Lakhmir Singh & Manjit Kaur: Carbon And Its Compounds, Solutions- 1 | Science Class 10 PDF Download
(Page No:220)
Question 1:
Name the element whose one of the allotropic forms is buckminsterfullerene.
Solution :
Carbon
Question 2:
What are the two properties of carbon which lead to the formation of a large number of carbon compounds ?
Solution :
Catenation(Self linking of carbon atoms to form long chains) and Tetravalency.
Question 3:
State whether the following statement is true or false :
Solution :
False
(Page No:221)
Question 4:
Name the scientist who disproved the ‘vital force theory’ for the formation of organic compounds.
Solution :
Friedrich Wohler.
Question 5:
Name the element whose allotropic form is graphite.
Solution :
Carbon.
Question 6:
In addition to some propane and ethane, LPG cylinders contain mainly two isomers of another alkane. Name the two isomers and write their condensed structural formulae.
Solution :
n-butane and iso-butane.
Question 7:
Buckminsterfullerene is a spherical molecule in which 60 carbon atoms are arranged in interlocking hexagonal and pentagonal rings of carbon atoms.
How many hexagons of carbon atoms are present in one molecule of buckminsterfullerene ?
How many pentagons of carbon atoms are present in one molecule of buckminsterfullerene ?
Solution :
(a) 20 hexagons
(b) 12 pentagons
Question 8:
Name the black substance of pencil. Will the current flow through the electrical circuit when we use the sharpened ends of the pencil to complete the circuit ?
Solution :
Graphite
Yes, current will flow through the circuit since graphite is a good conductor of electricity.
Question 9:
How does graphite act as a lubricant ?
Solution :
Graphite is used as a lubricant in the form of graphite powder or mixed with petroleum jelly or with any lubricant oil to form graphite grease.
Question 10:
Name the hardest natural substance known.
Solution :
Diamond
Question 11:
Which of the following molecule is called buckminsterfullerene ?
C90 C60 C70 C20
Solution :
C60 is called buckminsterfullerene
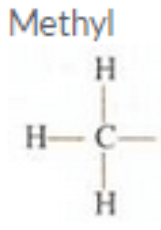
Question 12:
Give the name and structural formula of an alkyl group.
Solution :
Methyl
Question 13:
Write the electron-dot structures for : (i) ethane, (ii) ethene, and (iii) ethyne.
Solution :



Question 14:
Give the IUPAC name of the following compound :
C2H6
Solution :
Ethane.
Question 15:
Write the structural formula of propene.
Solution :

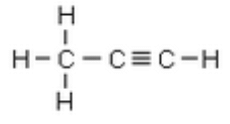
Question 16:
Write the structural formula of propyne.
Solution :
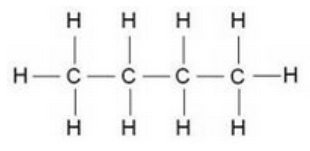
Question 17:
Write the structural formula of butane.
Solution :
Question 18:
What do you call the compounds having the same molecular formula but different structural arrangements of atoms ?
Solution :
Isomers.
Question 19:
Write the names of any two isomers represented by the molecular formula C5H12.
Solution :
Isopentane and neopentane.
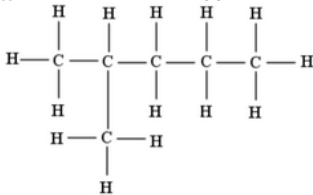
Question 20:
Write down (i) structural formula, and (ii) electron-dot formula, of any one isomer of hexane (C6H14), other than n-hexane.
Solution :
(i) Isomer of hexane: 2-methylpentane

