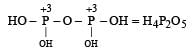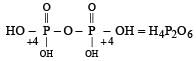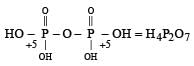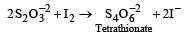All Exams >
NEET >
30-Day Revision Course for NEET >
All Questions
All questions of p-Block Elements for NEET Exam
Which is the strongest acid in the following : [NEET 2013]- a)HClO3
- b)HClO4
- c)H2SO3
- d)H2SO4
Correct answer is option 'B'. Can you explain this answer?
Which is the strongest acid in the following : [NEET 2013]
a)
HClO3
b)
HClO4
c)
H2SO3
d)
H2SO4

|
Surbhi Das answered |
HClO4 is the strongest acid amongst all because the oxidation state or Cl is maximum (+7).
Which of the following is a polar molecule ? [NEET 2013]- a)SF4
- b)SiF4
- c)XeF4
- d)BF3
Correct answer is option 'A'. Can you explain this answer?
Which of the following is a polar molecule ? [NEET 2013]
a)
SF4
b)
SiF4
c)
XeF4
d)
BF3

|
Rajesh Datta answered |
SF4 has 4 bond pairs and 1 lone pair of electrons, sp3d hybridisation leads to irregular shape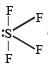 and resultant
and resultant
 and resultant
and resultantμ ≠ 0.
In the case of alkali metals, the covalent character decreases in the order: [2009]- a)MF > MCl > MBr > MI
- b)MF > MCl > MI > MBr
- c)MI > MBr > MCl > MF
- d)MCl > MI > MBr > MF
Correct answer is option 'C'. Can you explain this answer?
In the case of alkali metals, the covalent character decreases in the order: [2009]
a)
MF > MCl > MBr > MI
b)
MF > MCl > MI > MBr
c)
MI > MBr > MCl > MF
d)
MCl > MI > MBr > MF

|
Ruchi Chopra answered |
MI > MBr > MCl > MF. As the size of the anion decreases covalency decreases.
Among the following which is the strongest oxidising agent? [2009]- a)Br2
- b)I2
- c)Cl2
- d)F2
Correct answer is option 'D'. Can you explain this answer?
Among the following which is the strongest oxidising agent? [2009]
a)
Br2
b)
I2
c)
Cl2
d)
F2

|
Rajesh Datta answered |
Standard reduction potential of halogens are positive and decreases from fluorine to iodine. Therefore halogens act as strong oxidising agent and their oxidising power decreases from fluorine to iodine.
Which of the following is the most basic oxide?- a)Sb2O3
- b)Bi2O3 [2006]
- c)SeO2
- d)Al2O3
Correct answer is option 'B'. Can you explain this answer?
Which of the following is the most basic oxide?
a)
Sb2O3
b)
Bi2O3 [2006]
c)
SeO2
d)
Al2O3

|
Nilotpal Gupta answered |
More the oxidation state of the central atom (metal) more is its acidity. Hence SeO2 (O. S. of Se = +4) is acidic. Further for a given O.S., the basic character of the oxides increases with the increasing size of the central atom.
Thus Al2O3 and Sb2O3 are amphoteric and Bi2O3 is basic.
Thus Al2O3 and Sb2O3 are amphoteric and Bi2O3 is basic.
Which one of the following arrangements does not truly represent the property indicated against it?[2000]- a)Br2 < Cl2 < F2 : Electronegativity
- b)Br2 < F2 < Cl2 : Electron affinity
- c)Br2 < Cl2 < F2 : Bond energy
- d)Br2 < Cl2 < F2 : Oxidising power
Correct answer is option 'C'. Can you explain this answer?
Which one of the following arrangements does not truly represent the property indicated against it?[2000]
a)
Br2 < Cl2 < F2 : Electronegativity
b)
Br2 < F2 < Cl2 : Electron affinity
c)
Br2 < Cl2 < F2 : Bond energy
d)
Br2 < Cl2 < F2 : Oxidising power

|
Krish Khanna answered |
The bond energy of F2 < Cl2 due to more repulsion in between non-bonding electrons (2p) of F2 in comparision to non- bonding pair (3p) repulsion in Cl2, the bond energy of F2 < Cl2.
Bond energy (kJ mol–1) :
Bond energy (kJ mol–1) :

