Theory & Procedure, Surface Tension | Additional Study Material for NEET PDF Download
Objective
To determine the surface tension of a liquid by capillary rise method.
Theory
How do you define surface tension?
Surface tension is the property of a liquid, by virtue of which its free surface at rest behaves like an elastic skin or a stretched rubber membrane, with a tendency to contract so as to occupy minimum surface area. This property is caused by cohesion of molecules and is responsible for much of the behaviors of liquids.
The property of surface tension is revealed, for example, by the ability of some objects to float on the surface of water, even though they are denser than water. Surface tension is also seen in the ability of some insects, such as water striders, and even reptiles like basilisk, to run on the water’s surface.

The Theory Behind Surface Tension
Surface tension has been well- explained by the molecular theory of matter. According to this theory, cohesive forces among liquid molecules are responsible for the phenomenon of surface tension. The molecules well inside the liquid are attracted equally in all directions by the other molecules. The molecules on the surface experience an inward pull.
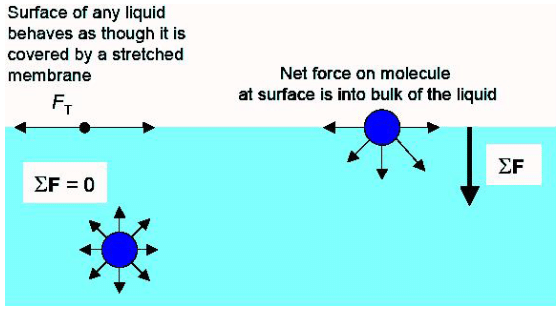
So, a network is formed against the inward pull, in order to move a molecule to the liquid surface. It results in a greater potential energy on surface molecules. In order to attain minimum potential energy and hence stable equilibrium, the free surface of the liquid tends to have the minimum surface area and thereby it behaves like a stretched membrane.
Surface tension is measured as the force acting normally per unit length on an imaginary line drawn on the free liquid surface at rest. It is represented by the symbol T (or S). It's S.I. The unit is Nm-1 and dimensional formula is M1L0T-2.
Capillarity in Liquids
When a capillary tube is dipped in a liquid, the liquid level either rises or falls in the capillary tube. The phenomena of rise or fall of a liquid level in a capillary tube is called capillarity or capillary action.
How do we define the surface tension of a liquid through the capillary rise method?
When a liquid rises in a capillary tube, the weight of the column of the liquid of density ρ inside the tube is supported by the upward force of surface tension acting around the circumference of the points of contact.
Thus, surface tension;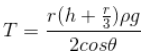
Where, h - height of the liquid column above the liquid meniscus
ρ - Density of the liquid
r - Inner radius of the capillary tube
θ - Angle of contact
Learning Outcomes
- Students understand the theory of the surface tension of liquids.
- Students correlate the property of surface tension with different natural phenomena.
- Students understand the concept of capillarity in liquids.
- They are able to relate surface tension and capillarity.
Materials required
- A clean and dry capillary tube
- A tipped pointer
- A beaker containing water
- A travelling microscope
- Adjustable wooden stand
- Clamps and stand
Real lab Procedure
To set up the apparatus :
- Place the adjustable height stand on the table and make its base horizontal by leveling the screws.
- Fix the capillary tube and the pointer in a cork, and clamp it in a rigid stand so that the capillary tubes and the pointer become vertical.
- Adjust the height of the vertical stand, so that the capillary tubes dip in the water in an open beaker.
- Adjust the position of the pointer, such that its tip just touches the water surface.
To find the capillary rise :
- Find the least count of the travelling microscope for the horizontal and the vertical scale.
- Make the axis of the microscope horizontal.
- Adjust the height of the microscope using the height adjusting screw.
- Bring the microscope in front of the capillary tube and clamp it when the capillary rise becomes visible.
- Make the horizontal cross wire just touch the central part of the concave meniscus.
- Note the reading of the position of the microscope on the vertical scale.
- Now, carefully remove the beaker containing water
- Move the microscope horizontally and bring it in front of the pointer.
- Lower the microscope and make the horizontal cross wire touch the tip of the pointer.
- Corresponding vertical scale readings are noted.
- The difference in the two readings (i.e., height of water meniscus and height of the tip of pointer) will give the capillary rise of the given liquid.
- We can repeat the experiment by changing the height of the wooden stand.
To find the internal diameter of the capillary tube :
- Place the capillary tube horizontally on the adjustable stand.
- Focus the microscope on the end dipped in water.
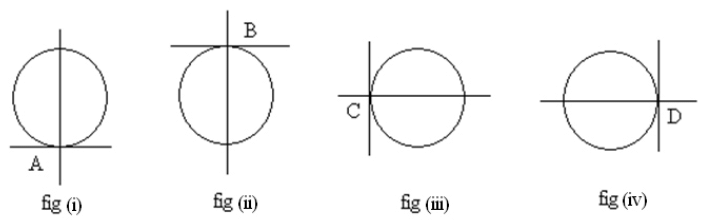
- Make the horizontal cross- wire touch the inner circle at A (fig i). Note microscope reading on the vertical scale.
- Raise the microscope to make the horizontal cross wire touch the circle at B (fig ii). Note the vertical scale reading.
- The difference between the two readings will give the vertical internal diameter (AB) of the tube.
- Move the microscope on the horizontal scale and make the vertical cross wire touch the inner circle at C (fig iii). Note microscope reading on the horizontal scale.
- Move the microscope to the right to make the vertical cross wire touch the circle at D (fig iv). Note the horizontal scale reading.
- The difference between the two readings will give the horizontal internal diameter (CD) of the tube.
- We can calculate the diameter of the tube by calculating the mean of the vertical and horizontal internal diameters. Half of the diameter will give the radius of the capillary tube.
Simulator Procedure (As performed through Online Labs)
- Select the liquid for which the coefficient of viscosity is to be measured, from the 'Select Solution' drop down list.
- Select the environment to perform the experiment from the 'Select Environment' drop down list.
- Use the ‘Select Temperature’ slider to change the temperature of the liquid.
- Use the ‘Capillary tube diameter’ slider to change the diameter of the capillary tube.
- Use the ‘Height of wooden stand slider to change the height of the wooden stand.
- Use the ‘Level of retort stand’ slider to change the height of the retort stand.
- Use the ‘Focus of microscope’ slider to focus the microscope.
- Use the arrows on the focusing screw also to focus the microscope.
- Use the ‘Height of microscope’ slider to change the height of the microscope.
- Use the arrows on the fine adjusting screw also to adjust the height of the microscope.
- Click on the beaker to see the zoomed view of that portion, to see whether the pointer tip just touches water level.
- Use the ‘Remove beaker’ button to remove the beaker.
- Click on the microscope to see the zoomed view of the vertical scale. Drag the scale to take the main scale and vernier scale readings.
- Now, calculations are done as per the real lab procedure and the surface tension of the selected liquid can be found out.
- Click on the ‘Reset’ button to redo the experiment.
|
26 videos|312 docs|64 tests
|
















