All Exams >
NEET >
Chemistry 31 Years NEET Chapterwise Solved Papers >
All Questions
All questions of Classification of Elements & Periodicity in Properties for NEET Exam
Which one of the following arrangements represents the correct order of least negative to most negative electron gain enthalpy for C, Ca, Al, F and O? [NEET Kar. 2013]- a)Ca < Al < C < O < F
- b)Al < Ca < O < C < F
- c)Al < O < C < Ca < F
- d)C < F < O < Al < Ca
Correct answer is option 'A'. Can you explain this answer?
Which one of the following arrangements represents the correct order of least negative to most negative electron gain enthalpy for C, Ca, Al, F and O? [NEET Kar. 2013]
a)
Ca < Al < C < O < F
b)
Al < Ca < O < C < F
c)
Al < O < C < Ca < F
d)
C < F < O < Al < Ca

|
Ritika Khanna answered |
As the nuclear charge increases, the force of attraction between the nucleus and the incoming electron increses and hence the elecron gain enthalpy becomes more negative, hence the correct order is Ca < Al < C < O < F
Identify the wrong statement in the following: [2012]- a)Amongst isoelectronic species, smaller the positive charge on the cation, smaller is the ionic radius.
- b)Amongst isoelectronic species, greater the negative charge on the anion, larger is the ionic radius.
- c)Atomic radius of the elements increases as one moves down the first group of the periodic table.
- d)Atomic radius of the elements decreases as one moves across from left to right in the 2nd period of the periodic table.
Correct answer is option 'A'. Can you explain this answer?
Identify the wrong statement in the following: [2012]
a)
Amongst isoelectronic species, smaller the positive charge on the cation, smaller is the ionic radius.
b)
Amongst isoelectronic species, greater the negative charge on the anion, larger is the ionic radius.
c)
Atomic radius of the elements increases as one moves down the first group of the periodic table.
d)
Atomic radius of the elements decreases as one moves across from left to right in the 2nd period of the periodic table.

|
Surbhi Das answered |
As the positive charge increases on metal cation, radius decreases. This is due to the fact that nuclear charge in the case of a cation is acting on lesser number of electrons and pulls them closer.
Pauling’s electronegativity values for elements are useful in predicting [1989]- a)Polarity of the molecules
- b)Position in the E.M.F. series
- c)Coordination numbers
- d)Dipole moments.
Correct answer is option 'A'. Can you explain this answer?
Pauling’s electronegativity values for elements are useful in predicting [1989]
a)
Polarity of the molecules
b)
Position in the E.M.F. series
c)
Coordination numbers
d)
Dipole moments.

|
Ruchi Chakraborty answered |
Pauling scale of electronegativity was helpful in predicting (i) Nature of bond between two atoms (ii) Stability of bond by calculating the difference in electronegativities polarity of bond can be calculated.
Which of the following pairs is correctly matched? [NEET 2023]- a)Basic oxides - In2O3, K2O, SnO2
- b)Neutral oxides - CO, NO2, N2O
- c)Acidic oxides - Mn2O7, SO2, TeO3
- d)Amphoteric oxides - BeO, Ga2O3, GeO
Correct answer is option 'C'. Can you explain this answer?
Which of the following pairs is correctly matched? [NEET 2023]
a)
Basic oxides - In2O3, K2O, SnO2
b)
Neutral oxides - CO, NO2, N2O
c)
Acidic oxides - Mn2O7, SO2, TeO3
d)
Amphoteric oxides - BeO, Ga2O3, GeO

