Alkenes: Nomenclature, Properties & Preparation
Alkenes
1. Introduction
Alkenes are hydrocarbons that contain at least one carbon-carbon double bond (C=C). They are also called olefins, a term historically derived from "oil-forming gas". Alkenes are important both in nature and industry. For example, ethylene (ethene) is the largest-volume industrial organic compound and is the monomer for polyethylene, used widely to make plastics and other consumer materials.
2. Structure and bonding in alkenes
- General formula: For an acyclic alkene with one double bond the general formula is CnH2n.
- Unsaturation: Alkenes are unsaturated hydrocarbons because the double bond contains π electrons in addition to σ bonds.
- Bond lengths and energies: In ethene the C=C bond length is about 1.34 Å and its bond energy is ≈ 146 kcal mol-1.
- Hybridisation and geometry: Each alkenic carbon in a C=C is sp2 hybridised. The three sp2 orbitals lie in one plane, forming σ bonds; the unhybridised p orbitals on the two carbons overlap side-by-side to form the π bond. The result is a planar arrangement about each double bonded carbon and restricted rotation about the C=C.
- π electron cloud: The π electrons are delocalised above and below the plane of the σ-bonded skeleton, producing the characteristic reactivity of the double bond.
- Isomerism and types of polyenes: Compounds with multiple double bonds can exist as conjugated polyenes (alternating single and double bonds), cumulated polyenes (adjacent double bonds), or isolated polyenes (double bonds separated by two or more single bonds).
- VSEPR and bond angles: Bond angles around sp2 carbons are ≈120°. When comparing an angle adjacent to a double bond with one adjacent to a single bond, repulsion due to the π electrons increases the angle beside the double bond (so angle a > b in common textbook diagrams).
IUPAC nomenclature of alkenes
- Select the longest carbon chain that contains the carbon-carbon double bond; that chain is the parent hydrocarbon.
- Replace the suffix 'ane' of the corresponding alkane by 'ene'. If there are two or more double bonds use 'diene', 'triene', etc.
- Number the parent chain so as to give the double bond the lowest possible locant; indicate the position by the number placed before the suffix (for example, 1-butene).
- If numbering from either end gives the same locant for the double bond, number so that substituents get the lowest possible set of locants.
- For multiple double bonds follow the lowest sum rule for locants.
- Name and locate substituents as prefixes, using alphabetical order (ignore multiplicative prefixes like di, tri for ordering).
Ex. 1 Write IUPAC names of:
(a)
Ans. (a) 2,3-Dimethylcyclohexene
(b) 1-(2-butenyl)cyclohex-1-ene
Ex. 2 Give the structure for each of the following:
(a) 4-Methyl-1,3-hexadiene
(b) 1-Isopropenylcyclopentene
Ans. (a)
3. Physical properties of alkenes
| Property | General trend in homologous series | Remarks / Isomeric effect |
|---|---|---|
| Physical state | C1-C3: gases; C4-C20: liquids; >C20: solids | - |
| Dipole moment | Depends on substituents | cis isomers usually have larger dipole moments than trans |
| Polarity | Generally nonpolar hydrocarbon | cis > trans for some C=C substitution patterns (cis more polar) |
| Melting point | Tends to change irregularly with chain length | trans > cis because trans isomers pack better |
| Boiling point | Increases with molecular mass in a homologous series | cis > trans (cis is more polar); branching lowers b.p. |
| Solubility | Practically insoluble in water; soluble in non-polar solvents (benzene, ether) | Polarity increases solubility in more polar solvents; cis > trans |
| Stability | - | trans isomers generally more stable than cis (less steric strain) |
Polarity increases → boiling point increases.
4. Laboratory test for alkenes
Alkenes give characteristic tests: they decolourise bromine in CCl4 (bromine test) and react with KMnO4 (cold, dilute) to give diols, decolourising purple KMnO4. These are commonly used qualitative tests for unsaturation.
5. Methods of preparation of alkenes
(i) Partial reduction of alkynes
Conversion of an alkyne to an alkene can be achieved selectively to give either the cis (syn) alkene or the trans (anti) alkene.
(a) Catalytic hydrogenation in presence of a poisoned catalyst (Lindlar hydrogenation)
This gives cis alkenes by syn addition of H2. Typical catalysts:
- Lindlar's catalyst: Pd deposited on CaCO3 poisoned with lead acetate and quinoline.
- P-2 catalyst (Ni2B): nickel boride.
General reaction: R-C≡C-R + H2 (poisoned catalyst) → R-CH=CH-R
Mechanism (summary): The alkyne and H2 are chemisorbed on the metal surface. Hydrogen atoms add from the same face of the π system to give the syn (cis) alkene. Quinoline partially blocks the catalyst surface preventing further hydrogenation to the alkane; it thus acts as a catalyst poison.
