2nd & 3d Law: Entropy Change & Spontaneity
Second Law of Thermodynamics
The second law of thermodynamics places restrictions on the direction of heat transfer and on the maximum possible efficiency of devices that convert heat to work. While the first law of thermodynamics (conservation of energy) tells us that energy cannot be created or destroyed, it does not tell us which processes can occur spontaneously or the quality of energy transferred. The second law supplies that criterion: a process is physically possible only if it satisfies both the first and second laws.
Statement (qualitative)
The second law can be expressed in several equivalent ways. A concise formulation useful for many purposes is:
For any spontaneous process, the entropy of the universe increases:
ΔSuniv > 0
Entropy - short reminder
Entropy (S) is a state function that measures the degree of disorder or the number of accessible microstates of a system. In an isolated system the tendency is toward greater disorder; the second law quantifies this as an increase of entropy.
Different classical statements of the second law
- Kelvin-Planck statement
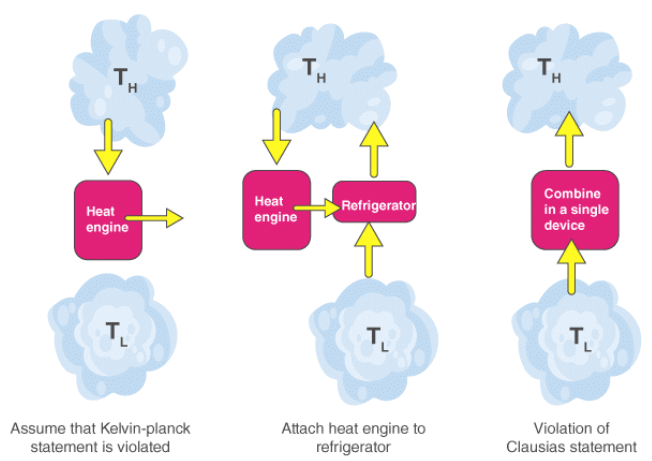
It is impossible to construct a heat engine that, operating in a complete cycle, produces net work while exchanging heat with a single heat reservoir only. In other words, no cyclic device can convert all heat absorbed from a single reservoir completely into work. - Clausius statement
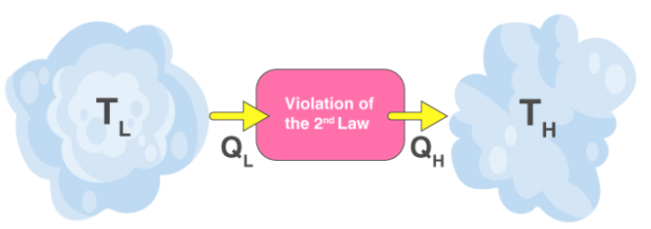
It is impossible to construct a cyclic device whose sole effect is to transfer heat from a colder body to a hotter body without external work being supplied (i.e. heat cannot spontaneously flow from cold to hot).
These two statements are equivalent: a violation of one leads to a violation of the other.
Carnot and the origin of the idea of limits
Sadi Carnot (often called the father of thermodynamics) analysed ideal heat engines and showed that the maximum possible efficiency of a heat engine depends only on the temperatures of the hot and cold reservoirs. His work anticipated the second law, though he used the caloric concept available in his time.
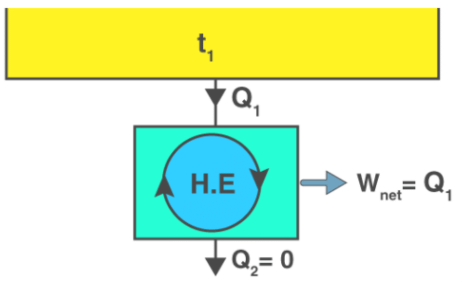
Perpetual motion machine of the second kind (PMM2)
A device that produces work while interacting with a single heat reservoir (thus converting heat completely into work in a cyclic process) would violate the second law. Such a hypothetical device is called a perpetual motion machine of the second kind (PMM2). Because the Kelvin-Planck statement forbids complete conversion of heat from a single reservoir into work, a PMM2 is impossible.
