Class 12 Exam > Class 12 Questions > Benzene diazonium chloride forms orange red d... Start Learning for Free
Benzene diazonium chloride forms orange red dye with:
- a)Nitrophenol
- b)Benzophenol
- c)Resorcinol
- d)Methanol
Correct answer is option 'C'. Can you explain this answer?
Most Upvoted Answer
Benzene diazonium chloride forms orange red dye with:a)Nitrophenolb)Be...
Free Test
FREE
| Start Free Test |
Community Answer
Benzene diazonium chloride forms orange red dye with:a)Nitrophenolb)Be...
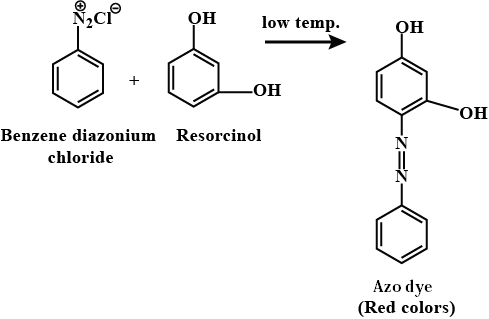
Benzene diazonium chloride and Resorcinol Reaction
The reaction between benzene diazonium chloride and resorcinol yields an orange-red dye. This is due to the formation of an azo compound, which is a compound containing a nitrogen-nitrogen double bond (-N=N-) linked to two aromatic rings.
Mechanism of Reaction
1. Diazotization: Benzene diazonium chloride reacts with hydrochloric acid to form diazonium salt.
C6H5N2Cl + HCl → C6H5N2Cl2
2. Coupling: The diazonium salt then reacts with resorcinol in an acidic medium to form an azo compound.
C6H5N2Cl2 + C6H4(OH)2 → C12H8N2O2 + 2HCl
Where C12H8N2O2 is the orange-red dye formed.
Explanation
Resorcinol contains two hydroxyl (-OH) groups that can react with benzene diazonium chloride in the presence of an acid to form an azo compound. This reaction is known as coupling. The nitrogen-nitrogen double bond (-N=N-) in the azo compound is responsible for its color. The presence of the electron-donating hydroxyl groups on the aromatic ring of resorcinol increases the electron density on the ring, making it more susceptible to attack by the diazonium ion.
Conclusion
The reaction between benzene diazonium chloride and resorcinol yields an orange-red dye due to the formation of an azo compound. This reaction is widely used in the textile industry for dyeing fabrics.
The reaction between benzene diazonium chloride and resorcinol yields an orange-red dye. This is due to the formation of an azo compound, which is a compound containing a nitrogen-nitrogen double bond (-N=N-) linked to two aromatic rings.
Mechanism of Reaction
1. Diazotization: Benzene diazonium chloride reacts with hydrochloric acid to form diazonium salt.
C6H5N2Cl + HCl → C6H5N2Cl2
2. Coupling: The diazonium salt then reacts with resorcinol in an acidic medium to form an azo compound.
C6H5N2Cl2 + C6H4(OH)2 → C12H8N2O2 + 2HCl
Where C12H8N2O2 is the orange-red dye formed.
Explanation
Resorcinol contains two hydroxyl (-OH) groups that can react with benzene diazonium chloride in the presence of an acid to form an azo compound. This reaction is known as coupling. The nitrogen-nitrogen double bond (-N=N-) in the azo compound is responsible for its color. The presence of the electron-donating hydroxyl groups on the aromatic ring of resorcinol increases the electron density on the ring, making it more susceptible to attack by the diazonium ion.
Conclusion
The reaction between benzene diazonium chloride and resorcinol yields an orange-red dye due to the formation of an azo compound. This reaction is widely used in the textile industry for dyeing fabrics.
 | Explore Courses for Class 12 exam |  |
Question Description
Benzene diazonium chloride forms orange red dye with:a)Nitrophenolb)Benzophenolc)Resorcinold)MethanolCorrect answer is option 'C'. Can you explain this answer? for Class 12 2026 is part of Class 12 preparation. The Question and answers have been prepared according to the Class 12 exam syllabus. Information about Benzene diazonium chloride forms orange red dye with:a)Nitrophenolb)Benzophenolc)Resorcinold)MethanolCorrect answer is option 'C'. Can you explain this answer? covers all topics & solutions for Class 12 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for Benzene diazonium chloride forms orange red dye with:a)Nitrophenolb)Benzophenolc)Resorcinold)MethanolCorrect answer is option 'C'. Can you explain this answer?.
Benzene diazonium chloride forms orange red dye with:a)Nitrophenolb)Benzophenolc)Resorcinold)MethanolCorrect answer is option 'C'. Can you explain this answer? for Class 12 2026 is part of Class 12 preparation. The Question and answers have been prepared according to the Class 12 exam syllabus. Information about Benzene diazonium chloride forms orange red dye with:a)Nitrophenolb)Benzophenolc)Resorcinold)MethanolCorrect answer is option 'C'. Can you explain this answer? covers all topics & solutions for Class 12 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for Benzene diazonium chloride forms orange red dye with:a)Nitrophenolb)Benzophenolc)Resorcinold)MethanolCorrect answer is option 'C'. Can you explain this answer?.
Solutions for Benzene diazonium chloride forms orange red dye with:a)Nitrophenolb)Benzophenolc)Resorcinold)MethanolCorrect answer is option 'C'. Can you explain this answer? in English & in Hindi are available as part of our courses for Class 12. Download more important topics, notes, lectures and mock test series for Class 12 Exam by signing up for free.
Here you can find the meaning of Benzene diazonium chloride forms orange red dye with:a)Nitrophenolb)Benzophenolc)Resorcinold)MethanolCorrect answer is option 'C'. Can you explain this answer? defined & explained in the simplest way possible. Besides giving the explanation of Benzene diazonium chloride forms orange red dye with:a)Nitrophenolb)Benzophenolc)Resorcinold)MethanolCorrect answer is option 'C'. Can you explain this answer?, a detailed solution for Benzene diazonium chloride forms orange red dye with:a)Nitrophenolb)Benzophenolc)Resorcinold)MethanolCorrect answer is option 'C'. Can you explain this answer? has been provided alongside types of Benzene diazonium chloride forms orange red dye with:a)Nitrophenolb)Benzophenolc)Resorcinold)MethanolCorrect answer is option 'C'. Can you explain this answer? theory, EduRev gives you an ample number of questions to practice Benzene diazonium chloride forms orange red dye with:a)Nitrophenolb)Benzophenolc)Resorcinold)MethanolCorrect answer is option 'C'. Can you explain this answer? tests, examples and also practice Class 12 tests.
 | Explore Courses for Class 12 exam |  |
Top Courses for Class 12
Explore CoursesSignup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.