Example:
(b) Birch reduction (Na (or Li, K) in liquid NH3)
Gives trans alkenes (anti addition of hydrogen) via solvated electrons and protonation steps.
General reagent: Na / liq NH3 (followed by proton source)
Mechanistic steps involve formation of a radical anion and subsequent protonation steps. Note that terminal alkynes are problematic with this reagent because they can give sodium alkynide salts.
Note: Terminal alkynes (R-C≡CH) can form sodium salts under these conditions and thus Birch reduction is unsuitable.
Ex. 3 Identify the reagent for following synthesis.
Ans. H2 / Lindlar's catalyst.
Ex. 4 Identify the products in the following reaction :
Ans.
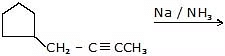
(ii) Dehalogenation of vicinal dihalides
Vicinal dihalides (halogens on adjacent carbons) can be converted to alkenes by elimination of the halogen atoms. Common reagents include Zn/AcOH, Zn/EtOH or NaI in acetone (Finkelstein conditions followed by elimination).
General: R-CHBr-CHBr-R → R-CH=CH-R
Example:
CH3-CHBr-CH2Br → CH3-CH=CH2
Mechanism with NaI in acetone involves anti elimination (E2) of halide ions.
Remarks:
- Both reactions are E2 type eliminations.
- They are stereospecific anti eliminations.
(iii) Dehydrohalogenation of alkyl halides
Removal of HX from an alkyl halide gives an alkene. This can proceed by E2 (one-step) or E1 (two-step, via carbocation) mechanisms depending on substrate and conditions. Common bases/reagents:
- Hot alcoholic KOH (or EtO-/EtOH) - common for E2 eliminations.
- NaNH2 (strong base) - used for forming alkynes or deprotonations.
- t-BuO-/t-BuOH - bulky base that often gives Hofmann (less substituted) product.
E2 mechanism (summary): A strong base abstracts a β-hydrogen while the leaving group leaves simultaneously. The reaction is stereospecific, often requiring anti-coplanar arrangement of the H and leaving group.
Example obeying Zaitsev (Saytzeff) rule: the more substituted (more stable) alkene is favoured.
Anti elimination stereospecificity is important for predicting stereochemistry of product alkenes.
Hofmann product
Bulky bases (for example t-BuO-) tend to abstract the least hindered proton and thus give the less substituted (Hofmann) alkene as the major product.
Ex. 5 What alkyl halide would yield each of the following pure alkene on reaction with alcoholic KOH?
(i)
(ii) CH3-CH2-CH2-CH=CH2
Ans.
(i)
(ii) CH3CH2CH2CH2CH2Cl
(iii)
Ex. 6 What are the various products due to loss of HBr from
Ans.
(iv) Dehydration of alcohols
Alcohols undergo β-elimination (loss of H2O) on heating with acid catalysts to give alkenes. Typical reagents and conditions:
- Conc. H2SO4, Δ (≈160°C)
- H3PO4, Δ
- P2O5, Δ
- Al2O3, 350°C (industrial dehydration)
General reaction: RCH(OH)-CH2R → R-CH=CH-R + H2O
Example:
(v) Pyrolysis of esters
Thermal decomposition (pyrolysis) of certain esters proceeds via a six-membered cyclic transition state to give an alkene and a carboxylic acid derivative. Because the transition state is cyclic, elimination occurs syn with both groups eliminated from the same face.
This syn elimination gives stereochemical control where applicable.
(vi) Hofmann elimination (quaternary ammonium bases)
Heating quaternary ammonium hydroxides under reduced pressure (100-200°C) gives alkenes. This elimination typically gives the less substituted (Hofmann) alkene as major product due to steric and leaving group effects.
(vii) Wittig reaction
Aldehydes and ketones react with phosphorus ylides (Wittig reagents, phosphoranes) to produce alkenes. The reaction proceeds via a cyclic oxaphosphetane intermediate and yields an alkene plus triphenylphosphine oxide.
(R, R', R" and R"' may be H or alkyl groups.)
Ex. 7 Complete the following reaction :
Ans.
