Isomerism & Nomenclature of Coordination Compounds
What is Isomerism?
Isomerism is the phenomenon in which two or more compounds have the same chemical formula but different structural or spatial arrangements of atoms, and therefore different physical and chemical properties. Compounds that exhibit this phenomenon are called isomers. Coordination compounds commonly show two broad categories of isomerism: structural (constitutional) isomerism and stereoisomerism.
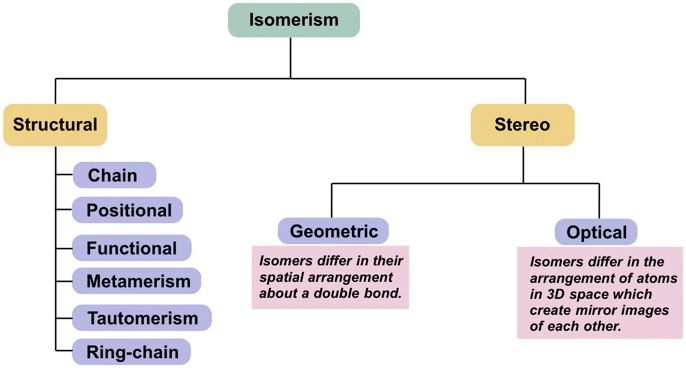
Types of Isomerism
Structural Isomerism
- Ionisation isomerism- These isomers arise when an anionic ligand inside the coordination sphere exchanges places with an anion outside the coordination sphere (i.e., between complex ion and counter-ion). Such isomers give different ions in solution and hence different chemical tests.
Example: [Co(NH3)5Br]SO4 and [Co(NH3)5SO4]Br. The first contains free SO42- in solution (gives a BaSO4 precipitate with BaCl2), while the second gives a precipitate with AgNO3 (free Br- present).
Other examples: [Pt(NH3)4Cl2]Br2 and [Pt(NH3)4Br2]Cl2.
- Hydrate (solvate) isomerism- These isomers differ by the number of water (or solvent) molecules coordinated to the metal and those present as water of crystallisation (outside the coordination sphere). Conductivity and chemical tests on solutions often distinguish them.
Example: Chromium(III) chloride with six waters, CrCl3·6H2O, has isomers such as:
[Cr(H2O)6]Cl3
[Cr(H2O)5Cl]Cl2·H2O
[Cr(H2O)4Cl2]Cl·2H2O
- Linkage (ambidentate) isomerism- Occurs when an ambidentate ligand (a ligand that can bind through two different atoms) binds to the metal through one atom in one isomer and through another atom in the other isomer.
Example: The nitrite ion, NO2-, can bind through N or O giving:
[Co(NH3)5ONO]Cl2 (pentaammine-nitrito-O-cobalt(III) chloride)
[Co(NH3)5NO2]Cl2 (pentaammine-nitro-N-cobalt(III) chloride)
Another classical example: [Mn(CO)5(SCN)]+ versus [Mn(CO)5(NCS)]+, where SCN binds through S in the first and through N in the second.
MULTIPLE CHOICE QUESTIONTry yourself: Linkage isomerism is seen in compounds having ________ ligand.
- Coordination (ion-exchange) isomerism- When both cationic and anionic species are coordination complexes, interchange of ligands between them produces coordination isomers.
Example: [{Pt(NH3)4}{PtCl4}] and [{Pt(NH3)3Cl}{PtCl3(NH3)}] represent different arrangements of the same set of ligands divided between cationic and anionic complex ions.
- Coordination position isomerism- In polynuclear or polyligated complexes, the position of a particular ligand may change from one metal centre to another, producing isomers.
- Polymerisation isomerism (formula isomerism) - Compounds having the same empirical formula but different molecular formulas (hence not true isomers in the strict structural sense) are sometimes referred to under this heading. For example, different platinum species such as Pt(NH3)2Cl2, Pt(NH3)4, PtCl4, etc., may have the same empirical formula but different molecular formulas and structures.
Stereoisomerism
Stereoisomerism arises when ligands are attached to the central metal ion in the same sequence (same bonds) but differ in their three-dimensional arrangements.
1. Geometrical Isomerism
Geometrical isomers have ligands arranged differently in space relative to each other (commonly classified as cis and trans forms). Geometrical isomerism depends on coordination number and geometry of the complex.
Coordination number 4: Tetrahedral (sp3) complexes generally do not show geometrical isomerism because all positions are equivalent. Square planar (dsp2) complexes commonly show geometrical isomerism.
Example: [Pt(NH3)2Cl2] exists as cis- and trans-isomers.
Example: [Pt(Gly)2] (glycine as monodentate/ambidentate representation) can also show geometric forms:
Coordination number 6: Octahedral complexes can show several geometrical isomers depending on ligand types and counts.
Example: [Co(NH3)4Cl2]+ can exist as cis and trans isomers.
Example: [Pt(NH3)2Cl2Br2] can show several geometric arrangements.
Try yourself: How many geometrical isomers are possible in [Al(C2O4)3]3-?