|
Ambition Institute answered |
Basic oxides: Basic oxides are typically oxides of metals that react with acids to form salts and water. Examples include
However,
Neutral oxides: Neutral oxides are oxides that do not react significantly with either acids or bases. CO,
In2O3
, K2O
, and SnO2
.However,
SnO2
is slightly amphoteric, so this option is not entirely accurate.Neutral oxides: Neutral oxides are oxides that do not react significantly with either acids or bases. CO,
NO2
, and N2O
are all neutral oxides.- CO (Carbon monoxide) is neutral.
- NO2(Nitrogen dioxide) andN2O(Dinitrogen monoxide) are neutral oxides as well.
- So, option (b) is incorrect.
Acidic oxides: Acidic oxides are oxides that react with water to form acids and can react with bases to form salts. Examples include
Mn2O7
, SO2
, and TeO3
.- Mn2O7(Manganese heptoxide) is acidic.
- SO2(Sulfur dioxide) is acidic.
- TeO3(Tellurium trioxide) is also acidic.
Therefore, option (c) is correct.
Amphoteric oxides: Amphoteric oxides can act both as acids and bases. Examples include BeO,
Amphoteric oxides: Amphoteric oxides can act both as acids and bases. Examples include BeO,
Ga2O3
, and GeO.- BeO (Beryllium oxide) is amphoteric.
- Ga2O3(Gallium oxide) is amphoteric.
- GeO (Germanium oxide) is also amphoteric.
- This matches well with the definition of amphoteric oxides, making option (d) correct.
Conclusion: The correct answer is (c) Acidic oxides -
Mn2O7
, SO2
, TeO3
.For the second period elements the correct increasing order of first ionisation enthalpy is: [NEET 2019]- a)Li < Be < B < C < N < O < F < Ne
- b)Li < B < Be < C < O < N < F < Ne
- c)Li < B < Be < C < N < O < F < Ne
- d)Li < Be < B < C < O < N < F < Ne
Correct answer is option 'B'. Can you explain this answer?
For the second period elements the correct increasing order of first ionisation enthalpy is: [NEET 2019]
a)
Li < Be < B < C < N < O < F < Ne
b)
Li < B < Be < C < O < N < F < Ne
c)
Li < B < Be < C < N < O < F < Ne
d)
Li < Be < B < C < O < N < F < Ne

