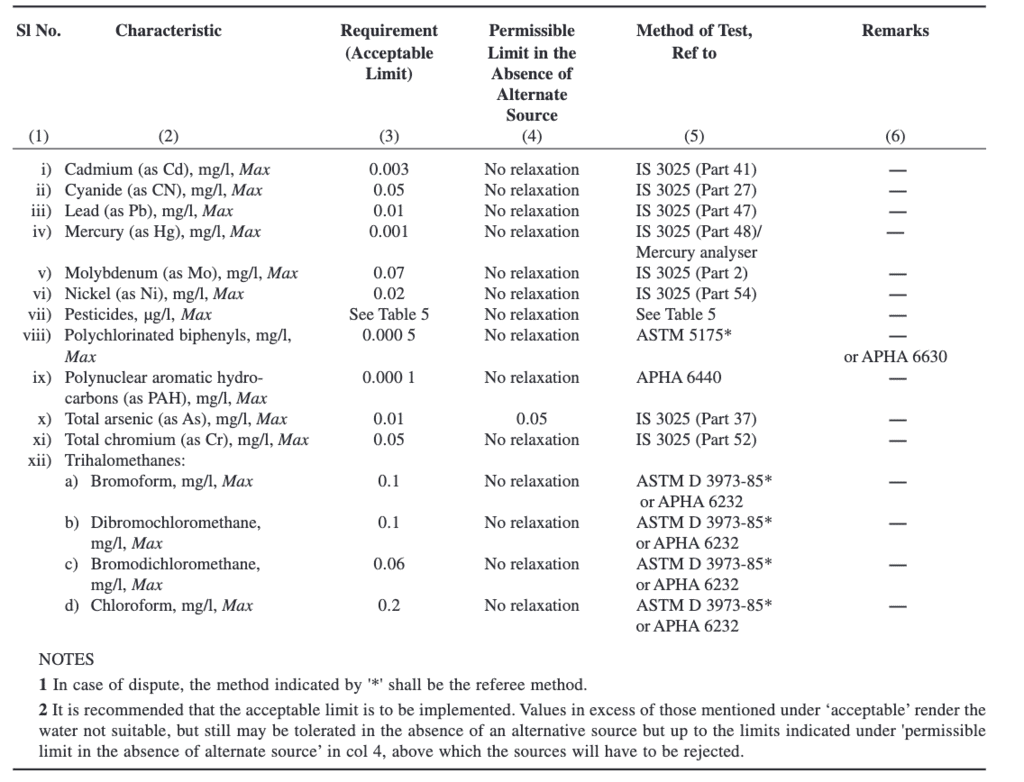
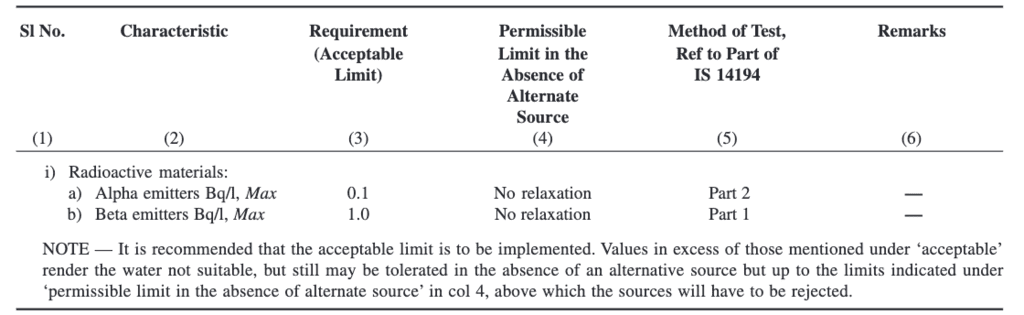
Parameters Concerning Radioactive Substances
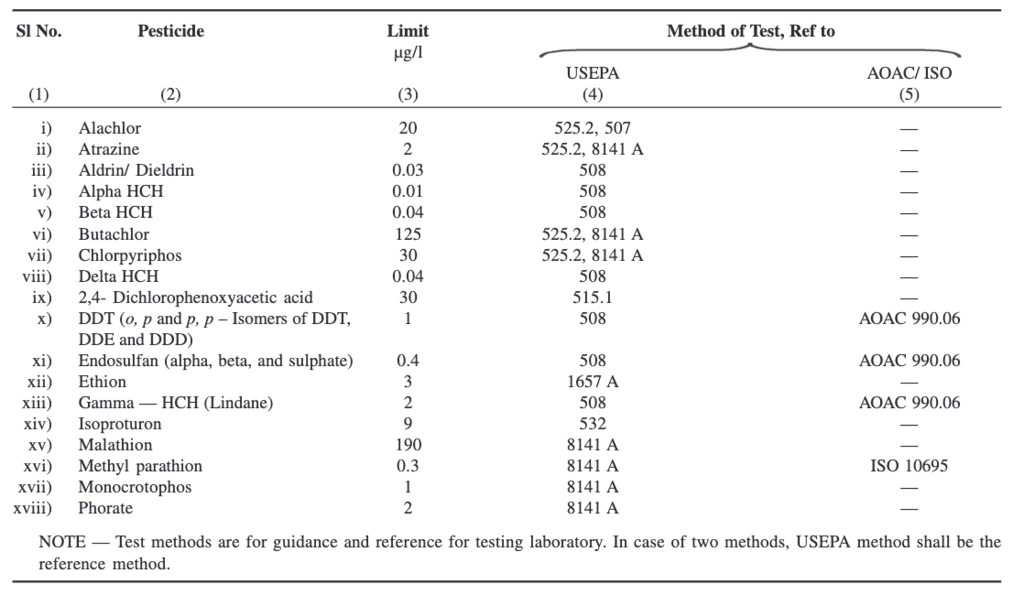
Pesticide Residues Limits and Test Method
Bacteriological Quality of Drinking Water

Classification of Water
- Salt Content Classification: Water is categorized based on salinity, with freshwater containing low dissolved salt levels (less than 0.5 ppt) and including lakes, rivers, and groundwater. Saltwater, with high salt concentrations like sodium chloride, is found in oceans.
- Source Classification: Surface water refers to water bodies like lakes and rivers exposed to the atmosphere, while groundwater resides in aquifers beneath the Earth's surface, accessed through wells and springs.
- Potability Classification: Potable water meets drinking standards after treatment to remove contaminants, while non-potable water is unfit for drinking and used for purposes like irrigation or industrial processes.
- Treatment Classification: Natural water exists without human intervention, encompassing rivers and lakes. Treated water undergoes processes to remove impurities and improve quality, suitable for drinking and household use.
- Nature Classification: Greywater originates from household activities like bathing and laundry, excluding sewage, and can be treated for reuse. Wastewater includes discharge from residential, commercial, and industrial sources, requiring extensive treatment before environmental release.
