NCERT Exemplar: Thermodynamics | Chemistry Class 11 - NEET PDF Download
Multiple Choice Questions
Q.1. Thermodynamics is not concerned about______.
(1) Energy changes involved in a chemical reaction.
(2) The extent to which a chemical reaction proceeds.
(3) The rate at which a reaction proceeds.
(4) The feasibility of a chemical reaction.
Ans. (3)
Solution.
Thermodynamics is not concerned about how and at what rate these energy transformations are carried out, but is based on initial and final states of a system undergoing the change. Laws of thermodynamics apply only when a system is in equilibrium or moves from one equilibrium state to another equilibrium state.
Q.2. Which of the following statements is correct?
(1) The presence of reacting species in a covered beaker is an example of open system.
(2) There is an exchange of energy as well as matter between the system and the surroundings in a closed system.
(3) The presence of reactants in a closed vessel made up of copper is an example of a closed system.
(4) The presence of reactants in a thermos flask or any other closed insulated vessel is an example of a closed system.
Ans. (3)
Solution.
The presence of reactants in a closed vessel made of conducting material e.g., copper or steel is an example of a closed system.
Q.3. The state of a gas can be described by quoting the relationship between___.
(1) Pressure, volume, temperature
(2) Temperature, amount, pressure
(3) Amount, volume, temperature
(4) Pressure, volume, temperature, amount
Ans. (4)
Solution.
Variables like p, V, T are called state variables or state functions because their values depend only on the state of the system and not on how it is reached.
Q.4. The volume of gas is reduced to half from its original volume. The specific heat will be ______.
(1) Reduce to half
(2) Be doubled
(3) Remain constant
(4) Increase four times
Ans. (3)
Solution.
Specific heat capacity is the quantity of heat required to raise the temperature of one unit mass of a substance by one degree celsius (or one kelvin). That is why it is an a intensive property which does not depend on mass.
Q.5. During complete combustion of one mole of butane, 2658 kJ of heat is released.The thermochemical reaction for above change is
(1) 2C4H10(g) + 13O2(g) → 8CO2(g) + 10H2O(l) ΔcH = -2658.0 kJ mol-1
(2) 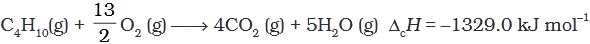
(3) 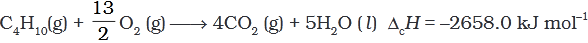
(3) 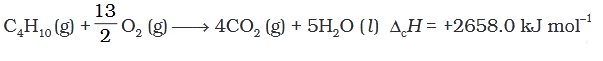
Ans. (3)
Solution.
Standard enthalpy of combustion is defined as the enthalpy change per mole (or per unit amount) of a substance, when it undergoes combustion and all the reactants and products being in their standard states at the specified temperature.
Q.6. ∆fUΘ of formation of CH4 (g) at certain temperature is –393 kJ mol–1. The value of ∆fHΘ is
(1) zero
(2) < ∆f UΘ
(3) > ∆f UΘ
(4) equal to ∆f UΘ
Ans. (2)
Solution.
∆fH = ∆fUΘ + ∆ngRT
For the reaction,
C(s) + 2H2(g) → CH4(g)
∴ ∆ng = 1 - 2 = -1
∴ ∆fHΘ = ∆fUΘ - 1 RT
or ∆fHΘ = ∆fUΘ + RT
or ∆fHΘ < ∆fUΘ
Q.7. In an adiabatic process, no transfer of heat takes place between system and surroundings. Choose the correct option for free expansion of an ideal gas under adiabatic condition from the following.
(1) q = 0, ∆T ≠0, w = 0
(2) q ≠ 0, ∆T =0, w = 0
(3) q = 0, ∆T =0, w = 0
(4) q = 0, ∆T <0, w ≠ 0
Ans. (3)
Solution.
For free expansion w = 0 for adiabatic process q = 0 from first law of thermodynamics,
∆U = q + w
= 0 + 0 = 0
Since there is no change of internal energy, hence temperature will also remain constant, i.e., ∆T = 0
Q.8. The pressure-volume work for an ideal gas can be calculated by using the expression 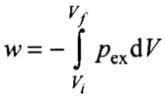
The work can also be calculated from the pV– plot by using the area under the curve within the specified limits. When an ideal gas is compressed
(a) reversibly or (b) irreversibly from volume Vi to Vf. choose the correct option.
(1) w(reversible) = w (irreversible)
(2) w(reversible) < w (irreversible)
(3) w(reversible) > w (irreversible)
(4) w(reversible) = w (irreversible) + pex.∆V
Ans. (2)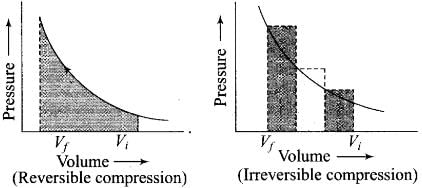
Area under the curve is greater in irreversible compression than that of reversible compression.
Q.9. The entropy change can be calculated by using the expression 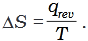
When water freezes in a glass beaker, choose the correct statement amongst the following :
(1) ∆S (system) decreases but ∆S (surroundings) remains the same.