Question 21:
Fill in the following blanks with suitable words :
(a) The form of carbon which is known as black lead is……………………..
(b) The form of carbon which is used as a lubricant at high temperature is……………………………
(c) Compounds of carbon with hydrogen alone are called…………………..
(d) CnH2n is the general formula of………………..
(e) Hydrocarbons having the general formula CnH2n-2 are called……………………..
(f) Ethene and ethyne are examples of…………….. hydrocarbons.
(g) Ethyne has…….. carbon-hydrogen single bonds.
(h) Carbon compounds have usually……………….. melting points and boiling points because they are……………… in nature.
(i) The property of carbon atoms to form long chains in compounds is called…………………………
(j) The general formula CnH2n for cycloalkanes is the same as that of……………………..
(k) The IUPAC name of ethylene is……………….
(l) The IUPAC name of acetylene is……………..
Solution :
(a) Graphite
(b) Graphite
(c) Hydrocarbons
(d) Alkene
(e) Alkynes
(f) Unsaturated
(g) Two
(h) Low; covalent
(i) Catenation
(j) Alkenes
(k) Ethene
(l) Ethyne
Question 22:
(a) What is the atomic number of carbon. Write its electronic configuration.
(b) What type of chemical bonds are formed by carbon ? Why ?
(c) Name the three allotropic forms of carbon.
Solution :
(a)The atomic number of carbon is 6. Its electronic configuration is 2,4.
(b)Carbon forms covalent bonds because it can achieve the inert gas electron arrangement only by sharing of electrons.
(c)Diamond, graphite and buckminsterfullerene.
Question 23:
(a) What is the general name of all the compounds made up of carbon and hydrogen ?
(b) Why does carbon form compounds mainly by covalent bonding ?
Solution :
(a) Hydrocarbons
(b) Carbon forms covalent bonds because it can achieve the inert gas electron
arrangement only by sharing of electrons.
Question 24:
(a) What is meant by catenation ? Name two elements which exhibit the property of catenation.
(b) Write the names and structural formulae of all the possible isomers of hexane.
Solution :
(a) The property of self combination of carbon atoms to form long chains is called catenation. Carbon and Silicon exhibit the property of catenation.





Question 25:
(a) What is buckminsterfullerene ? How is it related to diamond and graphite ?
(b) Why is diamond used for making cutting tools (like glass cutters) but graphite is not ?
(c) Why is graphite used for making dry cell electrodes but diamond is not ?
Solution :
(a) Buckminsterfullerene is an allotrope of carbon containing clusters of 60 carbon atoms joined together to form spherical molecules. It burns on heating to form carbon dioxide and nothing is left behind. This shows that it is made up of carbon only like diamond and graphite.
(b) Diamond used for making cutting tools but graphite is not because diamond is a very hard substance and graphite is a soft substance.
(c) Graphite is used for making dry cell electrodes but diamond is not b ecause graphite is a good conductor of electricity whereas diamond is a bad conductor of electricity.
(Page No:222)
Question 26:
(a) Give the general formula of an : (i) alkane (ii) alkene (in) alkyne.
(b) Classify the following compounds as alkanes, alkenes and alkynes :
C2H4, C3H4, C4H8, C5H12, C5H8, C3H8, C6H6
Solution :
(a) (i) CnH2n+2
(ii) CnH2n
(iii) CnH2n-2
Alkanes:
C5H12
C3H8
Alkenes:
C2H4
C4H8
Alkynes:
C3H4
C5H8
Question 27:
(a) Friedrich Wohler converted an inorganic compound into an organic compound in the laboratory.
(i) Give the name and formula of inorganic compound.
(ii) Write the name and formula of organic compound formed.
(b) Give the molecular formula of butane and mention the names of its two isomers. Name one fuel which contains both these isomers.
Solution :
(a) (i) Ammonium
cyanate, NH4CNO
(ii) Urea, CO(NH2)2
(b) The molecular
formula of butane is C4H10; Its isomers are n-butane and 2-methylpropane; LPG.
Question 28:
(a) Give IUPAC names and formulae of an organic compound containing single bonds and the other
containing a triple bond.
(b) Which of the following is the molecular formula of benzene ?
C6H6, C6H10, C6H12, C6H14
(c) Which of the two has a branched chain : isobutane or normal butane ?
Solution :
(a) Methane (single bond): CH4
Ethyne (triple bond): C2H2
(b) Benzene: C6H6
(c) Isobutene
Question 29:
Catenation is the ability of an atom to form bonds with other atoms of the same element. It is exhibited by both carbon and silicon. Compare the ability of catenation of the two elements. Give reasons.
Solution :
Carbon forms strong bonds among themselves and with other elements and this makes the carbon compounds stable whereas silicon shows catenation property due to which it forms compounds with hydrogen having chains of up to 7 or 8 silicon atoms; but due to weak bonds, these compounds are unstable.
Question 30:
(a) How can diamonds be made artificially ? How do synthetic diamonds differ from natural ones ?
(b) Give any two differences between the properties of diamond and graphite. What causes these differences ?
Solution :
(a)Diamonds can be made artificially by subjecting pure carbon to very high
pressure and temperature. The synthetic diamonds are small whereas natural
diamonds are big.
(b)(i) Diamond is hard whereas graphite is soft.
(ii)Diamond is a non-conductor of electricity whereas graphite is a good conductor of electricity. The difference in the physical properties of diamond and graphite arises because
of the different arrangements of carbon atoms in them.
Question 31:
(a) Why does the element carbon from a large number of carbon compounds ?
(b) Write down the structures and names of two isomers of butane (C4H10)
Solution :
(a) Carbon forms a large number of carbon compounds because carbon atoms can link with one another by means of covalent bonds to form long chains of carbon atoms.


