Theory & Procedure, Newton Law of Cooling | Additional Study Material for NEET PDF Download
To study the relationship between the temperature of a hot body and its time of cooling by plotting a cooling curve.
Theory
This Law of Cooling is named after the famous English Physicist Sir Isaac Newton, who conducted the first experiments on the nature of cooling.
Statement of the Law :
According to Newton’s Law of Cooling, the rate of cooling of a body is directly proportional to the difference in temperatures of the body (T) and the surrounding (T0), provided difference in temperature should not exceed by 300C.
From the above statement,
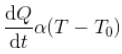
For a body of mass m, specific heat s, and temperature T kept in surrounding of temperature T0;
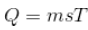
Now, the rate of cooling,
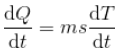
Hence ,
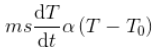
Since the mass and the specific heat of the body are taken as constants, the rate of change of temperature with time can be written as,
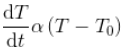
The above equation explains that, as the time increases, the difference in temperatures of the body and surroundings decreases and hence, the rate of fall of temperature also decreases.
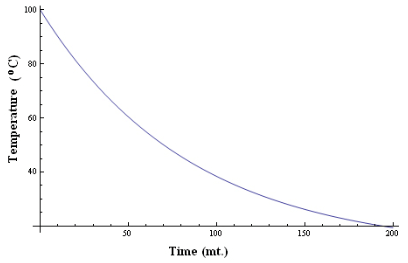
It can be graphically represented as,
 Learning Outcomes
Learning Outcomes
- Students understand the different modes of transfer of heat.
- Students identify the variables which affect the cooling rate of a substance.
- Students verify Newton’s Law of Cooling.
- Students understand the relationship between temperature and time of cooling of objects.
Materials Required:
- Copper calorimeter
- Stirrer
- Wooden box with a clamp and stand
- A wooden lid having a hole in the middle.
- Thermometer
- Stop watch
- Hot water of about 80 °C.
Real Lab Procedure:
- Fill about 2/3rd of the copper calorimeter containing stirrer with hot water of about 80 °C.
- Place the calorimeter inside the wooden box. The space between the wooden box and calorimeter is filled with cotton to avoid heat loss.
- Close the wooden box with its lid.
- Suspend the thermometer inside the hot water in the calorimeter from the clamp and stand.
- Stir water continuously to make it cool uniformly.
- When the temperature of hot water falls to 70°C, start the stop watch.
- Note the temperature reading at every five minutes.
- Continue the time temperature observation till the temperature becomes constant.
- Plot a graph between time along X-axis and temperature along Y-axis. This graph is called the cooling curve.
- The graph is an exponential curve and it shows that the temperature falls quickly at the beginning and then slowly as the difference of temperature goes on decreasing. This verifies the Newton’s Law of cooling.
Simulator Procedure (as performed through the Online Labs):
- Select the material of the calorimeter from the drop down list.
- Select the radius of the calorimeter using the slider.
- Select the mass of the calorimeter using the slider.
- Select the liquid sample from the drop down list.
- Select the temperature of the preheated liquid using the slider.
- Select the room temperature using the slider.
- Mass of the liquid is fixed as 250 g.
- Click on the ‘START’ button on the timer to start/stop the experiment.
- Click on the ‘Show cross section’ button to view the cross section area.
- Click on the ‘Show graph’ button to view the graph.
- To redo the experiment, click on the ‘Reset’ button.
Observations:
Sl. No. | Time for cooling, t (mt.) | Temperature of water in calorimeter, T(oC) | Temperature of water in enclosure, T0 (oC) | Difference of temperature, (T-T0) (oC) |
1 | 0 | 70 | ||
2 | 1 | |||
3 | 2 | |||
4 | 3 | |||
5 | 4 | |||
6 | 5 | |||
7 | 6 | |||
8 | 7 | |||
9 | 8 |
Calculations:
- Temperature of water in the enclosure is found to be a constant. If not, then take its mean as T0.
- From the observations, we can calculate the temperature difference (T-T0).
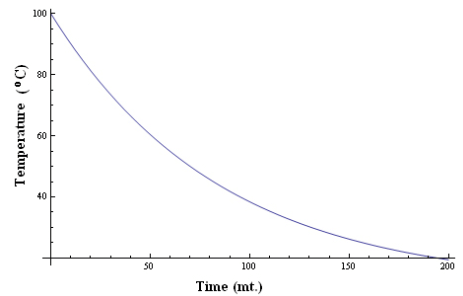
- Now, plot a graph between time (t) and temperature (T), taking time along X axis and temperature along Y axis.
Results:
The cooling curve of the liquid is plotted.
The temperature falls quickly in the beginning and then slowly as the difference of temperature goes on decreasing.
This is in agreement with Newton’s Law of cooling.
|
26 videos|312 docs|64 tests
|
FAQs on Theory & Procedure, Newton Law of Cooling - Additional Study Material for NEET
| 1. What is Newton's law of cooling? |  |
| 2. How can Newton's law of cooling be applied in practical situations? |  |
| 3. What is the procedure to calculate the temperature of an object using Newton's law of cooling? |  |
| 4. Can Newton's law of cooling be used for objects undergoing phase changes? |  |
| 5. How does Newton's law of cooling relate to the concept of thermal equilibrium? |  |

