(2) ∆S (system) increases but ∆S (surroundings) decreases.
(3) ∆S (system) decreases but ∆S (surroundings) increases.
(4) ∆S (system) decreases and ∆S (surroundings) also decreases.
Ans. (3)
Solution.
During the process of freezing energy is released, which is absorbed by the surroundings.
∴ 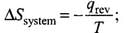

Therefore, the entropy of the system decreases and that of surroundings increases.
Q.10. On the basis of thermochemical equations (a), (b) and (c), find out which of the algebraic relationships given in options (i) to (iv) is correct.
(a) C (graphite) + O2(g) → CO2 (g) ;ΔrH = x kJ mol-1
(b) 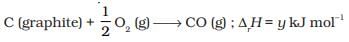
(c) 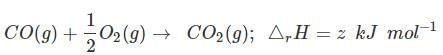
(1) z = x + y
(2) x = y – z
(3) x = y + z
(4) y = 2z – x
Ans. (3)
Solution.
C (graphite) + O2(g) → CO2 (g) ;ΔfH = x kJ mol-1 ...(i)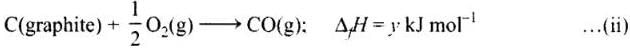
Subtracting equation (i) and (ii) we get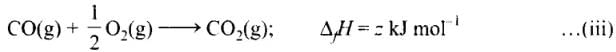
Equation (iii) is obtained by subtracting (ii) from (i)
Therefore,
z = x - y
or x = y + z
Q.11. Consider the reactions given below. On the basis of these reactions find out which of the algebraic relations given in options (i) to (iv) is correct?
(a) C(g) + 4H(g) → CH4(g); ∆rH = x kJ mol–1
(b) C(graphite,s) + 2H2(g) → CH4(g); ∆rH = y kJ mol–1
(1) x = y
(2) x = 2y
(3) x > y
(4) x < y
Ans. (3)
Solution.
Same bonds are formed in reaction (a) and (b) but bonds between the reactant molecules are broken only in reaction (b).
Q.12. The enthalpies of elements in their standard states are taken as zero. The enthalpy of formation of a compound
(1) Is always negative
(2) Is always positive
(3) May be positive or negative
(4) Is never negative
Ans. (3)
Solution.
Heat of formation of a compound may be positive or negative,
Example,
C(s) + O2(g) → CO2(g);
ΔHº = -393.5 kJ mol-1
ΔHº = +92 kJ mol-1
Q.13. Enthalpy of sublimation of a substance is equal to
(1) Enthalpy of fusion + enthalpy of vapourisation
(2) Enthalpy of fusion
(3) Enthalpy of vapourisation
(4) Twice the enthalpy of vapourisation
Ans. (1)
Solution.
Enthalpy of sublimation of a substance is equal to enthalpy of fusion + enthalpy of vapourisation.
Sublimation is direct conversion of solid to vapour, i.e., solid → vapour
Writing in two steps, we have solid → liquid → vapour.
Solid → liquid requires enthalpy of fusion.
Liquid →vapour requires enthalpy of vapourisation.
Q.14. Which of the following is not correct?
(1) ∆G is zero for a reversible reaction
(2) ∆G is positive for a spontaneous reaction
(3) ∆G is negative for a spontaneous reaction
(4) ∆G is positive for a non-spontaneous reaction
Ans. (2)
Solution.
∆G gives a criterion for spontaneity at constant pressure and temperature.
(a) If ∆G is negative (< 0). the process is spontaneous.
(b) If ∆G is positiv e (> 0). the process is non-spontaneous.
(c) If ∆G is zero then reaction is at equilibrium.
Q.15. Thermodynamics mainly deals with
(1) Interrelation of various forms of energy and their transformation from one form to another.
(2) Energy changes in the processes which depend only on initial and final states of the microscopic systems containing a few molecules.
(3) How and at what rate these energy transformations are carried out.
(4) The system in equilibrium state or moving from one equilibrium state to another equilibrium state.
Ans. (1, 4)
Solution.
Thermodynamics deals with interrelation of various forms of energy and their transformation into each other. It also deals with thermal or mechanical equilibrium. However, if does not tell anything about the rate of reaction.
Q.16. In an exothermic reaction, heat is evolved, and system loses heat to the surrounding. For such system
(1) qp will be negative
(2) ∆rH will be negative
(3) qp will be positive
(4) ∆rH will be positive
Ans. (1, 2)
Solution.
In an exothermic reaction, heat is evolved, and system loses heat to the surroundings. Therefore, qp will be negative and ∆rH will also be negative.
Q.17. The spontaneity means, having the potential to proceed without the assistance of external agency. The processes which occur spontaneously are
(1) Flow of heat from colder to warmer body.
(2) Gas in a container contracting into one corner.
(3) Gas expanding to fill the available volume.
(4) Burning carbon in oxygen to give carbon dioxide.
Ans. (1, 4)
Solution.
Gas expands or diffuses in available space spontaneously, e.g., leakage of cooking gas gives smell of ethyl mercaptan spontaneously. Moreover, burning of carbon to CO2 is also spontaneous.
