Raoult's Law
Who discovered Raoult's Law?
Raoult's law is named after the French chemist François-Marie Raoult, who, by experiments on mixtures of volatile liquids, observed that the vapour pressure of a solution falls when a solute is added. Raoult reported these findings in 1887. The law is a fundamental relationship in physical chemistry describing vapour pressures of ideal solutions.
Statement of Raoult's Law
For a solution of volatile liquids at a given temperature, the partial vapour pressure of each component above the solution is directly proportional to its mole fraction in the liquid phase.
Mathematical form
The partial pressure of component i in an ideal solution is given by
\(p_i = p_i^{\circ}\,x_i\)
where
- \(p_i\) is the partial vapour pressure of component i above the solution,
- \(p_i^{\circ}\) is the vapour pressure of pure component i at the same temperature,
- \(x_i\) is the mole fraction of component i in the liquid phase.
Total vapour pressure of a binary ideal solution
Consider an ideal binary solution of volatile liquids A and B. By Dalton's law of partial pressures, the total vapour pressure above the solution is the sum of the partial pressures of A and B.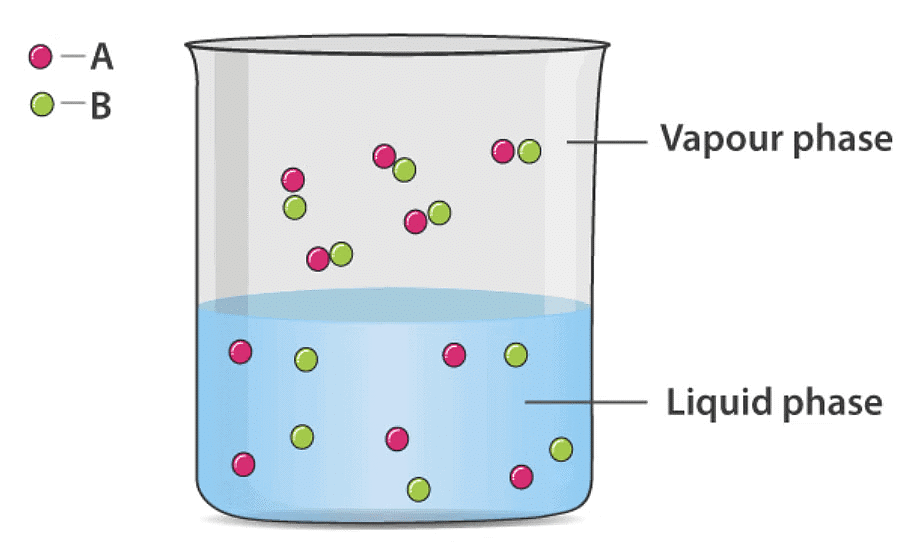 Ideal Solution of Two Volatile Liquids
Ideal Solution of Two Volatile Liquids
\(P_{\text{total}} = P_A + P_B\)
By Raoult's law for each component,
\(P_A = P_A^{\circ}\,x_A\)
\(P_B = P_B^{\circ}\,x_B\)
Therefore, for the solution
\(P_{\text{total}} = P_A^{\circ}\,x_A + P_B^{\circ}\,x_B\)
Using \(x_A + x_B = 1\), we may also write
\(P_{\text{total}} = P_A^{\circ} + (P_B^{\circ} - P_A^{\circ})\,x_B\)
This is a linear relation in \(x_B\); a plot of \(P_{\text{total}}\) versus \(x_B\) is a straight line with y-intercept \(P_A^{\circ}\) and slope \((P_B^{\circ}-P_A^{\circ})\).
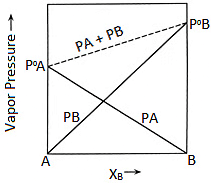 Plot of Vapour Pressure and Mole Fraction
Plot of Vapour Pressure and Mole Fraction The total pressure of a solution
\(P_{\text{solution}} = P_A + P_B = x_A P_A^{\circ} + x_B P_B^{\circ}\)
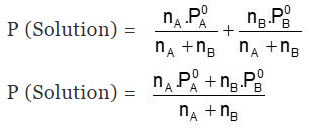
Try yourself: “Total pressure of gas mixture is the sum of individual pressures”. Which law is reflected in this statement?
Vapour composition above a binary solution
Vapour composition refers to the mole fractions of components in the vapour phase. For components A and B in the vapour phase, denote their mole fractions by \(y_A\) and \(y_B\), so \(y_A + y_B = 1\).