The formula for calcium chlorite is [1994]- a)Ca(ClO4)2
- b)Ca(ClO3)2
- c)CaClO2
- d)Ca(ClO2)2
Correct answer is option 'D'. Can you explain this answer?
The formula for calcium chlorite is [1994]
a)
Ca(ClO4)2
b)
Ca(ClO3)2
c)
CaClO2
d)
Ca(ClO2)2
|
|
Shraddha Choudhury answered |
The formula for calcium chlorite is Ca(ClO2)2. Let's break down the components of this compound to understand why this is the correct formula.
Calcium:
- Calcium is a metal with a +2 charge. It is represented by the symbol Ca in the periodic table.
Chlorite:
- Chlorite is a polyatomic ion with the formula ClO2-. It consists of one chlorine atom (Cl) bonded to two oxygen atoms (O), and it carries a -1 charge.
Formation of calcium chlorite:
- When calcium reacts with chlorite, the calcium ion (Ca2+) combines with two chlorite ions (ClO2-) to form calcium chlorite (Ca(ClO2)2).
- The calcium ion has a +2 charge, while each chlorite ion has a -1 charge. To balance the charges, two chlorite ions are needed for every calcium ion.
Explanation of options:
a) Ca(ClO4)2:
- This formula represents calcium perchlorate, not calcium chlorite. Perchlorate is a different polyatomic ion that consists of one chlorine atom and four oxygen atoms.
b) Ca(ClO3)2:
- This formula represents calcium chlorate, not calcium chlorite. Chlorate is another polyatomic ion that consists of one chlorine atom and three oxygen atoms.
c) CaClO2:
- This formula represents calcium hypochlorite, not calcium chlorite. Hypochlorite is a different polyatomic ion that consists of one chlorine atom and two oxygen atoms.
d) Ca(ClO2)2:
- This formula represents calcium chlorite, which is the correct compound. It consists of one calcium ion (Ca2+) bonded to two chlorite ions (ClO2-).
In summary, the correct formula for calcium chlorite is Ca(ClO2)2, as it consists of one calcium ion bonded to two chlorite ions.
Calcium:
- Calcium is a metal with a +2 charge. It is represented by the symbol Ca in the periodic table.
Chlorite:
- Chlorite is a polyatomic ion with the formula ClO2-. It consists of one chlorine atom (Cl) bonded to two oxygen atoms (O), and it carries a -1 charge.
Formation of calcium chlorite:
- When calcium reacts with chlorite, the calcium ion (Ca2+) combines with two chlorite ions (ClO2-) to form calcium chlorite (Ca(ClO2)2).
- The calcium ion has a +2 charge, while each chlorite ion has a -1 charge. To balance the charges, two chlorite ions are needed for every calcium ion.
Explanation of options:
a) Ca(ClO4)2:
- This formula represents calcium perchlorate, not calcium chlorite. Perchlorate is a different polyatomic ion that consists of one chlorine atom and four oxygen atoms.
b) Ca(ClO3)2:
- This formula represents calcium chlorate, not calcium chlorite. Chlorate is another polyatomic ion that consists of one chlorine atom and three oxygen atoms.
c) CaClO2:
- This formula represents calcium hypochlorite, not calcium chlorite. Hypochlorite is a different polyatomic ion that consists of one chlorine atom and two oxygen atoms.
d) Ca(ClO2)2:
- This formula represents calcium chlorite, which is the correct compound. It consists of one calcium ion (Ca2+) bonded to two chlorite ions (ClO2-).
In summary, the correct formula for calcium chlorite is Ca(ClO2)2, as it consists of one calcium ion bonded to two chlorite ions.
Noble gases do not react with other elements because [1994]- a)They are mono atomic
- b)They are found in abundance
- c)The size of their atoms is very small
- d)They are completely paired up and stable electron shells
Correct answer is option 'D'. Can you explain this answer?
Noble gases do not react with other elements because [1994]
a)
They are mono atomic
b)
They are found in abundance
c)
The size of their atoms is very small
d)
They are completely paired up and stable electron shells

|
Arnab Chavan answered |
Why noble gases do not react with other elements?
Noble gases, also known as inert gases, are a group of elements that include helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn). These gases are characterized by their extremely low reactivity, which is due to their stable electron configurations. The correct answer to the question is option 'D', which states that noble gases do not react with other elements because they have completely paired up and stable electron shells.
Explanation:
1. Noble gases have completely paired up electron shells:
- Noble gases have full valence electron shells, meaning that their outermost energy level is completely filled with electrons.
- For example, helium has two electrons in its outermost energy level, neon has eight, and so on.
- This complete electron configuration gives noble gases a high degree of stability and a lack of reactivity.
2. Stable electron configuration:
- The stability of noble gases arises from the fact that their electron configurations follow the octet rule.
- The octet rule states that atoms tend to gain, lose, or share electrons to achieve a complete outer shell with eight electrons (except for helium, which only requires two electrons).
- Noble gases already have this stable electron configuration, so they have no need to gain, lose, or share electrons with other elements.
3. Lack of unpaired electrons:
- In chemical reactions, elements typically react to achieve a stable electron configuration.
- Most elements achieve this stability by gaining, losing, or sharing electrons to fill their outer energy levels.
- Noble gases, however, already have their outer energy levels fully filled with electrons, so they do not require any additional electrons.
- This lack of unpaired electrons makes noble gases unreactive and unlikely to form chemical bonds with other elements.
Conclusion:
In summary, noble gases do not react with other elements because they have completely paired up and stable electron shells. Their full outer energy levels provide a high degree of stability, making them unreactive and giving them the name "inert gases." This lack of reactivity is a result of their already fulfilled electron configurations, which do not require any additional electrons to achieve stability.
Noble gases, also known as inert gases, are a group of elements that include helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn). These gases are characterized by their extremely low reactivity, which is due to their stable electron configurations. The correct answer to the question is option 'D', which states that noble gases do not react with other elements because they have completely paired up and stable electron shells.
Explanation:
1. Noble gases have completely paired up electron shells:
- Noble gases have full valence electron shells, meaning that their outermost energy level is completely filled with electrons.
- For example, helium has two electrons in its outermost energy level, neon has eight, and so on.
- This complete electron configuration gives noble gases a high degree of stability and a lack of reactivity.
2. Stable electron configuration:
- The stability of noble gases arises from the fact that their electron configurations follow the octet rule.
- The octet rule states that atoms tend to gain, lose, or share electrons to achieve a complete outer shell with eight electrons (except for helium, which only requires two electrons).
- Noble gases already have this stable electron configuration, so they have no need to gain, lose, or share electrons with other elements.
3. Lack of unpaired electrons:
- In chemical reactions, elements typically react to achieve a stable electron configuration.
- Most elements achieve this stability by gaining, losing, or sharing electrons to fill their outer energy levels.
- Noble gases, however, already have their outer energy levels fully filled with electrons, so they do not require any additional electrons.
- This lack of unpaired electrons makes noble gases unreactive and unlikely to form chemical bonds with other elements.
Conclusion:
In summary, noble gases do not react with other elements because they have completely paired up and stable electron shells. Their full outer energy levels provide a high degree of stability, making them unreactive and giving them the name "inert gases." This lack of reactivity is a result of their already fulfilled electron configurations, which do not require any additional electrons to achieve stability.
Which one of the following arrangements does not give the correct picture of the trends indicated against it ? [2008]- a)F2 > Cl2 > Br2 > I2 : Oxidizing power
- b)F2 > Cl2 > Br2 > I2 : Electron gain enthalpy
- c)F2 > Cl2 > Br2 > I2 : Bond dissociation energy
- d)F2 > Cl2 > Br2 > I2 : Electronegativity.
Correct answer is option 'B,C'. Can you explain this answer?
Which one of the following arrangements does not give the correct picture of the trends indicated against it ? [2008]
a)
F2 > Cl2 > Br2 > I2 : Oxidizing power
b)
F2 > Cl2 > Br2 > I2 : Electron gain enthalpy
c)
F2 > Cl2 > Br2 > I2 : Bond dissociation energy
d)
F2 > Cl2 > Br2 > I2 : Electronegativity.
|
|
Nisha Pillai answered |
From the given options we find option (a) is correct. The oxidising power of halogens follow the order F2 > Cl2 > Br2 > I2 . Option (b) is incorrect because it in not the correct order of electron gain enthalpy of halogens. The correct order is Cl2 > F2 > Br2 > I2 . The low value of F2 than Cl2 is due to its small size. Option (c) is incorrect. The correct order of bond dissociation energies of halogens is Cl2 > Br2 > F2 > I2 . Option (d) is correct. It is the correct order of electronegativity values of halogens. Thus option (b) and (c) are incorrect.
Which of the following oxy-acids has the maximum number of hydrogens directly attached to phosphorus? [1999]- a)H4P2O7
- b)H3PO2
- c)H3PO3
- d)H3PO4
Correct answer is option 'B'. Can you explain this answer?
Which of the following oxy-acids has the maximum number of hydrogens directly attached to phosphorus? [1999]
a)
H4P2O7
b)
H3PO2
c)
H3PO3
d)
H3PO4
|
|
Smrity answered |
In option a )no hydrogen attached directly to p
in option b) two H directly attached to P
in option c) one H directly attached to P
and in option d) no any hydrogen directly attached to P
so correct option is b
in option b) two H directly attached to P
in option c) one H directly attached to P
and in option d) no any hydrogen directly attached to P
so correct option is b
A solution of potassium bromide is treated with each of the following. Which one would liberate bromine ? [1993]- a)Hydrogen iodide
- b)Sulphur dioxide
- c)chlorine
- d)Iodine
Correct answer is option 'A'. Can you explain this answer?
A solution of potassium bromide is treated with each of the following. Which one would liberate bromine ? [1993]
a)
Hydrogen iodide
b)
Sulphur dioxide
c)
chlorine
d)
Iodine