Q.18. For an ideal gas, the work of reversible expansion under isothermal condition can be calculated by using the expression 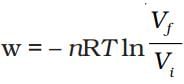
A sample containing 1.0 mol of an ideal gas is expanded isothermally and reversibly to ten times of its original volume, in two separate experiments.
The expansion is carried out at 300 K and at 600 K respectively. Choose the correct option.
(1) Work done at 600 K is 20 times the work done at 300 K.
(2) Work done at 300 K is twice the work done at 600 K.
(3) Work done at 600 K is twice the work done at 300 K.
(4) ∆U = 0 in both cases.
Ans. (3, 4)
Solution.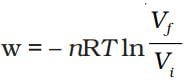 .
.
Here,
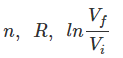
are same in both the cases,
therefore,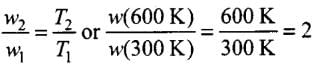
i.e., work done at 600 K is twice the work done at 300 K. Since each case involves isothermal expansion of an ideal gas, there is no change in internal energy,
i.e., ∆U = 0.
Q.19. Consider the following reaction between zinc and oxygen and choose the correct options out of the options given below:
2Zn(s) + O2 (g) → 2ZnO (s) ; ∆H = – 693.8 kJ mol–1
(1) The enthalpy of two moles of ZnO is less than the total enthalpy of two moles of Zn and one mole of oxygen by 693.8 kJ.
(2) The enthalpy of two moles of ZnO is more than the total enthalpy of two moles of Zn and one mole of oxygen by 693.8 kJ.
(3) 693.8 kJ mol–1 energy is evolved in the reaction.
(4) 693.8 kJ mol–1 energy is absorbed in the reaction.
Ans. (1, 3)
Solution.
In the reaction:
2Zn(s) + O2(g) → 2ZnO(s); ΔrH = -693.8 kJ mol-1
ΔrH = ∑ Enthalpy of formation of products
-∑ Enthalpy of formation of reactants
= [2ΔfHZnO] - [2ΔfHZn + Δf HO2]
For exothermic reactions, ΔrH is negative, i.e., 693.8 kJ heat is evolved.
∴ Enthalpy of 2 moles of ZnO will less than the enthalpy of two moles of zinc and one mole of O2.
SHORT ANSWER TYPE QUESTION
Q.20. 18.0 g of water completely vapourises at 100°C and 1 bar pressure and the enthalpy change in the process is 40.79 kJ mol–1. What will be the enthalpy change for vapourising two moles of water under the same conditions? What is the standard enthalphy of vapourisation for water?
Ans. Enthalpy of a reaction is the energy change per mole for the process.
18 g of H2O = 1 mole (∆Hvap = 40.79 kJ moE1)
Enthalpy change for vapourising 2 moles of H2O = 2 × 40.79 = 81.58 kJ ∆H°vap = 40.79 kJ mol -1
Q.21. One mole of acetone requires less heat to vapourise than 1 mol of water. Which of the two liquids has higher enthalpy of vapourisation?
Ans. Due to weak force of attraction between molecules, acetone requires less heat to vaporise. Hence, water has higher enthalpy of vaporisation.
Q.22. Standard molar enthalpy of formation, ∆fHΘ is just a special case of enthalpy of reaction, ∆rHΘ. Is the ∆rHΘ for the following reaction same as ∆fHΘ? Give reason for your answer.
CaO(s) + CO2(g) → CaCO3(s); ∆fHΘ = –178.3 kJ mol–1
Ans. No, standard molar enthalpy of formation is the enthalpy of the reaction when 1 mole of compound is formed from its constituent elements.
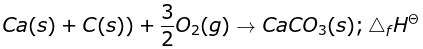
As the close reaction is different from the given reaction,
∴ ΔrHΘ ≠ ΔfHΘ
Q.23. The value of ∆fHΘ for NH3 is –91.8 kJ mol–1. Calculate enthalpy change for the following reaction:
2NH3(g) → N2(g) + 3H2(g)
Ans.
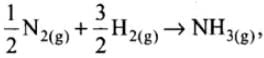
ΔfHΘ= -91.8 kJ mol-1
or N2(g) + 3H2(g) → 2NH3(g),
ΔfHΘ = -2 x 91.8 = -183.6 kJ mol-1
So, for the reverse reaction,
ΔrHΘ = 183.6 kJ mol-1.
∴ The value of ΔrHΘ for NH3 is +183.6 kJ mol-1
Q.24. Enthalpy is an extensive property. In general, if enthalpy of an overall reaction A→B along one route is ∆r H and ∆rH1, ∆rH2, ∆rH3 ..... represent enthalpies of intermediate reactions leading to product B. What will be the relation between ∆rH for overall reaction and ∆rH1 , ∆rH2 ..... etc. for intermediate reactions.
Ans. By Hess’s law ∆rH = ∆rH1 + ∆rH2 + ∆rH3……………………
Q.25. The enthalpy of atomisation for the reaction CH4(g) → C(g) + 4H (g) is 1665 kJ mol–1. What is the bond energy of C–H bond?
