First Law of Thermodynamics
Introduction
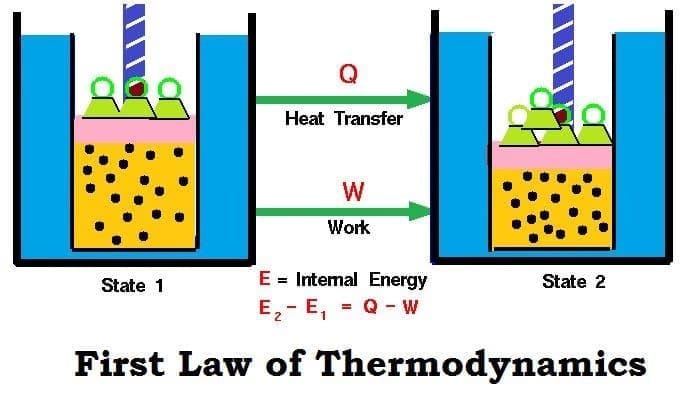
The first law of thermodynamics is a statement of energy conservation for thermodynamic systems. It states that energy can neither be created nor destroyed, but it can be transformed from one form to another. In other words, the energy of an isolated system is constant. Heat and work are two modes by which energy is exchanged between a system and its surroundings.
Definitions and basic concepts
- System - the part of the universe chosen for study.
- Surroundings - everything outside the system that can exchange energy or matter with the system.
- Isolated system - neither matter nor energy is exchanged with the surroundings.
- Closed system - matter is fixed; energy in the form of heat or work may be exchanged.
- Open system - both matter and energy can be exchanged with the surroundings.
- State function - a property that depends only on the state of the system (for example, internal energy U).
- Path function - a quantity that depends on the path taken (for example, heat q and work w).
- Internal energy (U or E) - the total microscopic energy of a system (kinetic + potential at molecular level). It is a state function.
Mathematical expression of the first law
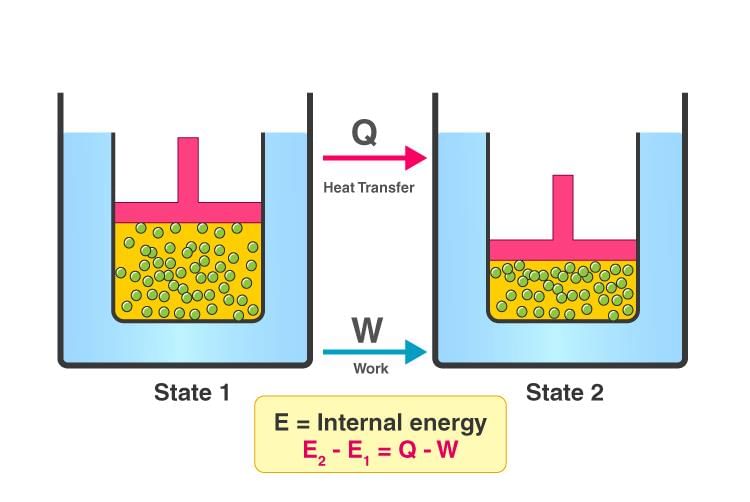
Consider a closed system whose internal energy in an initial equilibrium state is U1 and in a final equilibrium state is U2. If the system receives heat q from the surroundings and an amount of work w is done on the system, then the change in internal energy is
U2 - U1 = q + w
or
ΔU = q + w
Note on sign conventions:
- In the convention used in chemistry, w is positive when work is done on the system and negative when work is done by the system. With this convention, ΔU = q + w.
- Some texts define w as the work done by the system; with that convention ΔU = q - w(by). Both conventions are equivalent once you are consistent in using one.
PV (mechanical) work
For expansion or compression of a gas against an external pressure Pext, the mechanical work done by the system is
w = -∫initialfinal Pext dV
For a process against a constant external pressure,
w = -Pext ΔV
When the gas expands (ΔV > 0), the system does work on surroundings and w is negative (chemistry sign convention: w<0). when the gas is compressed (δv < 0), work is done on the system and w is>
Two illustrative cases
Case 1 - heat absorbed by the system and system does work
- Let UA and UB be internal energies in states A and B respectively.
- If heat q is absorbed by the system from surroundings, q > 0 (this tends to increase internal energy).
- If the system performs work w on the surroundings, then, in the chemistry sign convention, w is negative (this tends to decrease internal energy).
Therefore,
ΔU = q + (-w) = q - w
Case 2 - heat absorbed and work done on the system
If heat q is absorbed and work w is done on the system, both q and w are positive and
ΔU = q + w
Consequences and special cases
ΔU = q + w
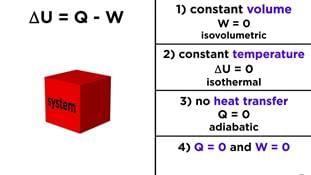
- (i) When ΔU = 0 - there is no net change in internal energy. The first law gives 0 = q + w, so q = -w. Heat absorbed equals work done by the system (and vice versa).
