Class 12 Exam > Class 12 Questions > A chemist isolated a compound A with molecula...
Start Learning for Free
A chemist isolated a compound A with molecular formula C7H13Br. A undergoes very fast SN1 reaction. Spectroscopic evidence indicated that compound A has the following structural characteristics : It contains five sp3-hybridised carbon atoms. Among those five sp3 carbon atoms, three are methyl groups, one CH2 group and one CH group.
It also contains two sp2-hybridised carbon atoms. Also there is only one hydrogen atom attached to sp2 carbons.
The compound contains a total of six aliylic hydrogen atoms.
The carbon atom that holds the Br has one H attached to it.
Working Space
When compound A reacts with boiling water, it undergoes a SN1 reaction and produces two principal products B and C. Both B and C are alcohols with their molecular formula C7H14O. Among the two alcohols, B has the —OH group attached to a sp3 carbon atom that has no H -atoms bonded to it.
It also contains two sp2-hybridised carbon atoms. Also there is only one hydrogen atom attached to sp2 carbons.
The compound contains a total of six aliylic hydrogen atoms.
The carbon atom that holds the Br has one H attached to it.
Working Space
When compound A reacts with boiling water, it undergoes a SN1 reaction and produces two principal products B and C. Both B and C are alcohols with their molecular formula C7H14O. Among the two alcohols, B has the —OH group attached to a sp3 carbon atom that has no H -atoms bonded to it.
Q.
What can be said about the isomerism shown by the two alcohols B and C ?
- a)Both B and C show stereoisomerism and can be resolved into enantiomers
- b)B and C are stereoisomers
- c)Both B and C show stereoisomerism but only C can be resolved into enantiomers
- d)Neither B nor C can be resolved into enantiomers
Correct answer is option 'A'. Can you explain this answer?
| FREE This question is part of | Download PDF Attempt this Test |
Verified Answer
A chemist isolated a compound A with molecular formula C7H13Br. A unde...
Alcohols B and C are:
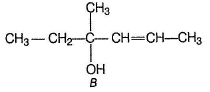
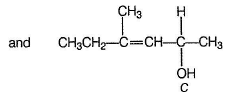
Both S and C have one chiral carbon each, both show optical isomerism.
Both S and C have one chiral carbon each, both show optical isomerism.
Most Upvoted Answer
A chemist isolated a compound A with molecular formula C7H13Br. A unde...
Explanation:
Isomerism in alcohols B and C:
- Both alcohols B and C have the same molecular formula C7H14O.
- Alcohol B has the OH group attached to a sp3 carbon atom with no H-atoms bonded to it.
- Alcohol C is the other product formed from the reaction.
- Since alcohol B has a chiral center (sp3 carbon with four different groups attached), it can exist as a pair of enantiomers.
Conclusion:
- Therefore, both alcohols B and C show stereoisomerism, with alcohol B being able to be resolved into enantiomers.
Isomerism in alcohols B and C:
- Both alcohols B and C have the same molecular formula C7H14O.
- Alcohol B has the OH group attached to a sp3 carbon atom with no H-atoms bonded to it.
- Alcohol C is the other product formed from the reaction.
- Since alcohol B has a chiral center (sp3 carbon with four different groups attached), it can exist as a pair of enantiomers.
Conclusion:
- Therefore, both alcohols B and C show stereoisomerism, with alcohol B being able to be resolved into enantiomers.

|
Explore Courses for Class 12 exam
|

|
Similar Class 12 Doubts
A chemist isolated a compound A with molecular formula C7H13Br. A undergoes very fast SN1 reaction. Spectroscopic evidence indicated that compound A has the following structural characteristics : It contains five sp3-hybridised carbon atoms. Among those five sp3 carbon atoms, three are methyl groups, one CH2 group and one CH group.It also contains two sp2-hybridised carbon atoms. Also there is only one hydrogen atom attached to sp2 carbons.The compound contains a total of six aliylic hydrogen atoms.The carbon atom that holds the Br has one H attached to it.Working SpaceWhen compound A reacts with boiling water, it undergoes a SN1 reaction and produces two principal products B and C. Both B and C are alcohols with their molecular formula C7H14O. Among the two alcohols, B has the —OH group attached to a sp3 carbon atom that has no H -atoms bonded to it.Q.What can be said about the isomerism shown by the two alcohols B and C ?a)Both B and C show stereoisomerism and can be resolved into enantiomersb)B and C are stereoisomersc)Both B and C show stereoisomerism but only C can be resolved into enantiomersd)Neither B nor C can be resolved into enantiomersCorrect answer is option 'A'. Can you explain this answer?