|
Aashna Mukherjee answered |
A stronger oxidising agent (Cl2) displaces a weaker oxidising agent (Br2) from its salt solution.
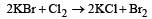
Polyanion formation is maximum in [1994]- a)Nitrogen
- b)Oxygen
- c)Sulphur
- d)Boron
Correct answer is option 'C'. Can you explain this answer?
Polyanion formation is maximum in [1994]
a)
Nitrogen
b)
Oxygen
c)
Sulphur
d)
Boron

|
Harshitha Dey answered |
Due to greater tendency for catenation, sulphur shows property of polyanion formation to a greater extent. For example, in polysulphides such as 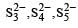
The acid which has a peroxy linkage is [1994]- a)Sulphurous acid
- b)Pyrosulphuric acid
- c)Dithionic acid
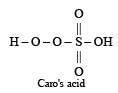
- d)Caro’s acid
Correct answer is option 'D'. Can you explain this answer?
The acid which has a peroxy linkage is [1994]
a)
Sulphurous acid
b)
Pyrosulphuric acid
c)
Dithionic acid
d)
Caro’s acid

|
Vaibhav Basu answered |
Caro’s acid is H2SO5 which contains one S – O – O – H peroxy linkage. It is also known as permonosulphuric acids.
Which of the following statements is false ? [1994]- a)Radon is obtained from the decay of radium
- b)Helium is inert gas
- c)Xenon is the most reactive among the rare gases
- d)The most abundant rare gas found in the atmosphere is helium
Correct answer is option 'D'. Can you explain this answer?
Which of the following statements is false ? [1994]
a)
Radon is obtained from the decay of radium
b)
Helium is inert gas
c)
Xenon is the most reactive among the rare gases
d)
The most abundant rare gas found in the atmosphere is helium
|
|
Abhijeet Menon answered |
False Statement:
The most abundant rare gas found in the atmosphere is helium. (Option D)
Explanation:
To determine the false statement among the given options, let's analyze each statement one by one:
a) Radon is obtained from the decay of radium:
This statement is true. Radon is a radioactive gas that is formed as a result of the decay of radium. Radium undergoes a series of radioactive decays, ultimately leading to the formation of radon.
b) Helium is inert gas:
This statement is true. Helium is classified as an inert gas because it has a stable electronic configuration with a complete outermost energy level. It does not readily react with other elements, making it chemically inert.
c) Xenon is the most reactive among the rare gases:
This statement is false. Xenon is actually the least reactive among the rare gases. Rare gases, also known as noble gases, are characterized by their low reactivity due to their stable electronic configurations. Xenon is particularly known for its low reactivity and is often used in lighting applications.
d) The most abundant rare gas found in the atmosphere is helium:
This statement is false. The most abundant rare gas found in the atmosphere is actually argon, not helium. Argon makes up about 0.93% of the Earth's atmosphere, making it the most abundant rare gas. Helium, on the other hand, is present in much smaller quantities and is only about 0.00052% of the atmosphere.
Therefore, the false statement is option D: The most abundant rare gas found in the atmosphere is helium.
The most abundant rare gas found in the atmosphere is helium. (Option D)
Explanation:
To determine the false statement among the given options, let's analyze each statement one by one:
a) Radon is obtained from the decay of radium:
This statement is true. Radon is a radioactive gas that is formed as a result of the decay of radium. Radium undergoes a series of radioactive decays, ultimately leading to the formation of radon.
b) Helium is inert gas:
This statement is true. Helium is classified as an inert gas because it has a stable electronic configuration with a complete outermost energy level. It does not readily react with other elements, making it chemically inert.
c) Xenon is the most reactive among the rare gases:
This statement is false. Xenon is actually the least reactive among the rare gases. Rare gases, also known as noble gases, are characterized by their low reactivity due to their stable electronic configurations. Xenon is particularly known for its low reactivity and is often used in lighting applications.
d) The most abundant rare gas found in the atmosphere is helium:
This statement is false. The most abundant rare gas found in the atmosphere is actually argon, not helium. Argon makes up about 0.93% of the Earth's atmosphere, making it the most abundant rare gas. Helium, on the other hand, is present in much smaller quantities and is only about 0.00052% of the atmosphere.
Therefore, the false statement is option D: The most abundant rare gas found in the atmosphere is helium.
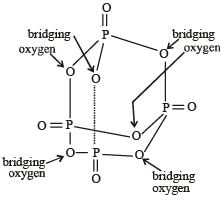
Which of the following statements is not valid for oxoacids of phosphorus? [2012]- a)Orthophosphoric acid is used in the manufacture of triple superphosphate.
- b)Hypophosphorous acid is a diprotic acid.
- c)All oxoacidscontaintetrahedral four coordinated phosphorus.
- d)All oxoacids contain atleast one P = O and one P — OH group.
Correct answer is option 'B'. Can you explain this answer?
Which of the following statements is not valid for oxoacids of phosphorus? [2012]
a)
Orthophosphoric acid is used in the manufacture of triple superphosphate.
b)
Hypophosphorous acid is a diprotic acid.
c)
All oxoacidscontaintetrahedral four coordinated phosphorus.
d)
All oxoacids contain atleast one P = O and one P — OH group.

