Cheat Sheet: Structure of the Atom
1. Charged Particles in Matter
Experiments with electricity and gas discharge tubes showed that atoms contain charged particles. These particles are called subatomic particles. Scientists discovered three important particles inside an atom: electrons, protons, and neutrons.
Subatomic Particles

Important points:
- Electrons carry negative charge.
- Protons carry positive charge.
- Neutrons have no charge.
- Protons and neutrons together are called nucleons.
2. Atomic Models
Scientists proposed several models to explain the structure of atoms.
Thomson's Model of the Atom
Thomson proposed the plum pudding model.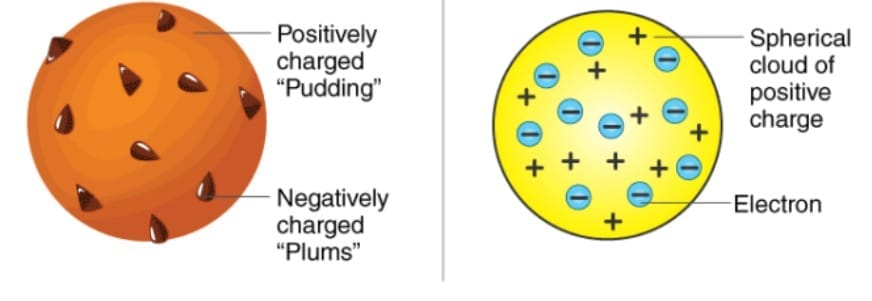
Main features
- Atom is a sphere of positive charge.
- Electrons are embedded in this sphere.
- Positive and negative charges balance each other.
- Therefore, the atom is electrically neutral.
Limitation
Could not explain the results of Rutherford's alpha particle experiment.
Rutherford's Model of the Atom