Question Description
A chemist isolated a compound A with molecular formula C7H13Br. A undergoes very fast SN1 reaction. Spectroscopic evidence indicated that compound A has the following structural characteristics : It contains five sp3-hybridised carbon atoms. Among those five sp3 carbon atoms, three are methyl groups, one CH2 group and one CH group.It also contains two sp2-hybridised carbon atoms. Also there is only one hydrogen atom attached to sp2 carbons.The compound contains a total of six aliylic hydrogen atoms.The carbon atom that holds the Br has one H attached to it.Working SpaceWhen compound A reacts with boiling water, it undergoes a SN1 reaction and produces two principal products B and C. Both B and C are alcohols with their molecular formula C7H14O. Among the two alcohols, B has the —OH group attached to a sp3 carbon atom that has no H -atoms bonded to it.Q.What can be said about the isomerism shown by the two alcohols B and C ?a)Both B and C show stereoisomerism and can be resolved into enantiomersb)B and C are stereoisomersc)Both B and C show stereoisomerism but only C can be resolved into enantiomersd)Neither B nor C can be resolved into enantiomersCorrect answer is option 'A'. Can you explain this answer? for Class 12 2024 is part of Class 12 preparation. The Question and answers have been prepared according to the Class 12 exam syllabus. Information about A chemist isolated a compound A with molecular formula C7H13Br. A undergoes very fast SN1 reaction. Spectroscopic evidence indicated that compound A has the following structural characteristics : It contains five sp3-hybridised carbon atoms. Among those five sp3 carbon atoms, three are methyl groups, one CH2 group and one CH group.It also contains two sp2-hybridised carbon atoms. Also there is only one hydrogen atom attached to sp2 carbons.The compound contains a total of six aliylic hydrogen atoms.The carbon atom that holds the Br has one H attached to it.Working SpaceWhen compound A reacts with boiling water, it undergoes a SN1 reaction and produces two principal products B and C. Both B and C are alcohols with their molecular formula C7H14O. Among the two alcohols, B has the —OH group attached to a sp3 carbon atom that has no H -atoms bonded to it.Q.What can be said about the isomerism shown by the two alcohols B and C ?a)Both B and C show stereoisomerism and can be resolved into enantiomersb)B and C are stereoisomersc)Both B and C show stereoisomerism but only C can be resolved into enantiomersd)Neither B nor C can be resolved into enantiomersCorrect answer is option 'A'. Can you explain this answer? covers all topics & solutions for Class 12 2024 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for A chemist isolated a compound A with molecular formula C7H13Br. A undergoes very fast SN1 reaction. Spectroscopic evidence indicated that compound A has the following structural characteristics : It contains five sp3-hybridised carbon atoms. Among those five sp3 carbon atoms, three are methyl groups, one CH2 group and one CH group.It also contains two sp2-hybridised carbon atoms. Also there is only one hydrogen atom attached to sp2 carbons.The compound contains a total of six aliylic hydrogen atoms.The carbon atom that holds the Br has one H attached to it.Working SpaceWhen compound A reacts with boiling water, it undergoes a SN1 reaction and produces two principal products B and C. Both B and C are alcohols with their molecular formula C7H14O. Among the two alcohols, B has the —OH group attached to a sp3 carbon atom that has no H -atoms bonded to it.Q.What can be said about the isomerism shown by the two alcohols B and C ?a)Both B and C show stereoisomerism and can be resolved into enantiomersb)B and C are stereoisomersc)Both B and C show stereoisomerism but only C can be resolved into enantiomersd)Neither B nor C can be resolved into enantiomersCorrect answer is option 'A'. Can you explain this answer?.