|
Subham Chavan answered |
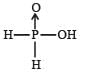
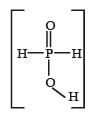 Hypophosphorous acid
Hypophosphorous acid(H3PO2) is a monobasic acid. i.e., it has only one ionisable hydrogen atom or one OH is present
Which of the following statements is not true ? [2003]- a)HF is a stronger acid than HCl
- b)Among halide ions, iodide is the most powerful reducing agent
- c)Fluorine is the only halogen that does not show a variable oxidation state
- d)HOCl is a stronger acid than HOBr
Correct answer is option 'A'. Can you explain this answer?
Which of the following statements is not true ? [2003]
a)
HF is a stronger acid than HCl
b)
Among halide ions, iodide is the most powerful reducing agent
c)
Fluorine is the only halogen that does not show a variable oxidation state
d)
HOCl is a stronger acid than HOBr

|
Sanjana Bajaj answered |
The correct answer is option A: HF is a stronger acid than HCl.
Explanation:
- In order to determine the strength of an acid, we need to compare their acidic properties, which can be measured by their ability to donate a proton (H+ ion) in a solution.
- The strength of an acid is determined by the stability of its conjugate base. A stronger acid will have a more stable conjugate base.
- In the given options, option A states that HF is a stronger acid than HCl. However, this statement is not true.
- HF is a weak acid, whereas HCl is a strong acid.
- The strength of an acid is based on the acidity constant (Ka). The higher the Ka value, the stronger the acid.
- For HF, the Ka value is relatively small (around 7 x 10^-4), indicating that it is a weak acid.
- On the other hand, HCl has a much larger Ka value (around 1 x 10^6), indicating that it is a strong acid.
- The strength of an acid is also influenced by the polarity of the bond between the hydrogen atom and the halogen atom. The more polar the bond, the stronger the acid.
- In the case of HF, the bond between hydrogen and fluorine is highly polar due to the large electronegativity difference between the two atoms, making HF a weak acid.
- However, in the case of HCl, the bond between hydrogen and chlorine is less polar, resulting in a stronger acid.
- Therefore, option A is not true as HF is a weaker acid compared to HCl.
Explanation:
- In order to determine the strength of an acid, we need to compare their acidic properties, which can be measured by their ability to donate a proton (H+ ion) in a solution.
- The strength of an acid is determined by the stability of its conjugate base. A stronger acid will have a more stable conjugate base.
- In the given options, option A states that HF is a stronger acid than HCl. However, this statement is not true.
- HF is a weak acid, whereas HCl is a strong acid.
- The strength of an acid is based on the acidity constant (Ka). The higher the Ka value, the stronger the acid.
- For HF, the Ka value is relatively small (around 7 x 10^-4), indicating that it is a weak acid.
- On the other hand, HCl has a much larger Ka value (around 1 x 10^6), indicating that it is a strong acid.
- The strength of an acid is also influenced by the polarity of the bond between the hydrogen atom and the halogen atom. The more polar the bond, the stronger the acid.
- In the case of HF, the bond between hydrogen and fluorine is highly polar due to the large electronegativity difference between the two atoms, making HF a weak acid.
- However, in the case of HCl, the bond between hydrogen and chlorine is less polar, resulting in a stronger acid.
- Therefore, option A is not true as HF is a weaker acid compared to HCl.
During its reactions, ozone [1999]- a)can only combine with hydrogen atoms
- b)accepts electrons
- c)loses electrons
- d)shows the role of electrons to be irrelevant
Correct answer is option 'A'. Can you explain this answer?
During its reactions, ozone [1999]
a)
can only combine with hydrogen atoms
b)
accepts electrons
c)
loses electrons
d)
shows the role of electrons to be irrelevant
|
|
Abhijeet Menon answered |
Answer:
Ozone (O3) is a molecule composed of three oxygen atoms. It is formed in the atmosphere through a series of chemical reactions involving oxygen molecules (O2) and ultraviolet radiation. Ozone is an important component of the Earth's atmosphere as it plays a crucial role in absorbing harmful ultraviolet (UV) radiation from the sun.
During its reactions, ozone can undergo various processes, including the following:
1. Ozone can combine with hydrogen atoms:
Ozone can react with hydrogen atoms to form water molecules. This reaction is known as hydrogenation, and it occurs under specific conditions. However, it is important to note that ozone can also react with other elements and compounds, not just hydrogen atoms.
2. Ozone accepts electrons:
Ozone is an oxidizing agent, meaning it has the ability to accept electrons from other substances. In many chemical reactions, ozone acts as an electron acceptor, gaining electrons to form stable compounds. This process is part of its role as an oxidant.
3. Ozone loses electrons:
While ozone can accept electrons, it can also lose electrons in certain reactions. When ozone loses an electron, it forms an oxygen molecule (O2). This process is known as reduction, where ozone acts as a reducing agent.
4. The role of electrons is relevant:
The statement that ozone shows the role of electrons to be irrelevant is incorrect. In fact, the role of electrons is crucial in ozone reactions. Electrons are involved in the transfer of charge, which allows ozone to act as an oxidizing or reducing agent. The gain or loss of electrons determines the outcome of the reaction and the formation of new compounds.
In summary, ozone can undergo various reactions, including combining with hydrogen atoms, accepting electrons, and losing electrons. The role of electrons is essential in these reactions, as they determine the behavior of ozone as an oxidizing or reducing agent. Therefore, the correct answer is option 'A' - ozone can only combine with hydrogen atoms.