|
Shivani Tiwari answered |
Understanding Ionization Enthalpy
Ionization enthalpy refers to the energy required to remove an electron from an atom in its gaseous state. For the second period elements (Li to Ne), the trend in ionization enthalpy is influenced by various factors such as nuclear charge, electron shielding, and electron configuration.
Factors Influencing Ionization Energy
- Nuclear Charge: As we move across the period from Li to Ne, the nuclear charge increases, which generally leads to higher ionization enthalpy due to stronger attraction between the nucleus and the electrons.
- Electron Shielding: This effect is minimal across the period because all elements have their outermost electrons in the same shell, meaning they experience similar shielding.
- Electron Configuration: The stability of electron configurations plays a crucial role. For example, elements like Be (with a completely filled s-orbital) have higher ionization enthalpies compared to those with half-filled or unfilled orbitals.
Correct Order of Increasing Ionization Enthalpy
The correct increasing order of first ionization enthalpy for the second period elements is:
- Li < b="">< be="">< c="">< n="">< o="">< f=""><>="">Explanation of Order
- Li has the lowest ionization enthalpy due to its larger atomic size and single valence electron.
- B has a lower ionization enthalpy than Be because the former has a single p-electron that is easier to remove.
- Be has a higher ionization enthalpy due to its stable electron configuration (full s-orbital).
- The transition from C to N sees an increase due to greater effective nuclear charge.
- O has a slightly lower ionization enthalpy than expected due to electron-electron repulsion in its paired p-orbitals.
- F has a high ionization enthalpy due to its high electronegativity and effective nuclear charge.
- Ne has the highest ionization enthalpy with a filled shell that is very stable.
In summary, the correct increasing order is best captured by option B: Li < b="">< be="">< c="">< o="">< n="">< f="">< ne.="">
Ionization enthalpy refers to the energy required to remove an electron from an atom in its gaseous state. For the second period elements (Li to Ne), the trend in ionization enthalpy is influenced by various factors such as nuclear charge, electron shielding, and electron configuration.
Factors Influencing Ionization Energy
- Nuclear Charge: As we move across the period from Li to Ne, the nuclear charge increases, which generally leads to higher ionization enthalpy due to stronger attraction between the nucleus and the electrons.
- Electron Shielding: This effect is minimal across the period because all elements have their outermost electrons in the same shell, meaning they experience similar shielding.
- Electron Configuration: The stability of electron configurations plays a crucial role. For example, elements like Be (with a completely filled s-orbital) have higher ionization enthalpies compared to those with half-filled or unfilled orbitals.
Correct Order of Increasing Ionization Enthalpy
The correct increasing order of first ionization enthalpy for the second period elements is:
- Li < b="">< be="">< c="">< n="">< o="">< f=""><>="">Explanation of Order
- Li has the lowest ionization enthalpy due to its larger atomic size and single valence electron.
- B has a lower ionization enthalpy than Be because the former has a single p-electron that is easier to remove.
- Be has a higher ionization enthalpy due to its stable electron configuration (full s-orbital).
- The transition from C to N sees an increase due to greater effective nuclear charge.
- O has a slightly lower ionization enthalpy than expected due to electron-electron repulsion in its paired p-orbitals.
- F has a high ionization enthalpy due to its high electronegativity and effective nuclear charge.
- Ne has the highest ionization enthalpy with a filled shell that is very stable.
In summary, the correct increasing order is best captured by option B: Li < b="">< be="">< c="">< o="">< n="">< f="">< ne.="">
Arrange the following elements in increasing order of electronegativity: [NEET 2024]
N, O, F, C, Si
Choose the correct answer from the options given below :- a)Si < C < N < O < F
- b)Si < C < O < N < F
- c)O < F < N < C < Si
- d)F < O < N < C < Si
Correct answer is option 'A'. Can you explain this answer?
Arrange the following elements in increasing order of electronegativity: [NEET 2024]
N, O, F, C, Si
Choose the correct answer from the options given below :
N, O, F, C, Si
Choose the correct answer from the options given below :
a)
Si < C < N < O < F
b)
Si < C < O < N < F
c)
O < F < N < C < Si
d)
F < O < N < C < Si
|
|
Janhavi Patel answered |
Understanding Electronegativity
Electronegativity is the tendency of an atom to attract electrons in a chemical bond. The values of electronegativity increase across a period and decrease down a group in the periodic table.
Electronegativity Values
- Fluorine (F): 4.0 (highest)
- Oxygen (O): 3.5
- Nitrogen (N): 3.0
- Carbon (C): 2.5
- Silicon (Si): 1.8 (lowest)
Arranging the Elements
To arrange the elements N, O, F, C, and Si in increasing order of electronegativity, we need to look at their respective values:
- Silicon (Si): 1.8
- Carbon (C): 2.5
- Nitrogen (N): 3.0
- Oxygen (O): 3.5
- Fluorine (F): 4.0
Increasing Order of Electronegativity
Based on the values mentioned, the increasing order of electronegativity is:
- Si < c="">< n="">< o="">< f="" this="" is="" reflected="" in="" option="">a)="" si="">< c="">< n="">< o="">< f,="" which="" is="" the="" correct="" answer.="">Conclusion
Understanding the trend of electronegativity helps in predicting the nature of bonds formed by these elements. Fluorine, being the most electronegative element, has a strong tendency to attract electrons, while silicon, being the least electronegative among the given elements, has a weaker attraction for electrons.
Electronegativity is the tendency of an atom to attract electrons in a chemical bond. The values of electronegativity increase across a period and decrease down a group in the periodic table.
Electronegativity Values
- Fluorine (F): 4.0 (highest)
- Oxygen (O): 3.5
- Nitrogen (N): 3.0
- Carbon (C): 2.5
- Silicon (Si): 1.8 (lowest)
Arranging the Elements
To arrange the elements N, O, F, C, and Si in increasing order of electronegativity, we need to look at their respective values:
- Silicon (Si): 1.8
- Carbon (C): 2.5
- Nitrogen (N): 3.0
- Oxygen (O): 3.5
- Fluorine (F): 4.0
Increasing Order of Electronegativity
Based on the values mentioned, the increasing order of electronegativity is:
- Si < c="">< n="">< o="">< f="" this="" is="" reflected="" in="" option="">a)="" si="">< c="">< n="">< o="">< f,="" which="" is="" the="" correct="" answer.="">Conclusion
Understanding the trend of electronegativity helps in predicting the nature of bonds formed by these elements. Fluorine, being the most electronegative element, has a strong tendency to attract electrons, while silicon, being the least electronegative among the given elements, has a weaker attraction for electrons.
Which of the following orders of ionic radii is correctly represented? [NEET 2014]- a)F- > O2- > Na+
- b)Al3+ > Mg2+ > N3-
- c)H- > H. > H
- d)Na+ > F > O2-
Correct answer is option 'C'. Can you explain this answer?
Which of the following orders of ionic radii is correctly represented? [NEET 2014]
a)
F- > O2- > Na+
b)
Al3+ > Mg2+ > N3-
c)
H- > H. > H
d)
Na+ > F > O2-