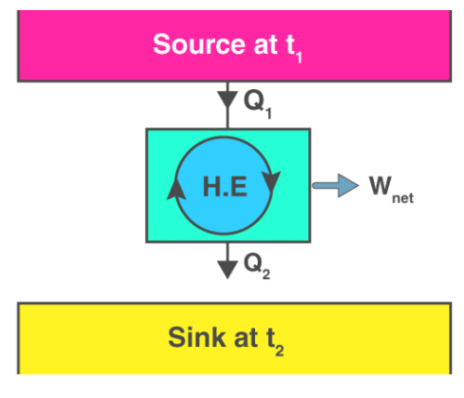
In practice a heat engine must exchange heat with at least two reservoirs at different temperatures to produce work in a cycle. If both reservoirs have finite heat capacities, the engine can produce work until the temperatures of the two reservoirs become equal.
Mathematical form of the second law
For any process occurring in the universe,
ΔSuniv = ΔSsystem + ΔSsurroundings ≥ 0
Equality holds for a reversible process (ΔSuniv = 0); strict inequality holds for irreversible (spontaneous) processes (ΔSuniv > 0).
Examples and simple calculations
Example 1: A heat pump uses 300 J of work to remove 400 J of heat from the low-temperature reservoir. How much heat is delivered to the higher temperature reservoir?
W = 300 J
QC = 400 J
QH = W + QC
QH = 300 J + 400 J
QH = 700 J
Answer: Heat delivered to the higher temperature reservoir is 700 J.
Example 2: A reversible heat engine receives 4000 kJ of heat from a reservoir at 600 K. The surroundings are at 300 K. Determine (a) the availability of heat energy and (b) unavailable heat.
Given:
Q1 = 4000 kJ
T1 = 600 K
T0 = 300 K
Change in entropy when heat Q1 is absorbed reversibly at T1 is
ΔS = Q1 / T1 = 4000 kJ / 600 K = 6.666... kJ K-1
The availability (also called exergy or useful energy at T0) is
A = Q1 - T0 ΔS
A = 4000 - 300 × 6.666... = 4000 - 2000 = 2000 kJ
Unavailable heat (U.A) = T0 ΔS = 300 × 6.666... = 2000 kJ
(a) Availability A = 2000 kJ
(b) Unavailable heat = 2000 kJ
Entropy
Entropy is a central concept in thermodynamics and statistical mechanics. It quantifies the degree of microscopic disorder or the number of accessible microstates consistent with the macroscopic state.
Definitions
- Thermodynamic definition: For a reversible, isothermal heat transfer, the change in entropy is the heat absorbed reversibly divided by the absolute temperature: ΔS = qrev / T.
- Statistical definition (Boltzmann): S = kB ln Ω, where kB is the Boltzmann constant and Ω is the number of accessible microstates.
Properties of entropy
- Entropy is a state function; it depends only on the state of the system, not the path by which the state was reached.
- Entropy is an extensive property: it scales with the size (amount) of the system.
- Standard symbol: S. Standard (molar) entropy often written as S°. SI unit: J K-1 mol-1.
- Typical order of entropy: gas > liquid > solid.
Note: A system at higher temperature usually has greater molecular disorder than the same system at a lower temperature. When a substance changes from solid → liquid → gas entropy generally increases. If a reaction produces more moles of gas, entropy usually increases.
Entropy change - general expressions
For a reversible process at temperature T,
ΔS = qrev,iso / T
For a process involving heat transfer between bodies at different temperatures, the total entropy change is
ΔStotal = ΔSsystem + ΔSsurroundings
If the system loses heat q at temperature T1 which is received by surroundings at temperature T2,
ΔSsystem = -q / T1
ΔSsurroundings = +q / T2
Therefore, ΔStotal = -q / T1 + q / T2
- If ΔStotal > 0 the process is spontaneous.