Ozone (O3) is a molecule composed of three oxygen atoms. It is formed in the atmosphere through a series of chemical reactions involving oxygen molecules (O2) and ultraviolet radiation. Ozone is an important component of the Earth's atmosphere as it plays a crucial role in absorbing harmful ultraviolet (UV) radiation from the sun.
During its reactions, ozone can undergo various processes, including the following:
1. Ozone can combine with hydrogen atoms:
Ozone can react with hydrogen atoms to form water molecules. This reaction is known as hydrogenation, and it occurs under specific conditions. However, it is important to note that ozone can also react with other elements and compounds, not just hydrogen atoms.
2. Ozone accepts electrons:
Ozone is an oxidizing agent, meaning it has the ability to accept electrons from other substances. In many chemical reactions, ozone acts as an electron acceptor, gaining electrons to form stable compounds. This process is part of its role as an oxidant.
3. Ozone loses electrons:
While ozone can accept electrons, it can also lose electrons in certain reactions. When ozone loses an electron, it forms an oxygen molecule (O2). This process is known as reduction, where ozone acts as a reducing agent.
4. The role of electrons is relevant:
The statement that ozone shows the role of electrons to be irrelevant is incorrect. In fact, the role of electrons is crucial in ozone reactions. Electrons are involved in the transfer of charge, which allows ozone to act as an oxidizing or reducing agent. The gain or loss of electrons determines the outcome of the reaction and the formation of new compounds.
In summary, ozone can undergo various reactions, including combining with hydrogen atoms, accepting electrons, and losing electrons. The role of electrons is essential in these reactions, as they determine the behavior of ozone as an oxidizing or reducing agent. Therefore, the correct answer is option 'A' - ozone can only combine with hydrogen atoms.
About 20 km above the earth, there is an ozone layer. Which one of the following statements about ozone and ozone layer is true? [1995]- a)ozone has a triatomic linear molecule
- b)it is harmful as it stops useful radiation
- c)it is beneficial to us as it stops U.V radiation
- d)conversion of O3 to O2 is an endothermic reaction
Correct answer is option 'C'. Can you explain this answer?
About 20 km above the earth, there is an ozone layer. Which one of the following statements about ozone and ozone layer is true? [1995]
a)
ozone has a triatomic linear molecule
b)
it is harmful as it stops useful radiation
c)
it is beneficial to us as it stops U.V radiation
d)
conversion of O3 to O2 is an endothermic reaction

|
Mehul Choudhary answered |
Understanding the Ozone Layer
The ozone layer plays a crucial role in protecting life on Earth. It is located about 20 km above the surface and absorbs most of the sun's harmful ultraviolet (UV) radiation.
Why Statement C is Correct
- Protection from UV Radiation: Ozone (O3) molecules effectively absorb UV radiation, particularly the more harmful UV-B and UV-C types. This absorption prevents these rays from reaching the Earth's surface, reducing the risk of skin cancer, cataracts, and other health issues in humans.
- Environmental Benefits: By filtering out harmful UV radiation, the ozone layer also protects ecosystems, including phytoplankton in oceans, which are vital for the marine food web and global oxygen production.
Why Other Statements are Incorrect
- Statement A (Triatomic Linear Molecule): Ozone is a triatomic molecule, but it is not linear; it has a bent molecular geometry due to the arrangement of its oxygen atoms.
- Statement B (Harmful as it Stops Useful Radiation): While ozone does absorb UV radiation, it does not stop all useful radiation. It selectively filters out harmful UV rays while allowing visible light to pass through, which is essential for life.
- Statement D (Endothermic Reaction): The conversion of O3 to O2 is actually an exothermic reaction, releasing energy. This process occurs naturally in the atmosphere and is crucial for maintaining the balance of ozone.
Conclusion
In summary, the ozone layer is beneficial as it protects living organisms from harmful UV radiation, making option C the correct statement regarding ozone and its layer.
The ozone layer plays a crucial role in protecting life on Earth. It is located about 20 km above the surface and absorbs most of the sun's harmful ultraviolet (UV) radiation.
Why Statement C is Correct
- Protection from UV Radiation: Ozone (O3) molecules effectively absorb UV radiation, particularly the more harmful UV-B and UV-C types. This absorption prevents these rays from reaching the Earth's surface, reducing the risk of skin cancer, cataracts, and other health issues in humans.
- Environmental Benefits: By filtering out harmful UV radiation, the ozone layer also protects ecosystems, including phytoplankton in oceans, which are vital for the marine food web and global oxygen production.
Why Other Statements are Incorrect
- Statement A (Triatomic Linear Molecule): Ozone is a triatomic molecule, but it is not linear; it has a bent molecular geometry due to the arrangement of its oxygen atoms.
- Statement B (Harmful as it Stops Useful Radiation): While ozone does absorb UV radiation, it does not stop all useful radiation. It selectively filters out harmful UV rays while allowing visible light to pass through, which is essential for life.
- Statement D (Endothermic Reaction): The conversion of O3 to O2 is actually an exothermic reaction, releasing energy. This process occurs naturally in the atmosphere and is crucial for maintaining the balance of ozone.
Conclusion
In summary, the ozone layer is beneficial as it protects living organisms from harmful UV radiation, making option C the correct statement regarding ozone and its layer.
By passing H2S gas in acidified KMnO4 solution, we get [1995]- a)S
- b)K2S
- c)MnO2
- d)K2SO3
Correct answer is option 'A'. Can you explain this answer?
By passing H2S gas in acidified KMnO4 solution, we get [1995]
a)
S
b)
K2S
c)
MnO2
d)
K2SO3