Ans. H4 → C + 4H, ∆H= + 1665 kJ mol-1
Bond energy of (C – H) bond = 1665/4 = 416.2 kJ mol
Q.26. Use the following data to calculate ∆latticeHΘ for NaBr.
∆subHΘ for sodium metal = 108.4 kJ mol–1
Ionization enthalpy of sodium = 496 kJ mol–1
Electron gain enthalpy of bromine = – 325 kJ mol–1
Bond dissociation enthalpy of bromine = 192 kJ mol–1
∆fHΘ for NaBr (s) = – 360.1 kJ mol–1
Ans.
(i) Na(s) → Na(g), ΔsubHΘ = 108.4 kJ mol-1
(ii) Na → Na+ + e-, ΔiHΘ = 496 kJ mol-1
(iii) 1/2 Br2 → Br, 1/2 ΔdissHΘ = 192/2 = 96 kJ mol-1
(iv) Br + e- → Br-, ΔegHΘ = -325 kJ mol-1
ΔfHΘ for NaBr = -360.1 kJ mol -1
Enthalpies in different steps involved in the formation of NaBr(s) are.
ΔfHΘ = ΔsubHΘ + 1/2 ΔsubHΘ + ΔiHΘ + ΔegHΘ + ∆latticeHΘ
∴ ∆latticeHΘ = - 360.1 - 108.4 - 96 - 496 + 325
= -735.5 kJ mol-1
Q.27. Given that ∆H = 0 for mixing of two gases. Explain whether the diffusion of these gases into each other in a closed container is a spontaneous process or not?
Ans. It is a spontaneous process. Although enthalpy change is zero but randomness or disorder (ΔS) increases and ΔS is positive. Therefore, in equation, ΔG = ΔH – TΔS, the term TΔS will be negative. Hence ΔG will be negative.
Q.28. Heat has randomising influence on a system and temperature is the measure of average chaotic motion of particles in the system. Write the mathematical relation which relates these three parameters.
Ans. Heat has randomising influence on a system and temperature is the measure of average chaotic motion of particles in the system. The mathematical relation which relates these three parameters is ΔS = qrev/ T
Here, ΔS = change in entropy
qrcv = heat of reversible reaction
T = temperature
Q.29. Increase in enthalpy of the surroundings is equal to decrease in enthalpy of the system. Will the temperature of system and surroundings be the same when they are in thermal equilibrium?
Ans. Yes. If the system is in thermal equilibrium with the surroundings, then the temperature of the surroundings is same as that of the system. Also, increase in enthalpy of the surroundings is equal to decrease in the enthalpy of the system.
Q.30. At 298 K. Kp for the reaction N2O4 (g) ⇌ 2NO2 (g) is 0.98. Predict whether the reaction is spontaneous or not.
Ans.
ΔrGΘ = -RT ln Kp
= -RT ln (0.98)
Since In (0.98) is negative
∴ ΔrGΘ is positive
⇒ the reaction is non spontaneous
Q.31. A sample of 1.0 mol of a monoatomic ideal gas is taken through a cyclic process of expansion and compression as shown in Figure. What will be the value of ∆H for the cycle as a whole?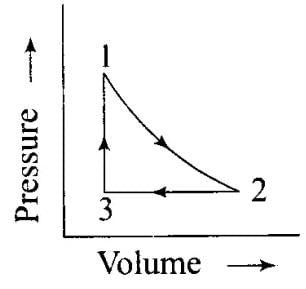 Ans. For a cyclic process, ΔH = 0
Ans. For a cyclic process, ΔH = 0
Q.32. The standard molar entropy of H2O (l) is 70 J K–1 mol–1. Will the standard molar entropy of H2O(s) be more, or less than 70 J K–1 mol–1?
Ans. The standard molar entropy of H2O (1) is 70 J K-1 mol-1. The solid form of H2O is ice. In ice, molecules of H2O are less random than in liquid water. Thus, molar entropy of H2O (s) < molar entropy of H2O (1). The standard molar entropy of H2O (s) is less than 70 J K1 mol-1.
Q.33. Identify the state functions and path functions out of the following: enthalpy, entropy, heat, temperature, work, free energy.
Ans. State functions: Enthalpy, entropy, temperature, free energy Path functions: Heat, work.
Short Answer Type Question
Q.34. The molar enthalpy of vapourisation of acetone is less than that of water. Why?
Ans. Molar enthalpy of vapourisation is more for water due to hydrogen bonding between water molecules.
Q.35. Which quantity out of ∆rG and ∆rGΘ will be zero at equilibrium?
Ans. Gibbs energy for a reaction in which all reactants and products are in standard state. ΔrGΘ is related to the equilibrium constant of the reaction as follows
ΔrG = ArGΘ + RT In K
At equilibrium, 0 = ΔrGΘ + RT In A– (ΔrG = 0) or ΔrG° = -RT lnK
ΔrG° = 0 when K= 1
For all other values of K, ArG° will be non-zero.
Q.36. Predict the change in internal energy for an isolated system at constant volume.
Ans. For an isolated system w = 0, q = 0
Since ΔU= q + w = 0 + 0 = 0, ΔU= 0
Q.37. Although heat is a path function but heat absorbed by the system under certain specific conditions is independent of path. What are those conditions? Explain.