- Treatment Classification (Alternative): Raw water is untreated water from natural sources, while processed water undergoes treatment to meet quality standards and remove contaminants, also known as potable water.
Water Quality Requirements
- Water quality requirements: Standards, guidelines, or regulations ensuring acceptable quality and safety levels for water usage.
- Drinking Water Quality Standards: Set limits for physical, chemical, and biological contaminants like microbes, heavy metals, and pesticides to protect public health.
- Recreational Water Quality Standards: Ensure safety for activities like swimming by controlling microbial indicators to prevent infections.
- Agricultural and Irrigation Water Quality Guidelines: Address parameters such as salinity and ion concentrations to maintain soil and crop health.
- Industrial Water Quality Requirements: Vary based on processes and include factors like pH and suspended solids, impacting operational efficiency and safety.
- Environmental Water Quality Standards: Preserving natural water bodies and ecosystems by defining acceptable parameter levels to sustain aquatic life and biodiversity.
|
2 videos|133 docs|55 tests
|
FAQs on Water Requirements: Quality Standard - Civil Engineering SSC JE (Technical) - Civil Engineering (CE)
| 1. What are the primary raw water sources used for drinking water supply? |  |
| 2. What are the key parameters used to assess water quality? |  |
| 3. What are the water quality benchmarks as per IS 10500:2012? |  |
| 4. Why is it important to maintain water quality standards? |  |
| 5. What are the consequences of failing to meet water quality requirements? |  |
