|
Pooja Choudhary answered |

Thus in this reaction sulphur (S) is produced.
Which of the following oxides will be the least acidic?[1996]- a)As4O6
- b)As4O10
- c)P4O10
- d)P4O6
Correct answer is option 'A'. Can you explain this answer?
Which of the following oxides will be the least acidic?[1996]
a)
As4O6
b)
As4O10
c)
P4O10
d)
P4O6

|
Dipanjan Chawla answered |
As th e O.N of the central atom of the compounds increases acidic strength of that compound also increases and on moving from top to bottom in groups acidic strength of oxides also decrease due to decreasing electronegativity in groups.
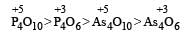
The correct order of increasing bond angles in the following species are : [2010]- a)
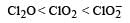
- b)
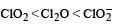
- c)
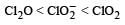
- d)
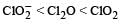
Correct answer is option 'C'. Can you explain this answer?
The correct order of increasing bond angles in the following species are : [2010]
a)
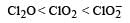
b)
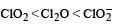
c)
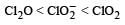
d)
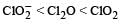

|
Yash Saha answered |
The correct order of increasing bond angle is 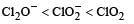
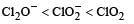
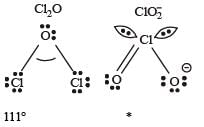
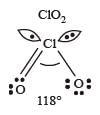
* In ClO2– there are 2 lone pairs of electrons present on the central chlorine atom. Therefore the bond angle in ClO2– is less than 118° which is the bond angle in ClO2 which has less number of electrons on chlorine.
Which one of the following orders correctly represents the increasing acid strengths of the given acids? [2007]- a)HOClO < HOCl < HOClO3 < HOClO2
- b)HOClO2 < HOClO3 < HOClO < HOCl
- c)HOClO3 < HOClO2 < HOClO < HOCl
- d)HOCl < HOClO < HOClO2 < HOClO3
Correct answer is option 'D'. Can you explain this answer?
Which one of the following orders correctly represents the increasing acid strengths of the given acids? [2007]
a)
HOClO < HOCl < HOClO3 < HOClO2
b)
HOClO2 < HOClO3 < HOClO < HOCl
c)
HOClO3 < HOClO2 < HOClO < HOCl
d)
HOCl < HOClO < HOClO2 < HOClO3
|
|
Poulomi Desai answered |
The correct order of increasing acid strength is:
d) HCOOH < ch3cooh="">< hno3="">< hclo4.="">
d) HCOOH < ch3cooh="">< hno3="">< hclo4.="">
In which of the following arrangements the given sequence is not strictly according to the property indicated against it ? [2012 M]- a)HF < HCl < HBr < HI : increasing acidic strength
- b)H2O < H2S < H2Se < H2Te : increasing pKa values
- c)NH3 < PH3 < AsH3 < SbH3 : increasing acidic character
- d)CO2 < SiO2 < SnO2 < PbO2 : increasing oxidising power
Correct answer is option 'B'. Can you explain this answer?
In which of the following arrangements the given sequence is not strictly according to the property indicated against it ? [2012 M]
a)
HF < HCl < HBr < HI : increasing acidic strength
b)
H2O < H2S < H2Se < H2Te : increasing pKa values
c)
NH3 < PH3 < AsH3 < SbH3 : increasing acidic character
d)
CO2 < SiO2 < SnO2 < PbO2 : increasing oxidising power

|
Bhargavi Choudhury answered |
If acidic nature is high, Ka is high and pka is low
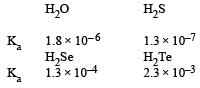
since pka = – log ka
Hence the order of pka will be H2O > H2S > H2Se > H2Te
Hence the order of pka will be H2O > H2S > H2Se > H2Te
Which one of the following orders is not in accordance with the property stated against is ? [2006]- a)HI > HBr > HCl > HF : Acidic property in water
- b)F2 > Cl2 > Br2 > I2 : Electronegativity
- c)F2 > Cl2 > Br2 > I2 : Bond dissociation energy
- d)F2 > Cl2 > Br2 > I2 : Oxidising power
Correct answer is option 'C'. Can you explain this answer?
Which one of the following orders is not in accordance with the property stated against is ? [2006]
a)
HI > HBr > HCl > HF : Acidic property in water
b)
F2 > Cl2 > Br2 > I2 : Electronegativity
c)
F2 > Cl2 > Br2 > I2 : Bond dissociation energy
d)
F2 > Cl2 > Br2 > I2 : Oxidising power

|
Shalini Saha answered |
Bond dissociation energy of fluorine is less because of its small size and repulsion between electrons of two atoms. So option (c) is wrong order. The correct order is [Cl2 > Br2 > F2 > I2]
Which of the following species has the highest dipole moment ? [1997]- a)NH3
- b)PH3
- c)AsH3
- d)SbH3
Correct answer is option 'A'. Can you explain this answer?
Which of the following species has the highest dipole moment ? [1997]
a)
NH3
b)
PH3
c)
AsH3
d)
SbH3