Ans.
At constant volume
q = ΔU + (-w)
-w = pΔ q = AU + pΔV
ΔV = 0 (at constant volume)
Hence,
qv = ΔU + 0 = ΔU= change in internal energy At constant pressure,
qp = AU + pΔV
Since ΔU + pΔV=ΔH
⇒ qp = ΔH change in enthalpy
Hence, at constant volume and at constant pressure, heat change is a state function because it is equal to ΔU and ΔH respectively which are state functions.
Q.38. Expansion of a gas in vacuum is called free expansion. Calculate the work done and the change in internal energy when 1 litre of ideal gas expands isothermally into vacuum until its total volume is 5 litre?
Ans. During free expansion, external pressure is zero, so Work done, w = -pextΔV
= -0(5 – 1) = 0
Since the gas is expanding isothermally, therefore, q = 0
ΔU = q + w = 0 + 0 = 0
Q.39. Heat capacity (Cp) is an extensive property but specific heat (c) is an intensive property. What will be the relation between Cp and c for 1 mol of water?
Ans.
For water, molar heat capacity = 18 × Specific heat or
Cp = 18 × c
But, specific heat,
C = 4.18 J g-1 K-1 Heat capacity,
Cp = 18 × 4.18 JK 1 = 75.24 JK-1
Q.40. The difference between CP and CV can be derived using the empirical relation H = U + pV. Calculate the difference between CP and CV for 10 moles of an ideal gas.
Ans. Given that, Cv = heat capacity at constant volume,
Cp = heat capacity at constant pressure Difference between Cp and Cv is equal to gas constant (R).
∴ Cp – Cv = nR (where, n = no. of moles)
= 10 × 8.314 = 83.14J
Q.41. If the combustion of 1g of graphite produces 20.7 kJ of heat, what will be molar enthalpy change? Give the significance of sign also.
Ans. Molar enthalpy change for graphite (ΔH)
= enthalpy change for 1 g × molar mass of C = -20.7 × 12 = -2.48 × 102 kJ mol-1
Since the sign of ΔH = -ve, it is an exothermic reaction.
Q.42. The net enthalpy change of a reaction is the amount of energy required to break all the bonds in reactant molecules minus amount of energy required to form all the bonds in the product molecules. What will be the enthalpy change for the following reaction.
H2(g) + Br2(g) → 2HBr(g)
Given that Bond energy of H2, Br2 and HBr is 435 kJ mol–1, 192 kJ mol–1 and 368 kJ mol–1 respectively.
Ans. ΔrHΘ ∑B.E.(reactants) -∑B.E.(products)
= B.E.H2 + B.E.Br2 - 2 × B.E. HBr
= 435 + 192 - (2 × 368)
= - 109 kJ mol-1
Q.43. The enthalpy of vapourisation of CCl4 is 30.5 kJ mol–1. Calculate the heat required for the vapourisation of 284 g of CCl4 at constant pressure. (Molar mass of CCl4 = 154 g mol–1).
Ans. Given that enthalpy of vaporization of 1 mole of CCI4 = 30.5 kJ/mol.
It means 1 mole of CCl4 = 154 g.
154 g requires heat of vaporization = 30.5 kJ/mol.
∴ Heat required for vapourisation of 284 g of CCl4
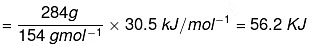 Q.44. The enthalpy of reaction for the reaction:
Q.44. The enthalpy of reaction for the reaction:
2H2(g) + O2(g) → 2H2O(l) is ∆rHΘ = – 572 kJ mol–1.
What will be standard enthalpy of formation of H2O(l) ?
Ans. The standard enthalpy change for the formation of one mole of a compound from its elements in their most stable states of aggregation (also known as reference states) is called Standard Molar Enthalpy of Formation. Its symbol is ∆fHΘ.
According to the definition of standard enthalpy of formation, the enthalpy change for the following reaction will be standard enthalpy of formation of H2O(l).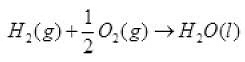 or the standard enthalpy of formation of H2O(l) will be half of the enthalpy of the given equation i.e., ∆rHΘ is also halved.
or the standard enthalpy of formation of H2O(l) will be half of the enthalpy of the given equation i.e., ∆rHΘ is also halved.
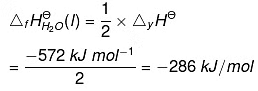 Q.45. What will be the work done on an ideal gas enclosed in a cylinder, when it is compressed by a constant external pressure, pext in a single step as shown in Figure. Explain graphically.
Q.45. What will be the work done on an ideal gas enclosed in a cylinder, when it is compressed by a constant external pressure, pext in a single step as shown in Figure. Explain graphically.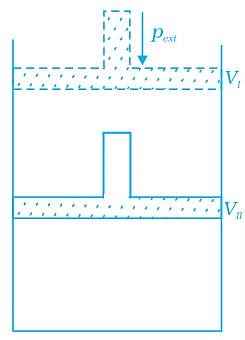 Ans. Let us consider a cylinder which contains one mole of an ideal gas fitted with a frictionless piston. Total volume of the gas is Vi and pressure of the gas inside is p. If external pressure is pext which is greater than p, piston is moved inward till the pressure inside becomes equal to pext. Let this change be achieved in a single step and the final volume be Vf. W = Pext (ΔV). Work done is equal to the shaded area ABVIVII.