- (ii) No work exchange (w = 0) - the first law reduces to ΔU = q. The change in internal energy equals heat exchanged.
- (iii) Adiabatic process (q = 0) - the first law reduces to ΔU = w. If work is done on the system, its internal energy increases; if work is done by the system, its internal energy decreases.
- (iv) Constant pressure expansion of a gas - if a gas expands against constant external pressure P and volume change ΔV, the mechanical work done by the gas is w = -P ΔV.
Solved numerical
Example 1. In a certain process, 670 J of heat is absorbed by a system while 264 J of work is done on the system. What is the change in internal energy of the process?
Solution.
q = +670 J (heat absorbed by the system)
w = +264 J (work done on the system)
ΔE = q + w
ΔE = 670 + 264
ΔE = 934 J
Example 2. If 150 J of heat is added to a system while the system does work equivalent of 255 J by expanding against the surrounding atmosphere, what is the value of ΔE for the system?
Solution.
q = +150 J (heat absorbed)
w = -255 J (work done by system; negative in chemistry convention)
ΔE = q + w
ΔE = +150 + (-255)
ΔE = -105 J
Example 3. If 500 calorie of heat energy are added to a system and the system does 350 calorie of work on the surroundings, what is the energy change of the system?
Solution.
q = +500 cal
w = -350 cal
ΔE = q + w
ΔE = 500 + (-350)
ΔE = 150 cal
Try Yourself!
Q.1. A 1.0 kJ of heat is added to 1.50 L of ideal gas in a cylinder at constant pressure of 1.00 atm, the volume increases to 2.30 L. Calculate ΔE for the process.
Ans. 919.2 J
Q.2. How much work is done on or by the system, if the internal energy of the system decreases by 100 J when 200 J of heat is supplied to the system.
Ans. - 300 J
Reversible processes

A thermodynamic process is reversible if it can be reversed at any stage by an infinitesimal change in external conditions such that both the system and surroundings are returned exactly to their initial states and no net change occurs elsewhere in the universe. Reversible processes are idealisations - they proceed infinitely slowly and pass through a continuous series of equilibrium states.
- Perfectly reversible processes do not occur in reality but are useful models to define maximum possible efficiency.
- An ideal reversible process takes infinite time to complete and is carried out by infinitesimal changes.
Formal statement: A process is reversible if it can be reversed by an infinitesimal modification of a variable so that the system and surroundings are restored to their original states.
- Examples of processes that can be approximated as reversible: extension of ideal springs, slow (quasi-static) adiabatic compression/expansion of gases, slow isothermal compression/expansion of gases, frictionless motion, and ideal electrolysis with negligible resistance.
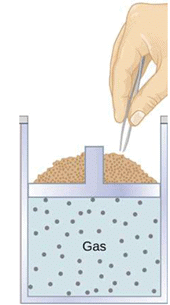
Illustration: An ideal gas in a cylinder fitted with a massless piston loaded with many tiny sand grains can be expanded reversibly by removing grains one by one. Each removal is infinitesimal so the gas remains in near-equilibrium throughout the process.
Irreversible processes
An irreversible process is one in which the system and surroundings cannot be returned to their original states by infinitesimal changes. Most natural processes are irreversible because of dissipative effects and spontaneous changes.
Causes of irreversibility include:
- Friction and viscous dissipation that convert mechanical energy into heat.
- Unrestrained (free) expansion of a gas where work recovery is impossible.
- Heat transfer over a finite temperature difference (finite ΔT) - the reverse process cannot proceed under the same finite ΔT conditions.
- Mixing of different substances that spontaneously increases entropy and cannot be undone without external work.
Examples of irreversible processes:
- Relative motion with friction.
- Throttling (flow through a restriction).
- Heat flow from hot to cold over a finite temperature difference.
- Diffusion and spontaneous mixing.
- Electric current through a resistive element (Joule heating).
Processes other than reversible processes are known as irreversible processes.
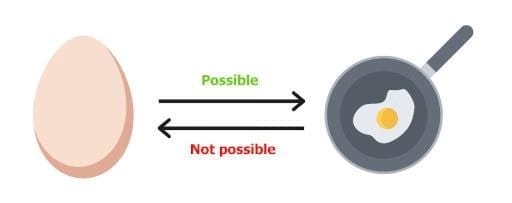 Reversible and Irreversible Process
Reversible and Irreversible Process
FAQs on First Law of Thermodynamics
| 1. What is the First Law of Thermodynamics? |  |
| 2. What is the mathematical expression of the First Law of Thermodynamics? |  |
| 3. Can you provide an example of a solved numerical problem related to the First Law of Thermodynamics? |  |
| 4. What is a reversible process in thermodynamics? |  |
| 5. What is an irreversible process in thermodynamics? |  |