|
Harshitha Dey answered |
Order of dipole moment decreases as NH3 > PH3 > AsH3 > SbH3 (Based upon electronegativity)
Which one is the correct order of the size of iodine species? [1997]- a)I > I+ > I–
- b)I > I– > I+
- c)I+ > I– > I
- d)I– > I > I+
Correct answer is option 'D'. Can you explain this answer?
Which one is the correct order of the size of iodine species? [1997]
a)
I > I+ > I–
b)
I > I– > I+
c)
I+ > I– > I
d)
I– > I > I+

|
Ayush Sengupta answered |
We know that positive ion is always smaller and negative ion is always larger than the corresponding atom. Therefore the correct order of the size is I- > I > I+
Which of the following oxide is amphoteric ?- a)SnO2
- b)CaO [2011 M]
- c)SiO2
- d)CO2
Correct answer is option 'A'. Can you explain this answer?
Which of the following oxide is amphoteric ?
a)
SnO2
b)
CaO [2011 M]
c)
SiO2
d)
CO2

|
Bhargavi Choudhury answered |
SnO2 is an amphoteric oxide because it reacts with acids as well as bases to form corresponding salts.
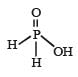
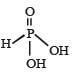
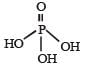
The structural formula of hypophosphorous acid is- a)
- b)
- c)
- d)
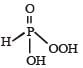
Correct answer is option 'A'. Can you explain this answer?
The structural formula of hypophosphorous acid is
a)

b)

c)

d)


|
Palak Khanna answered |
We know that empirical formula of hypophosphorus acid is H3PO2. In this only one ionisable hydrogen atom is present i.e. it is monobasic. Therefore option (1) is correct structural formula of it.
A certain compound (X) when treated with copper sulphate solution yields a brown precipitate. On adding hypo solution, the precipitate turns white.The compound is- a)K2CO3
- b)KI [1994]
- c)KBr
- d)K3PO4
Correct answer is option 'B'. Can you explain this answer?
A certain compound (X) when treated with copper sulphate solution yields a brown precipitate. On adding hypo solution, the precipitate turns white.The compound is
a)
K2CO3
b)
KI [1994]
c)
KBr
d)
K3PO4

|
Anand Jain answered |
KI reacts with CuSO4 solution to produce cuprous iodide (white precipitate) and I2 (which gives brown colour) Iodine reacts with hypo (Na2S2O35H2O) solution.
Decolourisaiton of solution shows the appearance of white precipitate.
Decolourisaiton of solution shows the appearance of white precipitate.
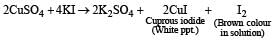
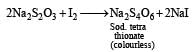
Match List - I (substan ces) with List - II (processes) employed in the manufacture of the substances and select the correct option. [2010]
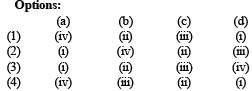
- a)a
- b)b
- c)c
- d)d
Correct answer is option 'A'. Can you explain this answer?
Match List - I (substan ces) with List - II (processes) employed in the manufacture of the substances and select the correct option. [2010]

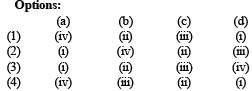
a)
a
b)
b
c)
c
d)
d

|
Maya Sengupta answered |
(a) Sulphuric acid (iv) Contact process
(b) Steel (ii) Bessemer ’s process
(c) Sodium hydroxide(iii) Leblanc process
(d) Ammonia (i) Haber ’s process
∴ Correct choice : (a)
(b) Steel (ii) Bessemer ’s process
(c) Sodium hydroxide(iii) Leblanc process
(d) Ammonia (i) Haber ’s process
∴ Correct choice : (a)
HI can be prepared by all the following methods, except[1994]- a)PI3 + H2O
- b)KI + H2SO4
- c)
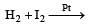
- d)I2 + H 2S
Correct answer is option 'B'. Can you explain this answer?
HI can be prepared by all the following methods, except[1994]
a)
PI3 + H2O
b)
KI + H2SO4
c)
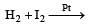
d)
I2 + H 2S

|
Pooja Choudhary answered |
HI cannot be prepared by heating iodides with concentrated H2SO4 because HI is a strong reducing agent and reduce H2SO4 to SO2 and get themselves oxidised to iodide.
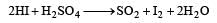
Which of the following fertilizers has the highest nitrogen percentage ? [1993]- a)Ammonium sulphate
- b)Calcium cyanamide
- c)Urea
- d)Ammonium nitrate
Correct answer is option 'C'. Can you explain this answer?
Which of the following fertilizers has the highest nitrogen percentage ? [1993]
a)
Ammonium sulphate
b)
Calcium cyanamide
c)
Urea
d)
Ammonium nitrate

|
Ayush Choudhury answered |
Urea (46.6%N). % of N in other compound are : ( NH4)2 SO4 = 21.2%; CaCN2 = 35.0% and NH4 NO3 = 35.0%
Repeated use of which one of the following fertilizers would increase the acidity of the soil?- a)Urea [1998]
- b)Superphosphate of lime
- c)Ammonium sulphate
- d)Potassium nitrate
Correct answer is option 'C'. Can you explain this answer?
Repeated use of which one of the following fertilizers would increase the acidity of the soil?
a)
Urea [1998]
b)
Superphosphate of lime
c)
Ammonium sulphate
d)
Potassium nitrate

|
Jatin Chakraborty answered |
Ammonium sulphate is a salt of weak base and strong acid, so it produces acidity. Hence aqueous solution of ammonium sulphate is increases the acidity of soil.
Which one of the following oxides of chlorine is obtained by passing dry chlorine over silver chlorate at 90°C ? [1994]- a)Cl2O
- b)ClO3
- c)ClO2
- d)ClO4
Correct answer is option 'C'. Can you explain this answer?
Which one of the following oxides of chlorine is obtained by passing dry chlorine over silver chlorate at 90°C ? [1994]
a)
Cl2O
b)
ClO3
c)
ClO2
d)
ClO4