- If ΔStotal < 0 the process is non-spontaneous.
- If ΔStotal = 0 the process is reversible / at equilibrium.
Entropy change for ideal gases (isothermal reversible)
For an ideal gas undergoing an isothermal, reversible expansion from V1 to V2:
ΔS = qrev / T
Using qrev = nRT ln(V2/V1) we obtain
ΔS = nR ln(V2/V1)
Entropy in adiabatic processes
For a reversible adiabatic process, q = 0, therefore ΔS = 0. Such a process is called isentropic. An irreversible adiabatic process is not isentropic and will have ΔS > 0.
Third Law of Thermodynamics
The third law of thermodynamics states that the entropy of a perfect crystal tends to zero as the temperature approaches absolute zero (0 K).
In statistical terms, a perfect crystal at 0 K has a single microstate (Ω = 1). Using Boltzmann's relation:
S = kB ln Ω
For Ω = 1, ln Ω = 0, hence S = 0.
Historical note: The third law (often called Nernst's theorem) was developed by Walther Nernst (1906-1912). Alternate statements by Nernst, Simon and by Lewis & Randall clarify practical and limiting aspects of reaching absolute zero.
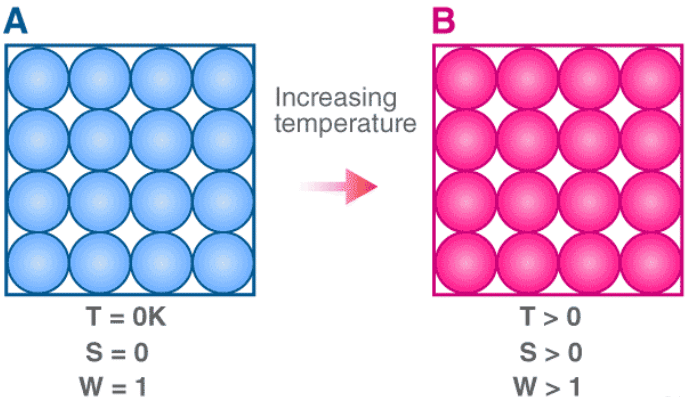
Consequences and implications
- It is impossible to reach absolute zero in a finite number of steps. As temperature approaches 0 K, the number of steps or the effort required to reduce the temperature further tends to infinity.
- The third law permits calculation of absolute entropies of substances by integrating heat capacities from 0 K to the temperature of interest.
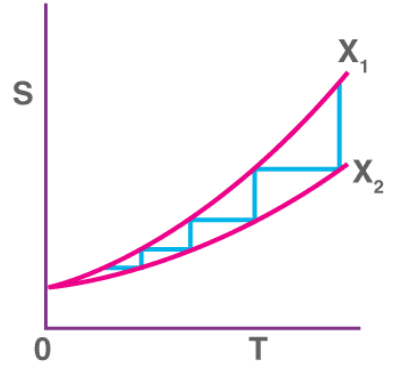
Mathematical use - absolute entropy
If S(0) is taken as zero for a perfect crystal at 0 K, the entropy at temperature T is given by
S(T) = ∫0T (Cp / T) dT
Practically, Cp is measured as a function of T and the integral evaluated. If Cp is approximately constant over a temperature range, a simplifying approximation is
S ≈ Cp ln T
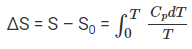
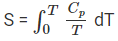
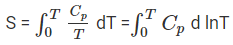
Limitations
- Not all solids have S(0) = 0 in practice. Examples are amorphous solids (glass), solids with structural disorder, or mixtures of isotopes where residual entropy remains at 0 K.
Entropy and Thermodynamic Laws (summary)
- First law: conservation of energy, heat is a form of energy.