Question 32:
(a) Give the name and structural formula of one member each of the following :
(i) alkane (ii) alkene (iii) alkyne (iv) cycloalkane
(b) Give the common name of (i) ethyne, and (ii) ethene.
(c) Write the molecular formula and structure of benzene.
Solution :



(b) (i) Acetylene
(ii) Ethylene
(c) C6H6;
Question 33:
(a) What is the unique property of carbon atom ? How is this property helpful to us ?
(b) Explain why, diamond is hard while graphite is soft (though both are made of carbon atoms).
Solution :
(a) The most unique property of carbon atom is its ability to combine itself, atom to atom to form long chains. This property of self combination is useful to us because it gives rise to an extremely large number of carbon compounds (or organic compounds).
(b) A diamond crystal is a giant molecule of carbon atoms. Each carbon atom in the diamond crystal is linked to four other carbon atoms by strong covalent bonds. The four surrounding atoms are at the four vertices of a regular tetrahedron. This rigid structure of diamond makes it a very hard substance.
The structure of graphite is very different from that of diamond. A graphite crystal consists of layers of carbon atoms or sheets of carbon atoms and these layers are held together by weak Van der Waals forces. Due to this sheet like structure, graphite is a comparatively soft substance.
Question 34:
(a) Giving their structures, state the number of single bonds, double bonds and triple bonds (if any) in the following compounds :
(i) ethyne (ii) ethene (iii) benzene
(b) Write the molecular formula and structure of cyclohexane. How many covalent bonds are there in a molecule of cyclohexane ?
Solution :
(a) (i) Ethyne:- Single bonds: Two; Triple bond: One
(ii) Ethene:- Single bonds: Four; Double bond: One
(iii) Ethane:- Single bonds: Nine; Double bonds: Three