Ex. 8 Identify (X), (Y), and (Z) in the following reactions
(i) PhCH2Br CH3 -
(ii) CH3I PhCOCH3
(iii) PhCH2Br PhCH=CHCHO
Ans. (X) = Ph-CH=C(CH3)2
(Y) = Ph-C(CH3)=CH2
(Z) = Ph-CH=CH-CH=CH-Ph
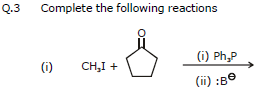
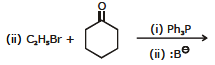
6. Chemical reactions of alkenes
(I) Catalytic hydrogenation of alkenes (heterogeneous hydrogenation)
Hydrogenation of an alkene (addition of H2 across C=C) is generally exothermic (ΔH° ≈ -120 kJ mol-1) and proceeds on metal catalysts such as Pt, Pd or Ni. Addition is usually syn (both hydrogens add from the same face) when occurring on a metal surface.
R-CH=CH-R + H2 → R-CH2-CH2-R
Example:
Ex. 9 Complete the following reactions :
CH3CH=CH2 + H2
Sol.
CH3CH=CH2 + H2
CH3CH2CH3
(II) Electrophilic addition reactions
Mechanism (general)
Step 1: An electrophile attacks the π bond to form a carbocation (or a bridged intermediate such as a halonium ion).
Step 2: A nucleophile attacks the carbocation (or opens the bridged intermediate) to give the addition product.
(i) Acid-catalysed hydration of alkenes
Alkenes add water in the presence of acid to yield alcohols following Markovnikov's rule (the proton adds to give the more stable carbocation). The reaction is reversible and carbocation rearrangements can occur.
Mechanism (summary):
Step 1: Protonation of the double bond to form a carbocation.
Step 2: Nucleophilic attack by water on the carbocation.
Step 3: Deprotonation to give the alcohol.
Ex. 10 Identify the product in following reaction:
Ans.
(ii)(a) Oxymercuration-demercuration
Oxymercuration adds H and OH across an alkene in a Markovnikov manner without rearrangement. The sequence uses Hg(OAc)2 followed by NaBH4 (demercuration).
In the oxymercuration step, an organomercurial intermediate is formed; NaBH4 later replaces the -HgOAc group with H, giving the alcohol with no rearrangement.
(ii)(b) Alkoxymercuration-demercuration
Analogous procedure where an alcohol (ROH) is added across the double bond to give ethers with Markovnikov regiochemistry.
Ex. 11 Supply the structures for (X) and (Y) in the following two-step reaction:
C3H7CH=CH2
Sol.
(X) = C3H7CH(OH)CH2-HgOAc
(Y) = C3H7CH(OH)CH3
(X is an organomercurial alcohol; Y is the demercurated alcohol.)
Ex. 12 Identify final product in the following :
(a)
(b)
Ans.
(a)
(b)
(iii) Hydroboration-oxidation (syn addition, anti-Markovnikov)
Hydroboration adds boron and hydrogen across the double bond in a syn fashion; subsequent oxidation with H2O2/OH- replaces boron with OH giving an alcohol with anti-Markovnikov regiochemistry.
Mechanistic summary: BH3 (or B2H6 in THF) adds across the double bond syn; oxidation retains stereochemistry.
Hydroboration-oxidation complements oxymercuration-demercuration in regiochemistry.
(i) Hydration with dilute H2SO4 proceeds via carbocation rearrangement.
(ii) Hydration via Hg(OAc)2/NaBH4 proceeds with Markovnikov addition without rearrangement.
(iii) Hydroboration-oxidation proceeds with anti-Markovnikov regiochemistry.
(iv) Addition of hydrogen halides (HX)
General: R-CH=CH2 + HX → R-CHX-CH3 (Markovnikov addition unless radical conditions present).
Notes:
- Anti-Markovnikov addition (radical chain) is observed for HBr in the presence of peroxides and light (peroxide effect).
- HF, HCl and HI typically give polar (ionic) addition and follow Markovnikov rule.
Ex. 13 Predict the major products of the following reactions and propose mechanism to support your predictions.
(A)
(B)
(C)
Sol.
(A)
(B)
(C)
Ex. 14 Identify the products in the following reactions :
(a) F3C-CH=CH2 + HCl →
(b) O2N-CH=CH2 + HCl →
(c) CH3O-CH=CH2 + HCl →
(d) PhCH=CHCH3 + HCl →
(e)
Q.6 Give the products of the following reactions :
Q.7 Give the reactant (alkene) of the following products.
(v) Addition of halogens (X2)
Halogens (Cl2, Br2) add across C=C to give vicinal dihalides. The reaction proceeds via a bridged halonium ion intermediate and is typically anti stereospecific (nucleophile attacks from the back side of the halonium ion).
Notes:
- F2 is not used: reaction is too reactive and often destructive (explosive) producing CO2 and H2O.
- I2 addition is often reversible and not generally useful for straightforward addition.
- Bromine addition is the basis for the qualitative bromine test for unsaturation.