A chemist isolated a compound A with molecular formula C7H13Br. A undergoes very fast SN1 reaction. Spectroscopic evidence indicated that compound A has the following structural characteristics : It contains five sp3-hybridised carbon atoms. Among those five sp3 carbon atoms, three are methyl groups, one CH2 group and one CH group.It also contains two sp2-hybridised carbon atoms. Also there is only one hydrogen atom attached to sp2 carbons.The compound contains a total of six aliylic hydrogen atoms.The carbon atom that holds the Br has one H attached to it.Working SpaceWhen compound A reacts with boiling water, it undergoes a SN1 reaction and produces two principal products B and C. Both B and C are alcohols with their molecular formula C7H14O. Among the two alcohols, B has the —OH group attached to a sp3 carbon atom that has no H -atoms bonded to it.Q.What can be said about the isomerism shown by the two alcohols B and C ?a)Both B and C show stereoisomerism and can be resolved into enantiomersb)B and C are stereoisomersc)Both B and C show stereoisomerism but only C can be resolved into enantiomersd)Neither B nor C can be resolved into enantiomersCorrect answer is option 'A'. Can you explain this answer? for Class 12 2024 is part of Class 12 preparation. The Question and answers have been prepared according to the Class 12 exam syllabus. Information about A chemist isolated a compound A with molecular formula C7H13Br. A undergoes very fast SN1 reaction. Spectroscopic evidence indicated that compound A has the following structural characteristics : It contains five sp3-hybridised carbon atoms. Among those five sp3 carbon atoms, three are methyl groups, one CH2 group and one CH group.It also contains two sp2-hybridised carbon atoms. Also there is only one hydrogen atom attached to sp2 carbons.The compound contains a total of six aliylic hydrogen atoms.The carbon atom that holds the Br has one H attached to it.Working SpaceWhen compound A reacts with boiling water, it undergoes a SN1 reaction and produces two principal products B and C. Both B and C are alcohols with their molecular formula C7H14O. Among the two alcohols, B has the —OH group attached to a sp3 carbon atom that has no H -atoms bonded to it.Q.What can be said about the isomerism shown by the two alcohols B and C ?a)Both B and C show stereoisomerism and can be resolved into enantiomersb)B and C are stereoisomersc)Both B and C show stereoisomerism but only C can be resolved into enantiomersd)Neither B nor C can be resolved into enantiomersCorrect answer is option 'A'. Can you explain this answer? covers all topics & solutions for Class 12 2024 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for A chemist isolated a compound A with molecular formula C7H13Br. A undergoes very fast SN1 reaction. Spectroscopic evidence indicated that compound A has the following structural characteristics : It contains five sp3-hybridised carbon atoms. Among those five sp3 carbon atoms, three are methyl groups, one CH2 group and one CH group.It also contains two sp2-hybridised carbon atoms. Also there is only one hydrogen atom attached to sp2 carbons.The compound contains a total of six aliylic hydrogen atoms.The carbon atom that holds the Br has one H attached to it.Working SpaceWhen compound A reacts with boiling water, it undergoes a SN1 reaction and produces two principal products B and C. Both B and C are alcohols with their molecular formula C7H14O. Among the two alcohols, B has the —OH group attached to a sp3 carbon atom that has no H -atoms bonded to it.Q.What can be said about the isomerism shown by the two alcohols B and C ?a)Both B and C show stereoisomerism and can be resolved into enantiomersb)B and C are stereoisomersc)Both B and C show stereoisomerism but only C can be resolved into enantiomersd)Neither B nor C can be resolved into enantiomersCorrect answer is option 'A'. Can you explain this answer?.
Solutions for A chemist isolated a compound A with molecular formula C7H13Br. A undergoes very fast SN1 reaction. Spectroscopic evidence indicated that compound A has the following structural characteristics : It contains five sp3-hybridised carbon atoms. Among those five sp3 carbon atoms, three are methyl groups, one CH2 group and one CH group.It also contains two sp2-hybridised carbon atoms. Also there is only one hydrogen atom attached to sp2 carbons.The compound contains a total of six aliylic hydrogen atoms.The carbon atom that holds the Br has one H attached to it.Working SpaceWhen compound A reacts with boiling water, it undergoes a SN1 reaction and produces two principal products B and C. Both B and C are alcohols with their molecular formula C7H14O. Among the two alcohols, B has the —OH group attached to a sp3 carbon atom that has no H -atoms bonded to it.Q.What can be said about the isomerism shown by the two alcohols B and C ?a)Both B and C show stereoisomerism and can be resolved into enantiomersb)B and C are stereoisomersc)Both B and C show stereoisomerism but only C can be resolved into enantiomersd)Neither B nor C can be resolved into enantiomersCorrect answer is option 'A'. Can you explain this answer? in English & in Hindi are available as part of our courses for Class 12.
Download more important topics, notes, lectures and mock test series for Class 12 Exam by signing up for free.