2. Optical Isomerism
Optical isomers (enantiomers) are non-superimposable mirror images of each other and rotate plane-polarised light in opposite directions; they are called dextrorotatory (d or +) and levorotatory (l or -). Optical isomerism is common in octahedral complexes with chelating (bidentate) ligands that create an asymmetric environment.
Example: [Co(en)2Cl2]+ has cis and trans forms; the cis form can exhibit optical isomerism (a pair of enantiomers), while the trans form is typically achiral.
Example: [Co(en)3]3+ is a classic optically active complex that exists as two enantiomers (Δ and Λ).
Try yourself: Which type of isomerism exhibits compounds with same chemical formula and bonds but different spatial arrangement?

Why do we need to name coordination compounds?
Systematic names give an unambiguous way to represent and communicate the composition and structure of coordination compounds. Correct nomenclature is essential for clear identification, especially when dealing with isomers that have the same formula but different structures or arrangements.
Basic rules of IUPAC nomenclature for coordination compounds
The following summary lists the main practical rules used for naming coordination compounds.
- Within a coordination entity (a complex ion or neutral complex), list the ligands first and the central metal atom/ion last.
- List ligands in alphabetical order when naming the complex entity (the prefixes di, tri, etc., used to indicate multiplicity are not considered for alphabetic order; however, prefixes such as bis, tris, tetrakis that are part of complex ligand names are ignored for alphabetic ordering except when they are part of the ligand name inside parentheses).
- Write the formula of the coordination entity in square brackets.
- When using ligand names that contain numerals or complex groups, enclose the ligand name in parentheses and use multiplicative prefixes such as bis, tris, tetrakis, etc., as required.
- No space should be left between ligands and the metal within the square brackets.
- The overall charges of ionic species must balance: the cation is named before the anion.
Detailed rules with examples
- Order of naming ions:Name the cation before the anion (this applies even if the complex is the anion).
Example: Na[Co(NH3)4Cl2] should be named with the cation first: Sodium tetraamminedichlorocobalt(II) (oxidation state depends on overall charge)-the exact oxidation state must be determined from ligand charges and overall charge.
Example: [Co(NH3)4Cl2]SO4 is named by giving the complex cation name first followed by the anion: tetraamminedichlorocobalt(III) sulphate (the oxidation state shown in parentheses corresponds to the metal in the complex).
- Alphabetical order of ligands: When different types of ligands are present, list ligand names in alphabetical order (ignoring multiplicative prefixes such as di-, tri- for this ordering). Anionic ligands end with the suffix -o, neutral ligands use their usual names (with special accepted names for some common ligands).
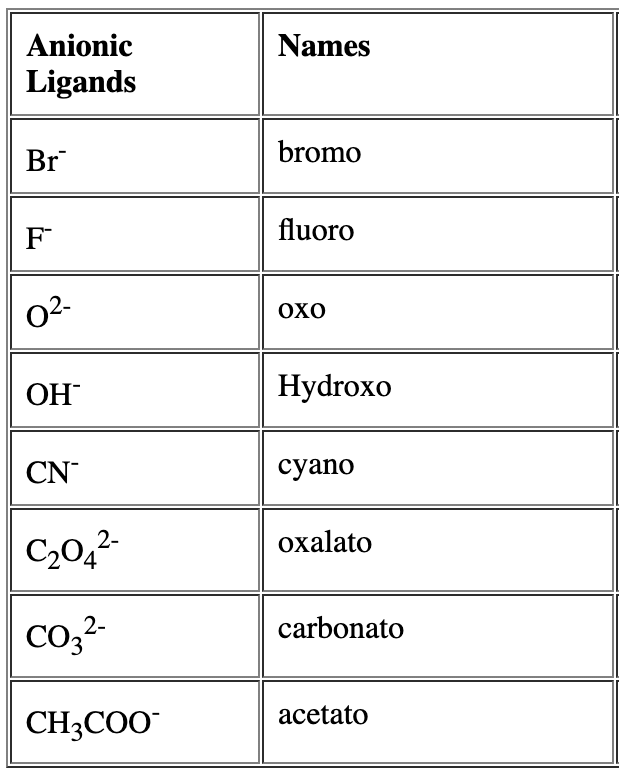

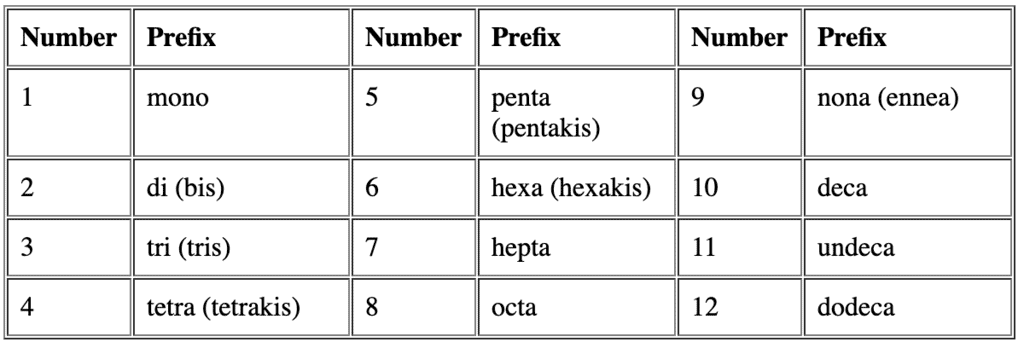
- Multiplicative prefixes: Use di, tri, tetra, penta, hexa for simple ligands and bis, tris, tetrakisfor ligands whose names already contain numbers or are polydentate or complex (enclose the ligand name in parentheses when using bis/tris/tetrakis).