Rutherford performed the alpha particle scattering experiment using a thin gold foil.
Observations
- Most alpha particles passed straight through the foil.
- Some particles were deflected slightly.
- A very small number of particles rebounded.
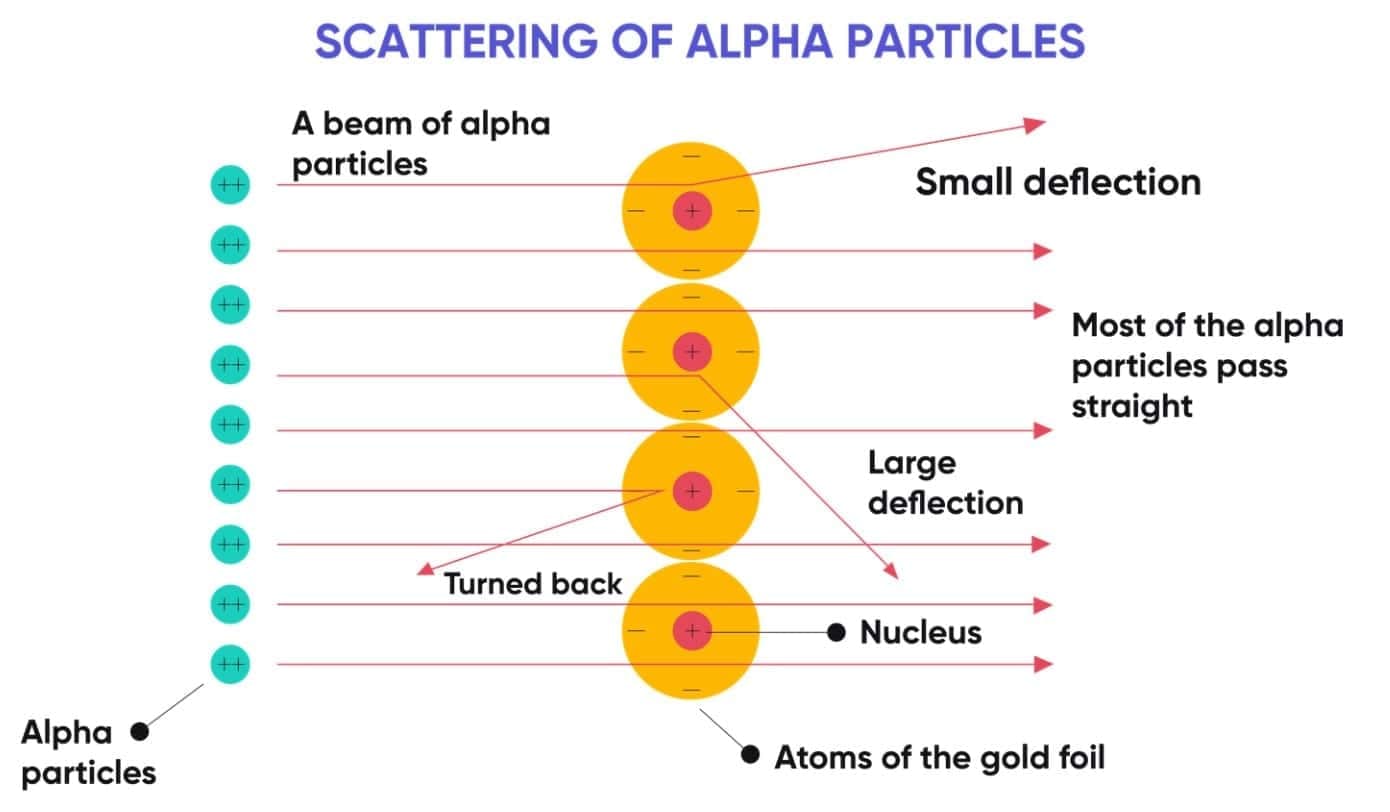
Conclusions
- Most of the atom is empty space.
- Positive charge is concentrated in a very small region called the nucleus.
- Nearly all the mass of the atom lies in the nucleus.
- Electrons revolve around the nucleus in circular paths.
Drawback
According to classical physics:
- Moving electrons should lose energy.
- They should eventually fall into the nucleus.
- This would make atoms unstable, which contradicts reality.
Bohr's Model of the Atom
Niels Bohr modified Rutherford's model to explain atomic stability.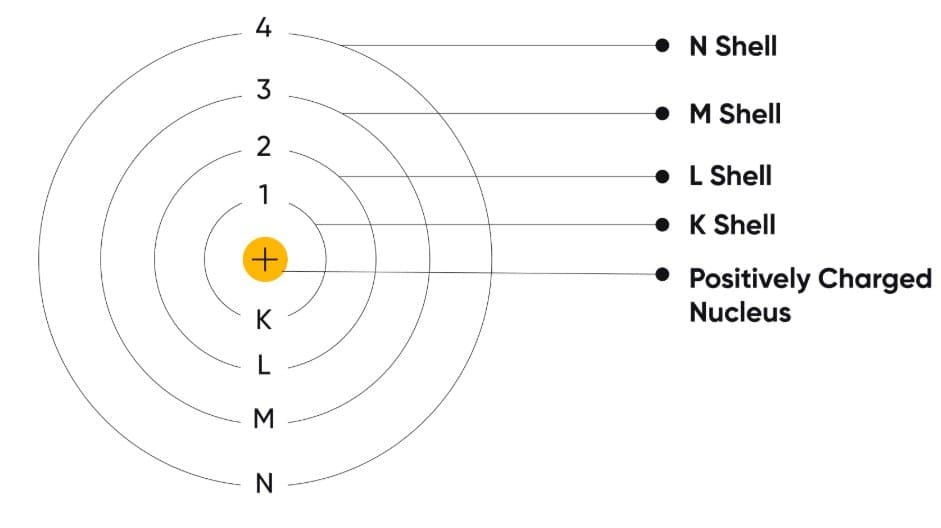
Main Postulates
- Electrons revolve around the nucleus in fixed circular paths called shells or energy levels.
- Electrons do not lose energy while moving in these shells.
- Each shell has a definite energy.