Here you can find the meaning of A chemist isolated a compound A with molecular formula C7H13Br. A undergoes very fast SN1 reaction. Spectroscopic evidence indicated that compound A has the following structural characteristics : It contains five sp3-hybridised carbon atoms. Among those five sp3 carbon atoms, three are methyl groups, one CH2 group and one CH group.It also contains two sp2-hybridised carbon atoms. Also there is only one hydrogen atom attached to sp2 carbons.The compound contains a total of six aliylic hydrogen atoms.The carbon atom that holds the Br has one H attached to it.Working SpaceWhen compound A reacts with boiling water, it undergoes a SN1 reaction and produces two principal products B and C. Both B and C are alcohols with their molecular formula C7H14O. Among the two alcohols, B has the —OH group attached to a sp3 carbon atom that has no H -atoms bonded to it.Q.What can be said about the isomerism shown by the two alcohols B and C ?a)Both B and C show stereoisomerism and can be resolved into enantiomersb)B and C are stereoisomersc)Both B and C show stereoisomerism but only C can be resolved into enantiomersd)Neither B nor C can be resolved into enantiomersCorrect answer is option 'A'. Can you explain this answer? defined & explained in the simplest way possible. Besides giving the explanation of
A chemist isolated a compound A with molecular formula C7H13Br. A undergoes very fast SN1 reaction. Spectroscopic evidence indicated that compound A has the following structural characteristics : It contains five sp3-hybridised carbon atoms. Among those five sp3 carbon atoms, three are methyl groups, one CH2 group and one CH group.It also contains two sp2-hybridised carbon atoms. Also there is only one hydrogen atom attached to sp2 carbons.The compound contains a total of six aliylic hydrogen atoms.The carbon atom that holds the Br has one H attached to it.Working SpaceWhen compound A reacts with boiling water, it undergoes a SN1 reaction and produces two principal products B and C. Both B and C are alcohols with their molecular formula C7H14O. Among the two alcohols, B has the —OH group attached to a sp3 carbon atom that has no H -atoms bonded to it.Q.What can be said about the isomerism shown by the two alcohols B and C ?a)Both B and C show stereoisomerism and can be resolved into enantiomersb)B and C are stereoisomersc)Both B and C show stereoisomerism but only C can be resolved into enantiomersd)Neither B nor C can be resolved into enantiomersCorrect answer is option 'A'. Can you explain this answer?, a detailed solution for A chemist isolated a compound A with molecular formula C7H13Br. A undergoes very fast SN1 reaction. Spectroscopic evidence indicated that compound A has the following structural characteristics : It contains five sp3-hybridised carbon atoms. Among those five sp3 carbon atoms, three are methyl groups, one CH2 group and one CH group.It also contains two sp2-hybridised carbon atoms. Also there is only one hydrogen atom attached to sp2 carbons.The compound contains a total of six aliylic hydrogen atoms.The carbon atom that holds the Br has one H attached to it.Working SpaceWhen compound A reacts with boiling water, it undergoes a SN1 reaction and produces two principal products B and C. Both B and C are alcohols with their molecular formula C7H14O. Among the two alcohols, B has the —OH group attached to a sp3 carbon atom that has no H -atoms bonded to it.Q.What can be said about the isomerism shown by the two alcohols B and C ?a)Both B and C show stereoisomerism and can be resolved into enantiomersb)B and C are stereoisomersc)Both B and C show stereoisomerism but only C can be resolved into enantiomersd)Neither B nor C can be resolved into enantiomersCorrect answer is option 'A'. Can you explain this answer? has been provided alongside types of A chemist isolated a compound A with molecular formula C7H13Br. A undergoes very fast SN1 reaction. Spectroscopic evidence indicated that compound A has the following structural characteristics : It contains five sp3-hybridised carbon atoms. Among those five sp3 carbon atoms, three are methyl groups, one CH2 group and one CH group.It also contains two sp2-hybridised carbon atoms. Also there is only one hydrogen atom attached to sp2 carbons.The compound contains a total of six aliylic hydrogen atoms.The carbon atom that holds the Br has one H attached to it.Working SpaceWhen compound A reacts with boiling water, it undergoes a SN1 reaction and produces two principal products B and C. Both B and C are alcohols with their molecular formula C7H14O. Among the two alcohols, B has the —OH group attached to a sp3 carbon atom that has no H -atoms bonded to it.Q.What can be said about the isomerism shown by the two alcohols B and C ?a)Both B and C show stereoisomerism and can be resolved into enantiomersb)B and C are stereoisomersc)Both B and C show stereoisomerism but only C can be resolved into enantiomersd)Neither B nor C can be resolved into enantiomersCorrect answer is option 'A'. Can you explain this answer? theory, EduRev gives you an
ample number of questions to practice A chemist isolated a compound A with molecular formula C7H13Br. A undergoes very fast SN1 reaction. Spectroscopic evidence indicated that compound A has the following structural characteristics : It contains five sp3-hybridised carbon atoms. Among those five sp3 carbon atoms, three are methyl groups, one CH2 group and one CH group.It also contains two sp2-hybridised carbon atoms. Also there is only one hydrogen atom attached to sp2 carbons.The compound contains a total of six aliylic hydrogen atoms.The carbon atom that holds the Br has one H attached to it.Working SpaceWhen compound A reacts with boiling water, it undergoes a SN1 reaction and produces two principal products B and C. Both B and C are alcohols with their molecular formula C7H14O. Among the two alcohols, B has the —OH group attached to a sp3 carbon atom that has no H -atoms bonded to it.Q.What can be said about the isomerism shown by the two alcohols B and C ?a)Both B and C show stereoisomerism and can be resolved into enantiomersb)B and C are stereoisomersc)Both B and C show stereoisomerism but only C can be resolved into enantiomersd)Neither B nor C can be resolved into enantiomersCorrect answer is option 'A'. Can you explain this answer? tests, examples and also practice Class 12 tests.

|
Explore Courses for Class 12 exam
|

|
Signup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.


