Example: [NiCl2(PPh3)2] is named dichloridobis(triphenylphosphine)nickel(II).
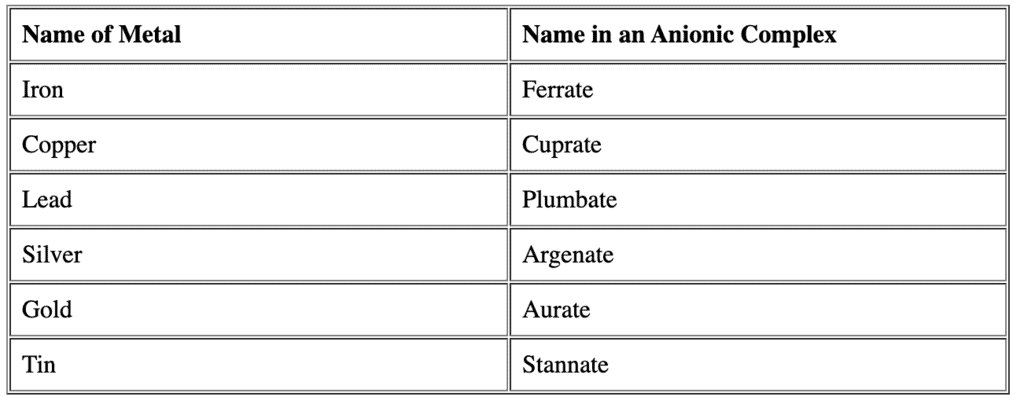
- Metal name and charge indication:After listing ligands in alphabetical order, give the name of the central metal.
If the complex ion is cationic, name the metal as the element (e.g., cobalt, iron).
If the complex ion is anionic, the metal name takes the suffix -ate and sometimes the Latin name of the metal is used (e.g., ferrate for iron, cuprate for copper). For many first-row transition metals, the English name plus -ate (e.g., cobalt → cobaltate) is used.
- Oxidation state: State the oxidation number (oxidation state) of the central metal in Roman numerals in parentheses immediately after the name of the metal.
- Neutral complexes: Name neutral complexes in the same way as complex cations (ligands first in alphabetical order, then the metal name and oxidation state if required).
- Ligands that can bind through different atoms (linkage isomers):Use prefixes to indicate the donor atom when necessary:
M-NO2 → nitro (bound through N)
M-ONO → nitrito (bound through O)
M-SCN → thiocyanato-S (bound through S) or simply thiocyanato when context is clear
M-NCS → isothiocyanato-N (bound through N)
- Solvates and hydrates:If solvent molecules or waters of crystallisation are present outside the coordination sphere, list these after naming the ionic components. Indicate the number of such molecules using Arabic numerals.
Example: KAl(SO4)2·12H2O is commonly named aluminium potassium sulphate 12-water (or aluminium potassium sulfate dodecahydrate).
(a) Complex cations IUPAC name (b) Complex anions (c) Organic groups (d) Bridging groups (e) Hydrates KAl(SO4)2·12H2O
Aluminium potassium sulphate 12-water - Writing the formula of a coordination compound:Enclose the coordination entity in square brackets. In the formula of the coordination entity, list the metal first followed by coordinated groups in the order: anionic ligands, neutral ligands, cationic ligands. Within each group, arrange ligands alphabetically by the first letter of the ligand name (ignoring multiplicative prefixes for ordering).
[M - negative ligands, neutral ligands, positive ligands]
n±
Summary
Coordination compounds can exhibit many types of isomerism; recognising the difference between structural and stereoisomerism is essential. Proper IUPAC naming requires a systematic order: name the cation before the anion, list ligands alphabetically (with appropriate ligand suffixes and multiplicative prefixes), name the metal (use -ate for anionic complexes where applicable), and indicate the metal oxidation state in Roman numerals. Mastery of these rules helps to distinguish and communicate isomers unambiguously.
FAQs on Isomerism & Nomenclature of Coordination Compounds
| 1. What is isomerism? |  |
| 2. What are the types of isomerism? |  |
| 3. Why do we need to name compounds? |  |
| 4. What is the relationship between isomerism and the nomenclature of coordination compounds? |  |
| 5. What are some frequently asked questions about isomerism and nomenclature of coordination compounds? |  |