Ans. Let us consider a cylinder which contains one mole of an ideal gas fitted with a frictionless piston. Total volume of the gas is Vi and pressure of the gas inside is p. If external pressure is pext which is greater than p, piston is moved inward till the pressure inside becomes equal to pext. Let this change be achieved in a single step and the final volume be Vf. W = Pext (ΔV). Work done is equal to the shaded area ABVIVII.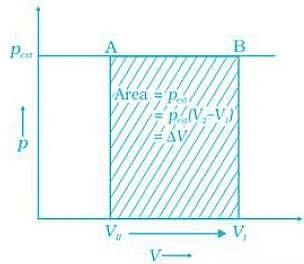 Q.46. How will you calculate work done on an ideal gas in a compression, when change in pressure is carried out in infinite steps?
Q.46. How will you calculate work done on an ideal gas in a compression, when change in pressure is carried out in infinite steps?
Ans. A process or change is said to be reversible, if a change is brought out in such a way that the process could, at any moment, be reversed by an infinitesimal change.
Work done can be calculated from pV-plot when pressure is not constant and changes are in infinite steps (reversible conditions) during compression from initial volume, Vi to final volume, Vf. Work done on the gas is represented by the shaded area.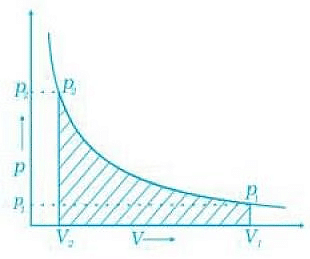 Q.47. Represent the potential energy/enthalpy change in the following processes graphically.
Q.47. Represent the potential energy/enthalpy change in the following processes graphically.
(a) Throwing a stone from the ground to roof In which of the processes potential energy/enthalpy change is contributing factor to the spontaneity?
In which of the processes potential energy/enthalpy change is contributing factor to the spontaneity?
Ans. (a) On throwing stone from ground to the roof we need to supply energy to the stone. Graphically, it can be represented as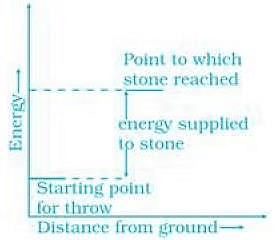 (b) In the reaction heat is evolved it means the process is followed by decrease in energy. It can be represented as
(b) In the reaction heat is evolved it means the process is followed by decrease in energy. It can be represented as
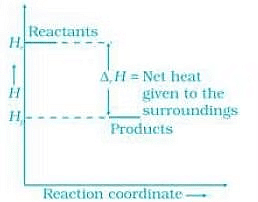
Energy increases in (a) part and decreases in (b). Hence, in process (b), the enthalpy change is the contributing factor to the spontaneity.
Q.48. Enthalpy diagram for a particular reaction is given in Figure. Is it possible to decide spontaneity of a reaction from given diagram. Explain.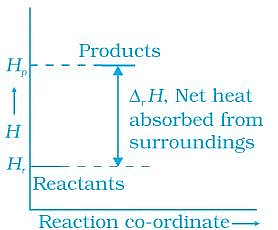 Ans. No, enthalpy is not the only criteria for spontaneity we need to consider enthalpy and entropy both.
Ans. No, enthalpy is not the only criteria for spontaneity we need to consider enthalpy and entropy both.
Q.49. 1.0 mol of a monoatomic ideal gas is expanded from state (1) to state (2) as shown in Figure. Calculate the work done for the expansion of gas from state (1) to state (2) at 298 K.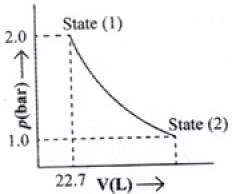 Ans. From the given diagram, it is very clear that it is a reversible change because the process is
Ans. From the given diagram, it is very clear that it is a reversible change because the process is
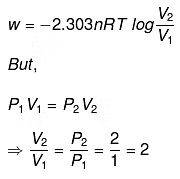 carried out in infinite no. of steps
carried out in infinite no. of steps
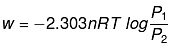 = - 2.303 x 1 mol x 8.314 J mol-1 K-1 x 298 K-1 x log 2
= - 2.303 x 1 mol x 8.314 J mol-1 K-1 x 298 K-1 x log 2
= - 2.303 x 8.314 x 298 x 0.3010 J = -1717.46 J
Q.50. An ideal gas is allowed to expand against a constant pressure of 2 bar from 10 L to 50 L in one step. Calculate the amount of work done by the gas. If the same expansion were carried out reversibly, will the work done be higher or lower than the earlier case? (Given that 1 L bar = 100 J)
Ans. Work done in the process can be calculated as
w = -Pex (Vf -Vt)= -2 × 40 = -80 bar = -8 kJ
The negative sign shows that work is done by the system on the surroundings. Work done will be more in the reversible expansion because internal pressure and external pressure are almost same at every step.