Question 35:
(a) Write two points of difference in the structures of diamond and graphite.
(b) Explain why, graphite can be used as a lubricant but diamond cannot.
(c) Explain why, diamond can be used in rock drilling equipment but graphite cannot.
(d) State one use of diamond which depends on its ‘extraordinary brilliance’ and one use of graphite which depends on its being ‘black and quite soft’.
Solution :
(a) Diamond:
(i) Each carbon atom is linked to four other carbon atoms.
(ii) A diamond crystal has a tetrahedral arrangement of carbon atoms.
Graphite:
(i) Each carbon atom is joined to only three other carbon atoms.
(ii) A graphite crystal has flat hexagonal rings structure.
(b) Due to its softness, powdered graphite can be used a lubricant whereas diamond being extremely hard can not be used as lubricant.
(c) Due to its rigid structure, diamond is the hardest known substance to man. Hence, it is used in rock drilling equipments but graphite is soft and hence not used in rock drilling equipments.
(d) Diamonds are used for making jewellery.
Graphite is used for making pencil cores or ‘pencil leads’.
Question 36:
(a) What is diamond ? Of what substance is diamond made ?
(b) Describe the structure of diamond. Draw a simple diagram to show the arrangement of carbon atoms in diamond.
(c) Explain why, diamond has a high melting point.
(d) State any two uses of diamond.
Solution :
(a) Diamond is a colourless transparent substance having extraordinary brilliance. It is made up of carbon.
(b) A diamond crystal is a giant molecule of carbon atoms. Each carbon atom in the diamond crystal is linked to four other carbon atoms by strong covalent bonds. The four surrounding carbon atoms are at the four vertices of a regular tetrahedron. This rigid structure of diamond makes it a very hard substance.

(c) Diamond has a high melting point because a lot of heat energy is required to break the network of strong covalent bonds in the diamond crystal.
(d) (i) Used in rock drilling equipment (ii) Used in making jewellery.
Question 37:
(a) What is graphite ? Of what substance is graphite made ?
(b) Describe the structure of graphite with the help of a labelled diagram.
(c) Why is graphite a good conductor of electricity but diamond is a non-conductor of electricity ?
(d) State any two uses of graphite.
Solution :
(a) Graphite is a greyish-black opaque substance. It is made up of carbon.
(b) The structure of graphite is very different from that of diamond. A graphite crystal consists of layers of carbon atoms or sheets of carbon atoms. Each carbon atom in a graphite layer is joined to other three carbon atoms by strong covalent bonds to form flat hexagonal rings. The various layers of carbon atoms in graphite are held together by weak Van der Waals forces. Due to this sheet like structure, graphite is a comparatively soft substance.
(c) Due to the presence of free electrons in a graphite crystal, it conducts electricity however; a diamond crystal does not have free electrons so it does not conduct electricity.
(d) (i) Used as a lubricant (ii) Used for making pencil leads.
Question 38:
(a) Explain the term ‘isomers’. Give one example of isomers.
(b) Write (i) structural formula, and (ii) electron-dot structure, of any one isomer of n-heptane (C7H16)
(c) Write IUPAC name of the compound having the formula n-C4H10.
(d) Give the IUPAC names for the following :
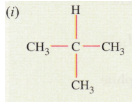
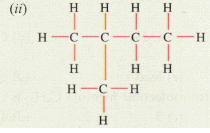
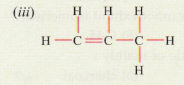
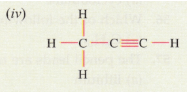
Solution :
(a) The organic compounds having the same molecular formula but different structures are known as isomers for ex: n-butane and iso-butane are isomers.
(b) Isomer of n-heptane:2-methylhexane
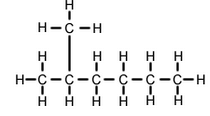
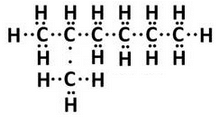
(c) Butane
(d) (i) 2-methylpropane
(ii) 2-methylbutane
(iii) Propene
(iv) Propyne
|
85 videos|437 docs|75 tests
|
FAQs on Lakhmir Singh & Manjit Kaur: Carbon And Its Compounds, Solutions- 1 - Science Class 10
| 1. What are some examples of carbon compounds? |  |
| 2. How is carbon dioxide formed? |  |
| 3. What is the difference between organic and inorganic compounds? |  |
| 4. How does carbon form multiple bonds with other atoms? |  |
| 5. What is the significance of carbon compounds in our daily lives? |  |

|
Explore Courses for Class 10 exam
|

|


