- Second law: entropy of the universe tends to increase; spontaneous processes increase total entropy; complete conversion of heat into work without losses is impossible.
- Third law: entropy of a perfect crystal approaches zero as T → 0 K, allowing determination of absolute entropies.
Entropy changes during phase transitions
- Entropy of fusion (melting)
ΔfusS = ΔfusH / Tf, where ΔfusH is enthalpy of fusion and Tf is the melting temperature. Normally ΔfusS > 0 because molecules gain freedom on melting. Exceptions at very low temperatures (e.g. helium isotopes) are known in specialised cases. - Entropy of vaporisation
ΔvapS = ΔvapH / Tb, where ΔvapH is enthalpy of vaporisation and Tb is boiling temperature. Vaporisation normally shows a large positive entropy change because gas phase has far greater disorder than liquid.
Standard entropy of formation
The standard molar entropy of a compound is the entropy change when one mole of the compound in its standard state is formed from the elements in their standard states. Standard absolute entropies (S°) are tabulated and positive for all substances except a perfect crystal at 0 K.
Spontaneity
Spontaneity judged by entropy: a process is spontaneous if the total entropy change of system plus surroundings is positive (ΔStotal > 0). This criterion requires knowledge of both the system and surroundings.
- Exothermic reactions often make ΔSsurroundings positive (heat released to surroundings), favouring spontaneity.
- Endothermic reactions can be spontaneous if the system entropy increases sufficiently to make ΔStotal positive.
- Free energy (Gibbs free energy) is a more convenient single-system criterion in many practical cases: a process at constant T and P is spontaneous when ΔG < 0 (this relation is consistent with entropy criteria because ΔG = ΔH - TΔS and ΔG relates to ΔSuniv by ΔSuniv = -ΔG / T).
Negentropy
Negentropy (negative entropy) refers to local decreases in entropy (increased order). Such local decreases are permitted if compensated by a larger increase of entropy elsewhere, so that the entropy of the universe still increases. Organised systems (for example, biological order or planetary systems) show local negentropy while increasing total entropy of the surroundings.
Worked and Practice Questions
Q.1. The entropy of an isolated system can never ____?
(a) increase
(b) decrease
(c) be zero
(d) none of the mentioned
Ans: b
Explanation: The entropy of an isolated system always increases and remains constant only when the process is reversible.
Q.2. According to the entropy principle, the entropy of an isolated system can never decrease and remains constant only when the process is reversible?
(a) true
(b) false
Ans: a
Explanation: This is the statement for the principle of increase of entropy.
Q.3. Entropy may decrease locally in some region within the isolated system. How can this statement be justified?
(a) this cannot be possible
(b) this is possible because the entropy of an isolated system can decrease
(c) it must be compensated by a greater increase of entropy somewhere within the system.
(d) none of the mentioned
Ans: c
Explanation: The net effect of an irreversible process is an entropy increase of the whole system.
Q.4. Clausius summarized the first and second laws of thermodynamics as?
(a) the energy of the world is constant
(b) the entropy of the world tends towards a maximum
(c) both of the mentioned
(d) none of the mentioned
Ans: c
Explanation: These two statements were given by Clausius.
Q.5. The entropy of an isolated system always ____ and becomes a ____ at the state of equilibrium?
(a) decreases, minimum
(b) increases, maximum
(c) increases, minimum
(d) decreases, maximum
Ans: b
Explanation: If the entropy of an isolated system varies with some parameter, then there is a certain value of that parameter that maximizes the entropy.
FAQs on 2nd & 3d Law: Entropy Change & Spontaneity
| 1. What is entropy and how does it relate to entropy change in a system? |  |
| 2. What factors affect the entropy change in a system? |  |
| 3. How can we determine the spontaneity of a process based on entropy change? |  |
| 4. Can entropy decrease in a system? |  |
| 5. How does the concept of entropy relate to everyday life? |  |