By Dalton's law, the partial pressure of A equals its mole fraction in the vapour times the total pressure:
\(P_A = y_A\,P_{\text{total}}\)
Therefore
\(y_A = \dfrac{P_A}{P_{\text{total}}} = \dfrac{x_A P_A^{\circ}}{x_A P_A^{\circ} + x_B P_B^{\circ}}\)
Similarly,
\(y_B = \dfrac{P_B}{P_{\text{total}}} = \dfrac{x_B P_B^{\circ}}{x_A P_A^{\circ} + x_B P_B^{\circ}}\)
Special note:
\(x_A\) and \(x_B\) are the mole fractions of A and B in the liquid phase and satisfy \(x_A + x_B = 1\).
\(y_A\) and \(y_B\) are the mole fractions of A and B in the vapour phase and satisfy \(y_A + y_B = 1\).
Raoult's Law as a special case of Henry's Law
- For a dilute solution of a gas or volatile solute in a solvent, Henry's law states that the partial pressure of the gas above the solution is proportional to its mole fraction in the solution: \(p = K_H\,x\), where \(K_H\) is Henry's constant for that gas-solvent pair at the given temperature.
- Raoult's law, \(p_i = p_i^{\circ} x_i\), has the same mathematical form. For a solvent that is volatile and behaves ideally, the proportionality constant \(p_i^{\circ}\) plays the role of Henry's constant. Thus Raoult's law is a limiting case of Henry's law when the component behaves like the pure liquid (that is, when the proportionality constant equals the vapour pressure of the pure component).
- Therefore, Raoult's law turns into a unique instance of Henry's law in which KH get to be equivalent to pi˚
Limitations and deviations from Raoult's Law
Raoult's law applies exactly only to ideal solutions. Real solutions often deviate from ideal behaviour because intermolecular forces between unlike molecules differ from those between like molecules. Key points:
- Raoult's law is valid when interactions between A-A, B-B and A-B molecules are similar so that the solution behaves ideally. True ideal solutions are uncommon.
- When interactions differ, the solution shows deviation from Raoult's law. Deviations are classified as:
- Negative deviation: The observed vapour pressure is lower than predicted by Raoult's law. This happens when A-B interactions are stronger than A-A and B-B interactions. Example: mixtures with hydrogen bonding such as chloroform and acetone (strong specific interactions) can show negative deviation.
- Positive deviation: The observed vapour pressure is higher than predicted. This occurs when A-B interactions are weaker than A-A and B-B interactions, so each component escapes more readily into the vapour. Examples include mixtures like benzene and methanol (where unlike interactions are weaker).
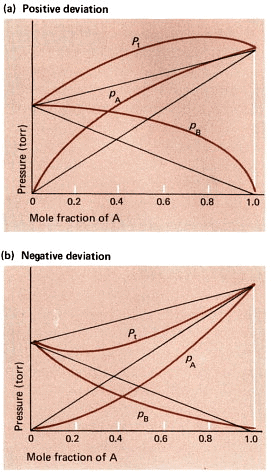 Positive and Negative Deviation
Positive and Negative DeviationSolved examples
Example 1. The vapour pressures of methanol and ethanol at a given temperature are 88.7 mm Hg and 44.5 mm Hg respectively. An ideal solution is formed at the same temperature by mixing 60 g of ethanol with 40 g of methanol. Calculate the total vapour pressure of the solution.
Solution.
Calculate moles of each component.
\( \text{Moles of ethanol (C}_2\text{H}_5\text{OH)} = \dfrac{60\text{ g}}{46.0\text{ g mol}^{-1}} = 1.3043\text{ mol}\)
\( \text{Moles of methanol (CH}_3\text{OH)} = \dfrac{40\text{ g}}{32.0\text{ g mol}^{-1}} = 1.2500\text{ mol}\)
Compute mole fractions (take A = methanol, B = ethanol if matching vapour pressures):
\(x_{\text{methanol}} = \dfrac{1.2500}{1.2500 + 1.3043} = 0.4896\)
\(x_{\text{ethanol}} = 1 - 0.4896 = 0.5104\)
Apply Raoult's law for each component.
\(P_{\text{methanol}} = x_{\text{methanol}} \, P_{\text{methanol}}^{\circ} = 0.4896 \times 88.7 = 43.44\ \text{mm Hg}\)
\(P_{\text{ethanol}} = x_{\text{ethanol}} \, P_{\text{ethanol}}^{\circ} = 0.5104 \times 44.5 = 22.71\ \text{mm Hg}\)
Therefore total vapour pressure:
\(P_{\text{total}} = 43.44 + 22.71 = 66.15\ \text{mm Hg}\)
Example 2. The composition of the vapour over a binary ideal solution depends on the composition of the liquid. If \(x_A\) and \(y_A\) are the mole fractions of A in the liquid and vapour phases respectively, find the value of \(x_A\) for which \(y_A - x_A\) has a minimum. What is the value of the total pressure at this composition?
Solution.