Names of Shells
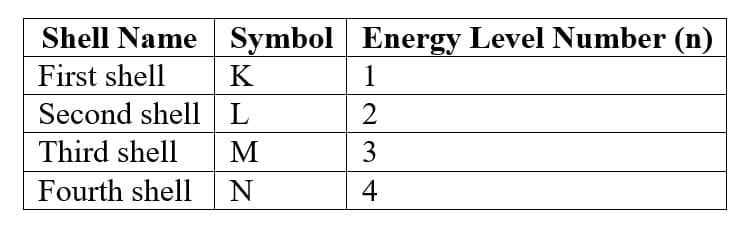
3. Distribution of Electrons in Shells
Bohr and Bury proposed rules for electron distribution.
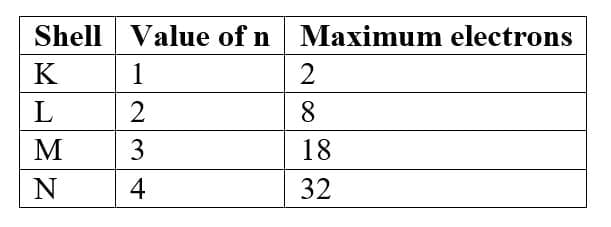
Rule 1: Maximum electrons in a shell
The maximum number of electrons in a shell is given by:
2n²
where n = shell number
Rule 2
The outermost shell cannot contain more than 8 electrons.
Rule 3
Electrons are filled in shells from inner shell to outer shell.
Example:
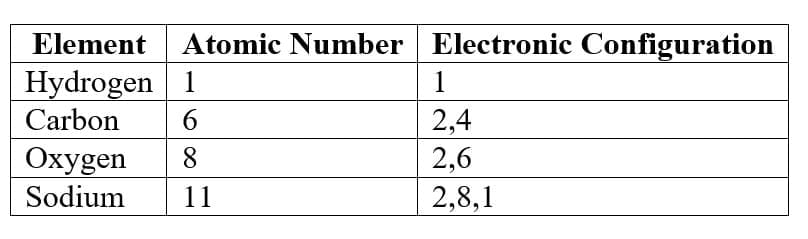
4. Valency
Definition
Valency is the combining capacity of an atom. It depends on the number of electrons in the outermost shell (valence electrons).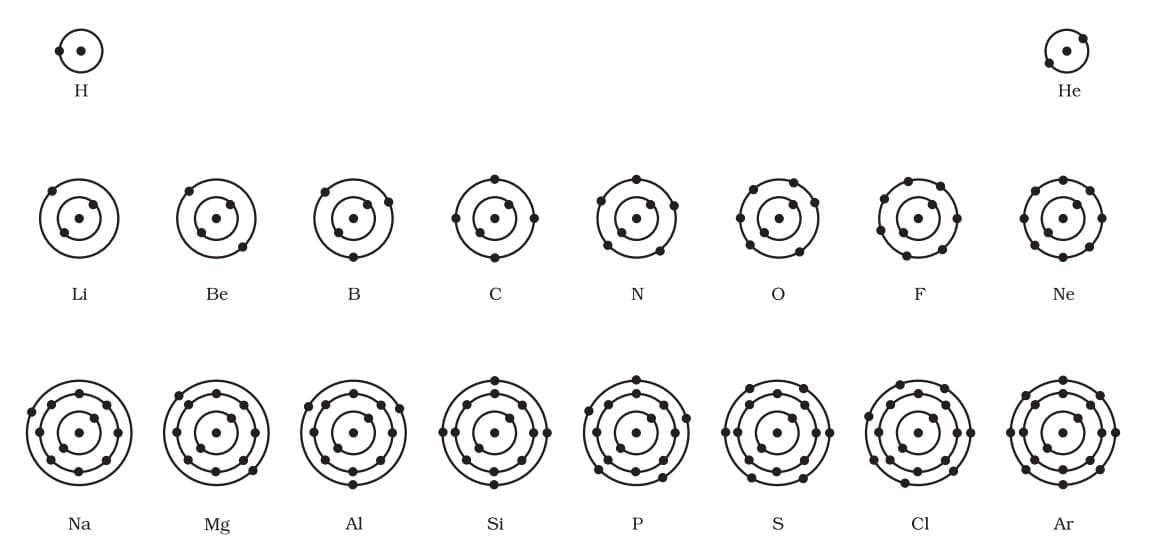 Schematic atomic structure of the first eighteen elements
Schematic atomic structure of the first eighteen elements
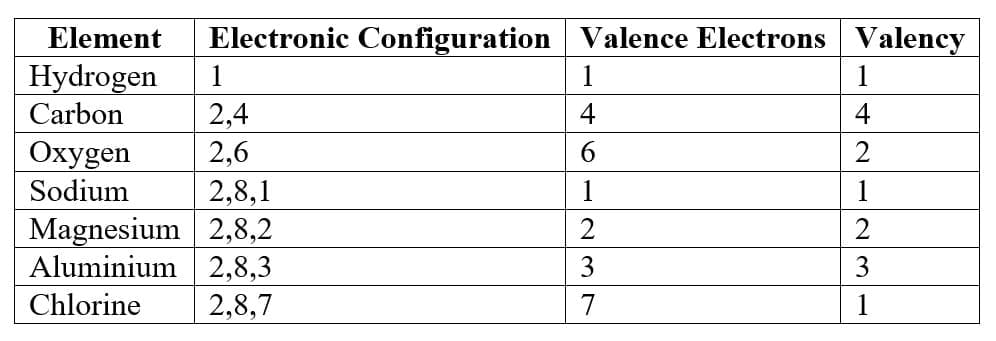
Determination of Valency
If outer shell electrons ≤ 4
Valency = number of electrons.
If outer shell electrons > 4
Valency = 8 - number of electrons.
Examples
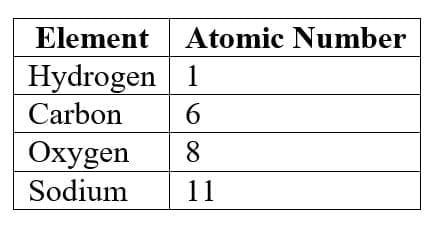
5. Atomic Number
Definition
Atomic number (Z) is the number of protons present in the nucleus of an atom.
Important facts:
- All atoms of the same element have the same atomic number.
- In a neutral atom:
Number of protons = Number of electrons
Examples
6. Mass Number
Definition
Mass number (A) is the total number of protons and neutrons in the nucleus.
Formula:
A = Number of protons + Number of neutrons
Example:
Carbon atom
Protons = 6
Neutrons = 6
Mass number = 12
7. Representation of an Atom
Atoms are represented as:

Where:
A = Mass number
Z = Atomic number
X = Symbol of element
Example:
¹⁴₇N
This means:
- Atomic number = 7
- Mass number = 14
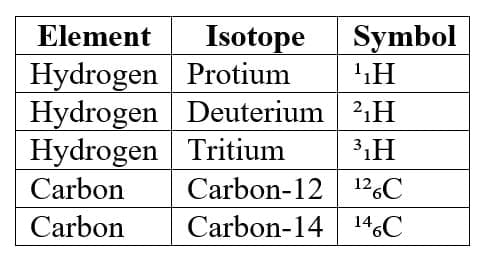
8. Isotopes
Definition
Isotopes are atoms of the same element that have:
- Same atomic number
- Different mass numbers.
Examples
Properties of Isotopes
Chemical properties: Same
Physical properties: Different
Uses of Isotopes

9. Isobars
Definition
Isobars are atoms of different elements having:
- Same mass number
- Different atomic numbers.
Example

FAQs on Cheat Sheet: Structure of the Atom
| 1. What are sub-atomic particles and their significance in atomic structure? |  |
| 2. Describe the main atomic models developed throughout history. |  |
| 3. What is the Bohr-Bury scheme for electron distribution? |  |
| 4. How do atomic number and mass number differ? |  |
| 5. What are isotopes and how do they differ from one another? |  |