Matching Answer Type Question
Q.51. In the following questions more than one correlation is possible between options of both columns.
Match the following :
| A | B |
| (i) Adiabatic process | (a) Heat |
| (ii) Isolated system | (b) At constant volume |
| (iii) Isothermal change | (c) First law of thermodynamics |
| (iv) Path function | (d) No exchange of energy and matter |
| (v) State function | (e) No transfer of heat |
| (vi) ∆U = q | (f) Constant temperature |
| (vii) Law of conservation of energy | (g) Internal energy |
| (viii) Reversible process | (h) pext = 0 |
| (ix) Free expansion | (i) At constant pressure |
| (x) ∆H = q | (j) Infinitely slow process which proceeds through a series of equilibrium states. |
| (xi) Intensive property | (k) Entropy |
| (xii) Extensive property | (l) Pressure |
| (m) Specific heat |
Ans. (i) → (e); (ii) → (d); (iii) → (f); (iv) → (a); (v) → (g), (k), (L); (vi) → (b); (vii) → (c); (viii) →(j); (ix) → (h); (x) → (i); (xi) → (a), (l), (m); (xii) → (g), (k)
Q.52. Match the following processes with entropy change:
| Reaction | Entropy change |
| (i) A liquid vapourises | (a) ∆S = 0 |
| (ii) Reaction is non-spontaneous at all temperatures and ∆H is positive | (b) ∆S = positive |
| (iii) Reversible expansion of an ideal gas | (c) ∆S = negative |
Ans.
| Reaction | Entropy change |
| (i) A liquid vapourises | When liquid changes into vapour, randomness of the molecules increases thus entropy increases. |
| (ii) Reaction is non-spontaneous at all temperatures and AH is positive. | When ΔH is positive, Energy factor opposes. ΔG is positive because the process is non-spontaneous so, ΔS =-ve |
| (iii) Reversible expansion of an ideal gas | The process remains in equilibrium at every stage. |
Q.53. Match the following parameters with description for spontaneity:
| ∆ (Parameters) ∆rHΘ ∆rSΘ ∆rGΘ | Description |
| (i) + – + | (a) Non-spontaneous at high temperature. |
| (ii) – – + at high T | (b) Spontaneous at all temperatures |
| (iii) – + – | (c) Non-spontaneous at all temperatures |
Ans. (i) → (c); (ii) → (a); (iii) → (b)
Solution.
| ∆ (Parameters) ∆rHΘ ∆rSΘ ∆rGΘ | Description |
| (i) + – + | (a) When enthalpy change is positive, ∆G is positive and ∆S is negative, then the process is non-spontaneous at all temperatures. |
| (ii) – – + at high T | (b) When enthalpy change is -ve and ∆G is +ve, then the process will be non-spontaneous at very high temperature. T ∆ S > ∆ H. |
| (iii) – + – | (c) When ∆ H = -ve, ∆S = +ve then ∆G = -ve and the process will be spontaneous at all temperatures. |
Q.54. Match the following :
| (i) Entropy of vapourisation | (a) decreases |
| (ii) K for spontaneous process | (b) is always positive |
| (iii) Crystalline solid state | (c) lowest entropy |
| (iv) ∆U in adiabatic expansion of ideal gas | (d) 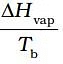 |
Ans. (i) → (b), (d); (ii) → (b); (iii) → (c); (iv) → (a)
Solution.
| Column I | Column II |
| (i) Entropy of vaporisation | ∆Hvap/Tb |
| (ii) K for spontaneous process | Since ∆rGΘ = ∆rHΘ - T ∆rSΘ = -RT |
| (iii) Crystalline solid state | Always has the lowest entropy because the molecules in solid crystal are in most ordered form. |
| (iv) ∆ U in adiabatic expansion of ideal gas | Since ∆U = q-w in case of expansion because work is done by the system and q = 0 and ∆U = -w |
ASSERTION AND REASON ANSWER TYPE QUESTION
Q.55. In the following questions a statement of Assertion (A) followed by a statement of Reason (R) is given. Choose the correct option out of the choices given below each question.
Assertion (A): Combustion of all organic compounds is an exothermic reaction.
Reason (R): The enthalpies of all elements in their standard state are zero.
(1) Both A and R are true and R is the correct explanation of A.
(2) Both A and R are true but R is not the correct explanation of A.
(3) A is true but R is false.
(4) A is false but R is true.
Ans. (2)
Solution.
In combustion reaction, enthalpy of the reactants is always greater than the enthalpy of the product.
Q.56. In the following questions a statement of Assertion (A) followed by a statement of Reason (R) is given. Choose the correct option out of the choices given below each question.
Assertion (A): Spontaneous process is an irreversible process and may be reversed by some external agency.
Reason (R): Decrease in enthalpy is a contributory factor for spontaneity.
(1) Both A and R are true and R is the correct explanation of A.
(2) Both A and R are true but R is not the correct explanation of A.
(3) A is true but R is false.
(4) A is false but R is true.
Ans. (2)
Solution.
For spontaneous process, energy factor should be favourable means ∆H = -ve and randomness should be positive.