Start with the expression for \(y_A\):
\(y_A = \dfrac{x_A P_A^{\circ}}{x_A P_A^{\circ} + (1-x_A) P_B^{\circ}}\)
Define the function to be minimised:
\(f(x_A) = y_A - x_A = \dfrac{x_A P_A^{\circ}}{x_A P_A^{\circ} + (1-x_A) P_B^{\circ}} - x_A\)
Simplify the expression algebraically to obtain a form suitable for differentiation and set the derivative to zero. The algebra leads to the condition
\(x_A = \dfrac{\sqrt{P_B^{\circ}}}{\sqrt{P_A^{\circ}} + \sqrt{P_B^{\circ}}}\)
At this composition, the total vapour pressure evaluates to
\(P_{\text{total}} = \sqrt{P_A^{\circ} P_B^{\circ}}\)
Thus the minimum difference between vapour and liquid composition occurs when the liquid mole fraction of A equals the ratio of \(\sqrt{P_B^{\circ}}\) to the sum \(\sqrt{P_A^{\circ}} + \sqrt{P_B^{\circ}}\), and the total pressure is the geometric mean of the pure component vapour pressures.
Example 3. A very small amount of a non-volatile, non-dissociating solute is dissolved in 56.8 cm3 of benzene (density = 0.889 g cm-3). At room temperature the vapour pressure of the solution is 99.88 mm Hg while that of pure benzene is 100.00 mm Hg. Find the molality of the solution. If the freezing point of the solution is 0.73 °C lower than that of pure benzene, calculate the molal freezing point-depression constant \(K_f\) for benzene.
Solution.
Calculate mass and moles of benzene present.
\(\text{Mass of benzene} = 56.8\ \text{cm}^3 \times 0.889\ \text{g cm}^{-3} = 50.4952\ \text{g}\)
\(\text{Moles of benzene} = \dfrac{50.4952\ \text{g}}{78.11\ \text{g mol}^{-1}} = 0.6466\ \text{mol}\)
Vapour pressure lowering:
\(\Delta P = P^{\circ} - P = 100.00 - 99.88 = 0.12\ \text{mm Hg}\)
For a non-volatile solute, Raoult's law gives \(P = x_{\text{solvent}}\,P^{\circ}\). For dilute solutions, the mole fraction of solute \(x_{\text{solute}} \approx \dfrac{\Delta P}{P^{\circ}}\).
Number of moles of solute (approximately):
\(n_{\text{solute}} \approx \dfrac{\Delta P}{P^{\circ}} \times n_{\text{solvent}} = \dfrac{0.12}{100.00} \times 0.6466 = 0.0007759\ \text{mol}\)
Molality (moles solute per kg solvent):
\(\text{Molality} = \dfrac{0.0007759\ \text{mol}}{0.0504952\ \text{kg}} = 0.01536\ \text{m}\)
Using freezing point depression \(\Delta T_f = K_f \times m\):
\(K_f = \dfrac{\Delta T_f}{m} = \dfrac{0.73\ \text{K}}{0.01536\ \text{mol kg}^{-1}} = 47.5\ \text{K kg mol}^{-1}\)
Therefore, the molality of the solution is approximately 0.01536 m and the molal freezing point-depression constant \(K_f\) for benzene is approximately 47.5 K kg mol-1.
Example 4. What is the composition of the vapour which is in equilibrium at 30 °C with a benzene-toluene solution with a mole fraction of benzene of 0.400? (Given \(P_{\text{benzene}}^{\circ} = 119\ \text{torr}\) and \(P_{\text{toluene}}^{\circ} = 37.0\ \text{torr}\).)
Solution.
Given \(x_{\text{benzene}} = 0.400\). Then \(x_{\text{toluene}} = 0.600\).
Total pressure using Raoult's law:
\(P_{\text{total}} = x_{\text{benzene}} P_{\text{benzene}}^{\circ} + x_{\text{toluene}} P_{\text{toluene}}^{\circ}\)
\(P_{\text{total}} = 0.400\times119 + 0.600\times37.0 = 47.6 + 22.2 = 69.8\ \text{torr}\)
Partial pressure of benzene:
\(P_{\text{benzene}} = 0.400\times119 = 47.6\ \text{torr}\)
Mole fraction of benzene in vapour:
\(y_{\text{benzene}} = \dfrac{P_{\text{benzene}}}{P_{\text{total}}} = \dfrac{47.6}{69.8} = 0.6819\)
Hence the mole fraction of toluene in the vapour is
\(y_{\text{toluene}} = 1 - 0.6819 = 0.3181\)
FAQs on Raoult's Law
| 1. Who discovered Raoult's Law? |  |
| 2. What is Raoult’s Law? |  |
| 3. How can we find vapor composition using Raoult's Law? |  |
| 4. How is Raoult's Law related to Henry's Law? |  |
| 5. What are the limitations of Raoult’s Law? |  |