|
Dipanjan Chawla answered |
Pure ClO2 is obtained by passing dry Cl2 over AgClO3 at 90°C.
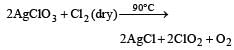
Which among the following is paramagnetic ? [1994]- a)Cl2O
- b)ClO2
- c)Cl2O7
- d)Cl2O6
Correct answer is option 'B'. Can you explain this answer?
Which among the following is paramagnetic ? [1994]
a)
Cl2O
b)
ClO2
c)
Cl2O7
d)
Cl2O6

|
Akshat Chavan answered |
ClO2 contain s 7 + 12 i.e. 19 electr ons (valence) which is an odd number, i.e. there is (are) free electron(s). Hence it is paramagnetic in nature.
Which is the best description of the behaviour of bromine in the reaction given below ? [2004] H2O + Br2 → HOBr + HBr- a)Proton acceptor only
- b)Both oxidized and reduced
- c)Oxidized only
- d)Reduced only
Correct answer is option 'B'. Can you explain this answer?
Which is the best description of the behaviour of bromine in the reaction given below ? [2004] H2O + Br2 → HOBr + HBr
a)
Proton acceptor only
b)
Both oxidized and reduced
c)
Oxidized only
d)
Reduced only

|
Palak Khanna answered |
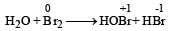
Thus here oxidation number of Br increases from 0 to +1 and also decreases from 0 to –1.
Thus it is oxidised as well as reduced
Thus it is oxidised as well as reduced
Which of the following structures is the most preferred and hence of lowest energy for SO3 ? [2011 M]- a)
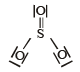
- b)
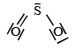
- c)
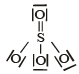
- d)
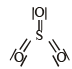
Correct answer is option 'D'. Can you explain this answer?
Which of the following structures is the most preferred and hence of lowest energy for SO3 ? [2011 M]
a)
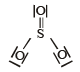
b)
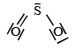
c)
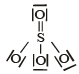
d)
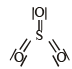

|
Maya Sengupta answered |
Formal charges help in the selection of the lowest energy structure from a number of possible Lewis structures for a given compound. The lowest energy structure means the structure with the smallest formal charge on each atom of the compound. A Lewis dot structure is preferable when all formal charges are zero.
A one litre flask is full of brown bromine vapour.The intensity of brown colour of vapour will not decrease appreciably on adding to the flask some[1998]- a)pieces of marble
- b)animal charcoal powder
- c)carbon tetrachloride
- d)carbon disulphide
Correct answer is option 'A'. Can you explain this answer?
A one litre flask is full of brown bromine vapour.The intensity of brown colour of vapour will not decrease appreciably on adding to the flask some[1998]
a)
pieces of marble
b)
animal charcoal powder
c)
carbon tetrachloride
d)
carbon disulphide

|
Nilotpal Gupta answered |
Due to absence of reaction between marble and bromine
Nitrogen forms N2, but phosphorus is converted into P4 from P, the reason is [2001]- a)Triple bond is present between phosphorus atom
- b)pπ – pπ bonding is strong
- c)pπ – pπ bonding is weak
- d)Multiple bond is formed easily
Correct answer is option 'C'. Can you explain this answer?
Nitrogen forms N2, but phosphorus is converted into P4 from P, the reason is [2001]
a)
Triple bond is present between phosphorus atom
b)
pπ – pπ bonding is strong
c)
pπ – pπ bonding is weak
d)
Multiple bond is formed easily

|
Moumita Khanna answered |
Nitrogen form N2 (i.e. N ≡ N) but phosphorus form P4, because in P2, pπ — pπ bonding is present which is a weaker bonding.
Brown ring test is used to detect [1994]- a)Iodine
- b)Nitrate
- c)Iron
- d)Bromide
Correct answer is option 'B'. Can you explain this answer?
Brown ring test is used to detect [1994]
a)
Iodine
b)
Nitrate
c)
Iron
d)
Bromide

|
Jatin Chakraborty answered |
Brown ring test is done for confirmation of
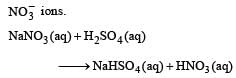
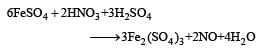

Regarding F– and Cl– which of the following statements is/are correct? [1996]
(i) Cl– can give up an electron more easily than F –
(ii) Cl– is a better reducing agent than F–
(iii) Cl– is smaller in size than F–
(iv)F– can be oxidized more readily than Cl–- a)(i) and (ii)
- b)(i), (ii) and (iv)
- c)(iii) and (iv)
- d)only (i)
Correct answer is option 'D'. Can you explain this answer?
Regarding F– and Cl– which of the following statements is/are correct? [1996]
(i) Cl– can give up an electron more easily than F –
(ii) Cl– is a better reducing agent than F–
(iii) Cl– is smaller in size than F–
(iv)F– can be oxidized more readily than Cl–
(i) Cl– can give up an electron more easily than F –
(ii) Cl– is a better reducing agent than F–
(iii) Cl– is smaller in size than F–
(iv)F– can be oxidized more readily than Cl–
a)
(i) and (ii)
b)
(i), (ii) and (iv)
c)
(iii) and (iv)
d)
only (i)

|
Charvi Shah answered |
The halide ions act as reducing agents . F– ion does not show any reducing nature but Cl–, Br– & I– ion act as reducing agents and their reducing nature is in increasing order
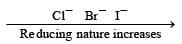
Chapter doubts & questions for p-Block Elements - 30-Day Revision Course for NEET 2026 is part of NEET exam preparation. The chapters have been prepared according to the NEET exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for NEET 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of p-Block Elements - 30-Day Revision Course for NEET in English & Hindi are available as part of NEET exam.
Download more important topics, notes, lectures and mock test series for NEET Exam by signing up for free.
;
Signup to see your scores
go up
within 7 days!
within 7 days!
Takes less than 10 seconds to signup