|
Infinity Academy answered |
Cation loose electrons are smaller in size than the parent atom, where anions gain electrons are larger in size than the parent atom.
Hence the order is H- > H. > H+
Hence the order is H- > H. > H+
Which of the following represents the correct order of increasing electron gain enthalpy with negative sign for the elements O, S, F and Cl ? [2010]- a)CI < F < O < S
- b)O < S < F < CI
- c)F < S < O < CI
- d)S < O < CI < F
Correct answer is option 'B'. Can you explain this answer?
Which of the following represents the correct order of increasing electron gain enthalpy with negative sign for the elements O, S, F and Cl ? [2010]
a)
CI < F < O < S
b)
O < S < F < CI
c)
F < S < O < CI
d)
S < O < CI < F

|
Prasenjit Pillai answered |
O < S < F < CI
Electron gain enthalpy – 141 – 200 – 333 – 349 kJ mol–1
What is the value of electron gain enthalpy of Na+ if IE1 of Na = 5.1 eV ? [2011M]- a)–5.1 eV
- b)–10.2 eV
- c)+2.55 eV
- d)+10.2 eV
Correct answer is option 'A'. Can you explain this answer?
What is the value of electron gain enthalpy of Na+ if IE1 of Na = 5.1 eV ? [2011M]
a)
–5.1 eV
b)
–10.2 eV
c)
+2.55 eV
d)
+10.2 eV

|
Pankaj Banerjee answered |
IE1 of Na = – Electron gain enthalpy of Na+ = – 5.1 Volt.
Which among the following electronic configurations belong to main group elements? [NEET 2025]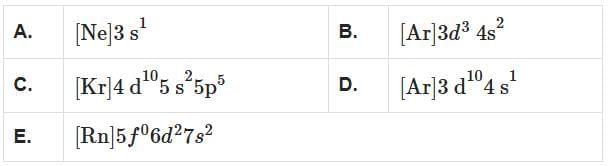 Choose the correct answer from the option given below:
Choose the correct answer from the option given below:- a)D and E only.
- b)A, C and D only.
- c)B and E only.
- d)A and C only.
Correct answer is option 'D'. Can you explain this answer?
Which among the following electronic configurations belong to main group elements? [NEET 2025]

Choose the correct answer from the option given below:
a)
D and E only.
b)
A, C and D only.
c)
B and E only.
d)
A and C only.

|
Stepway Academy answered |
Analyze the given electronic configurations to determine whether they belong to main group elements:
- A. [Ne]3s1: This corresponds to sodium (Na), which is in Group 1. It is a main group element.
- B. [Ar]3d34s2: This corresponds to vanadium (V), which is a transition metal (Group 5). It is not a main group element.
- C. [Kr]4d105s25p5: This corresponds to iodine (I), which is in Group 17. It is a main group element.
- D. [Ar]3d104s1: This corresponds to copper (Cu), which is a transition metal (Group 11). It is not a main group element.
- E. [Rn]5f06d27s2: This corresponds to thorium (Th), which is an actinide and not a main group element.
So, only A and C belong to main group elements.
Chapter doubts & questions for Classification of Elements & Periodicity in Properties - Chemistry 31 Years NEET Chapterwise Solved Papers 2026 is part of NEET exam preparation. The chapters have been prepared according to the NEET exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for NEET 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Classification of Elements & Periodicity in Properties - Chemistry 31 Years NEET Chapterwise Solved Papers in English & Hindi are available as part of NEET exam.
Download more important topics, notes, lectures and mock test series for NEET Exam by signing up for free.
;
Signup to see your scores
go up within 7 days!
Access 1000+ FREE Docs, Videos and Tests
Takes less than 10 seconds to signup