- Halogen addition in inert solvent is stereospecific anti addition; in sunlight radical additions may occur giving different outcomes.
- Relative reactivity in radical addition: F2 (explosive) > Cl2 > Br2 > I2.
Mechanism (summary):
Step 1: Formation of a halonium ion by electrophilic attack of X+ on the π bond.
Step 2: Back-side attack by X- to open the halonium ion giving anti addition product.
X- attacks from the back side of the halonium ion.
(vi) Addition of dihydrogen
Hydrogenation of alkenes with H2 over Pd, Pt or Ni catalysts yields alkanes (see catalytic hydrogenation under (I)).
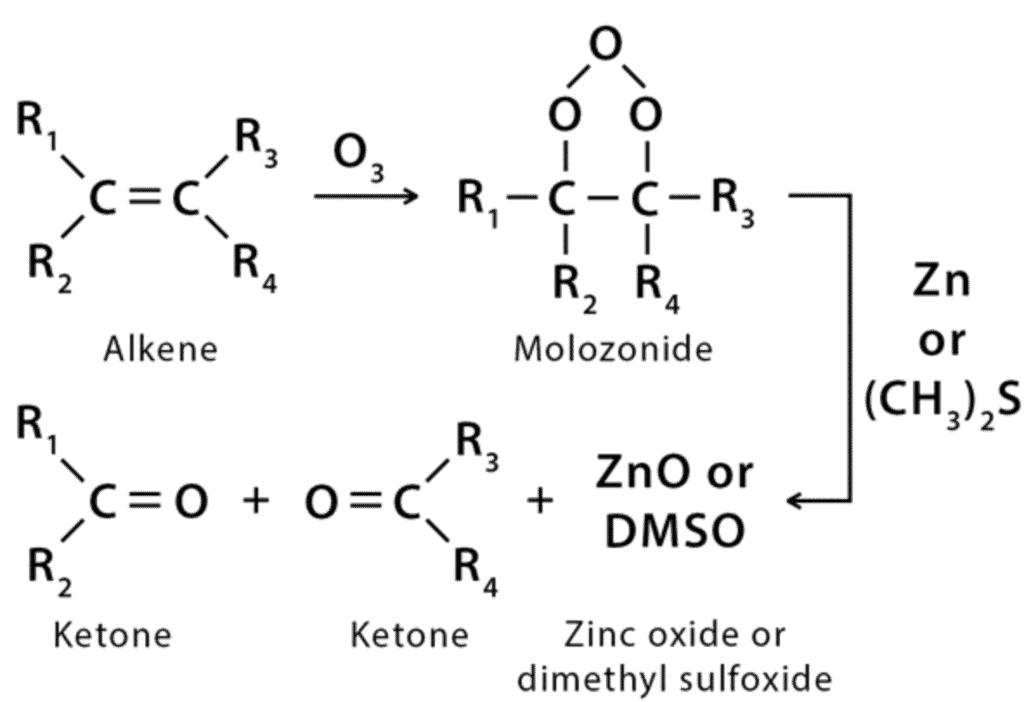
(vii) Ozonolysis
Ozonolysis cleaves double bonds using ozone (O3), converting alkenes into carbonyl fragments (aldehydes, ketones) or further oxidised products (carboxylic acids) depending on work-up conditions (reductive workup gives carbonyls; oxidative workup can give acids).
- Alkenes ozonised with reductive workup (Zn/CH3COOH or (CH3)2S) give aldehydes/ketones.
- Alkynes give diketones or carboxylic acid derivatives depending on conditions.
- Ozonolysis of elastomers causes ozone cracking (cleavage of C=C bonds).
- Ozonolysis of azo compounds may yield nitrosamines or other nitrogen-containing fragments.
(viii) Polymerisation
Many alkenes polymerise to form long-chain macromolecules (polymers). Ethene polymerises under appropriate temperature, pressure and catalyst to give polythene (polyethylene). Other alkenes, such as propene, give polypropene (polypropylene).

Uses of polymers:
- Polymers are widely used to manufacture plastic bags, bottles, containers, pipes, toys, radio and TV cabinets and many household items.
- Polypropene is used for crates, buckets and many moulded articles.
- Excessive use of non-biodegradable polymers (polythene, polypropylene) presents environmental concerns and waste management challenges.
FAQs on Alkenes: Nomenclature, Properties & Preparation
| 1. What is the IUPAC nomenclature for alkenes? |  |
| 2. What are the properties of alkenes? |  |
| 3. How can alkenes be prepared? |  |
| 4. What are the physical properties of alkenes? |  |
| 5. How do the properties of alkenes differ from alkanes? |  |