Q.57. In the following questions a statement of Assertion (A) followed by a statement of Reason (R) is given. Choose the correct option out of the choices given below each question.
Assertion (A): A liquid crystallises into a solid and is accompanied by decrease in entropy.
Reason (R): In crystals, molecules organise in an ordered manner.
(1) Both A and R are true and R is the correct explanation of A.
(2) Both A and R are true but R is not the correct explanation of A.
(3) A is true but R is false.
(4) A is false but R is true.
Ans. (1)
Solution.
When a liquid crystallizes, its entropy decreases. Since in crystalline form, the molecules are more ordered.
LONG ANSWER TYPE QUESTION
Q.58. Derive the relationship between ∆H and ∆U for an ideal gas. Explain each term involved in the equation.
Ans. We know that the heat absorbed at constant volume is equal to change in the internal energy i.e., ∆U = qV. But most of chemical reactions are carried out not at constant volume, but in flasks or test tubes under constant atmospheric pressure ∆U = qp - p∆V at constant pressure, where qp is heat absorbed by the system and - p∆V represent expansion work done by the system.
Let us represent the initial state by subscript 1 and final state by 2.
We can rewrite the above equation as
U2 - U1 = p(V2 - V1)
On rearranging, we get
qp = (U2 + pV2) - (U1 + pV1) ....(1)
Now, we can define another thermodynamic function, the enthalpy H [Greek word enthalpie, to warm or heat content] as:
H = U + pV ...( 2)
So, equation (1) becomes
qp = H2 - H1 = AH
Although, q is a path dependent function, H is a state function because it depends on U, p and V, all of which are state functions. Therefore, ∆H is independent of path. Hence, qp is also independent of path.
For finite changes at constant pressure, we can write equation (2) as
∆H = ∆U + ∆pV
Since p is constant, we can write:
∆H = ∆U + p∆V ...(3)
It is important to note that when heat is absorbed by the system at constant pressure, we are actually measuring changes in the enthalpy.
Remember ∆H = qp, heat absorbed by the system at constant pressure.
∆H is negative for exothermic reactions which evolve heat during the reaction and ∆H is positive for endothermic reactions which absorb heat from the surroundings.
At constant volume (∆V = 0). ∆U = qv, therefore, equation (3) becomes
∆H = ∆U = qv
Let us consider a reaction involving gases. If VA is the total volume of the gaseous reactants, VB is the total volume of the gaseous products, nA is the number of moles of gaseous reactants and nB is the number of moles of gaseous products, all at constant pressure and temperature, then using the ideal gas law, we write,
pVA = nART
and pVB = nBRT
Thus, pVB - pVA = nBRT - nART = (nB - nA)RT
or p(VB -VA) = (nB - nA)RT
or p ∆V = ∆n gRT ... (4)
Here, ∆ng refers to the number of moles of gaseous products minus the number of moles of gaseous reactants.
Substituting the value of p∆V from equation (4) in equation (3), we get
∆H = ∆U + ∆ngRT ...(5)
Q.59. Extensive properties depend on the quantity of matter but intensive properties do not. Explain whether the following properties are extensive or intensive.
Mass, internal energy, pressure, heat capacity, molar heat capacity, density, mole fraction, specific heat, temperature and molarity.
Ans. In thermodynamics, a distinction is made between extensive properties and intensive properties. An extensive property is a property whose value depends on the quantity or size of matter present in the system. For example, the following are extensive properties: mass, volume, internal energy, enthalpy, heat capacity.
Those properties which do not depend on the quantity or size of matter present, are known as intensive properties.
For example, the following are intensive properties: temperature, density, pressure. A molar property, χm, is the value of an extensive property χ of the system for 1 mol of the substance. If n is the amount of matter, χm = χ/n , is independent of the amount of matter. The ratio of two extensive properties is always an intensive property.
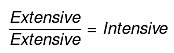 Example:
Example: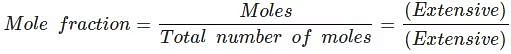
Q.60. Graphically show the total work done in an expansion when the state of an ideal gas is changed reversibly and isothermally from (pi , Vi) to (pf , Vf). With the help of a pV plot compare the work done in the above case with that carried out against a constant external pressure pf.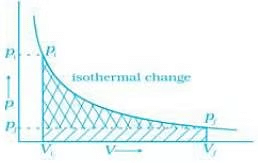 Ans. (i) Reversible work is represented by the combined areas
Ans. (i) Reversible work is represented by the combined areas and
and .(ii) Work against constant pressure, pf is represented by the area
.(ii) Work against constant pressure, pf is represented by the area  Work (i) > Work (ii).
Work (i) > Work (ii).
|
114 videos|263 docs|74 tests
|
FAQs on NCERT Exemplar: Thermodynamics - Chemistry Class 11 - NEET
| 1. What is the first law of thermodynamics? |  |
| 2. What is the difference between heat and temperature? |  |
| 3. How does a heat engine work? |  |
| 4. What is entropy in thermodynamics? |  |
| 5. What is the significance of the Carnot cycle in thermodynamics? |  |






















