Bonding in Metal Complexes & Applications of Coordination Compounds
Bonding in Metal Complexes
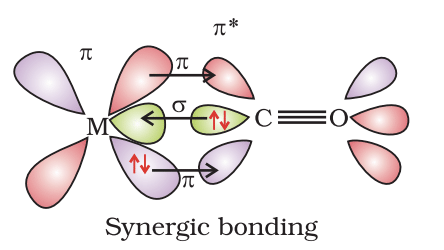
Complexes in which carbon monoxide acts as a ligand are called metal carbonyls. These provide clear examples of the types of bonding that occur between a metal centre and a ligand.
In metal carbonyls two principal interactions occur between the metal and CO ligand:
- Sigma (σ) donation: The lone pair on the carbon atom of CO is donated into a suitable vacant orbital of the metal (often a vacant d, s or hybrid orbital). This gives an M←CO σ bond.
- Pi (π) back-donation: Electrons in filled metal d orbitals are donated back into the vacant antibonding π* orbitals of CO. This produces M→CO π-backbonding.
Because both processes occur simultaneously the bonding is described as synergic-σ donation strengthens the M-C interaction and π back-donation strengthens the M-C interaction while weakening the C-O bond in CO. The extent of backbonding affects observable properties such as the C-O stretching frequency in IR spectroscopy (stronger backbonding → lower ν(CO)).
Example: Ni(CO)4 (tetracarbonyl nickel(0)) and Fe(CO)5 (pentacarbonyl iron(0)).
The bonding in these complexes can be rationalised using the 18-electron rule. For example, Ni(0) has 10 valence electrons and each CO donates 2 electrons; in Ni(CO)4 the total electron count is 10 + 4×2 = 18, a stable configuration. Similarly, Fe(0) in Fe(CO)5 reaches 18 electrons (8 + 5×2 = 18).
Applications of Coordination Compounds
Coordination compounds (complexes) have many practical applications because complex formation can change the chemical and physical properties (solubility, colour, volatility, reactivity) of a metal atom or ion. The following are major applications.
Detection and Estimation
The formation of characteristic coloured or sparingly soluble complexes is widely used for qualitative detection and quantitative estimation of metal ions.
(i) Detection and gravimetric estimation
The detection and estimation of Ni2+ is based on the formation of a scarlet-red complex with dimethylglyoxime (DMG). Formation of a coloured or insoluble complex provides a simple test and a means for gravimetric estimation.
(a) Fe3+ is detected by formation of a blood-red complex with thiocyanate (KSCN).
(b) Many organic reagents (ligands) are used for the gravimetric estimation of metal ions. Common pairs are shown in the table below.
| Metal ion to be estimated | Cu2+ | Ni2+ | Fe3+ | Al3+ | Co2+ |
| Organic reagent used | Benzoin oxime | Dimethyl glyoxime | 1,10-Phenanthroline | 8-Hydroxyquinoline | α-Nitroso-β-naphthol |
(c) EDTA is used as a complexing agent in volumetric (complexometric) analysis of metal ions such as Ca2+, Mg2+ and Zn2+.
(d) The variety of colours exhibited by transition metal complexes is utilised in colorimetric analysis for the estimation of many metals.
Metallurgical Processes
(ii) Complex formation is used in extraction and purification of metals.
(a) Extraction of silver and gold: Native silver and gold can be dissolved by formation of cyanide complexes. Silver ore treated with sodium cyanide solution in the presence of oxygen dissolves as a soluble cyanide complex; silver is later precipitated by addition of zinc (cementation).
1. Leaching of silver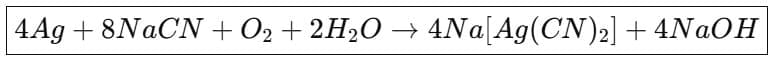
2. Formation of silver cyanide complex from argentite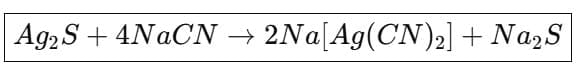 3. Oxidation of sodium sulphide (by air)
3. Oxidation of sodium sulphide (by air)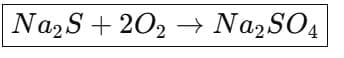
4. Precipitation of silver by zinc (cementation)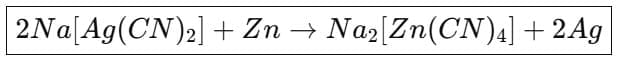
Silver and gold are precipitated from their cyanide complexes by addition of scrap zinc.
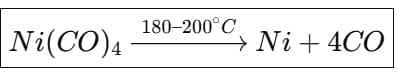
(b) Mond's process for the purification of nickel is based on the formation of the volatile organometallic complex Ni(CO)4. Nickel reacts with carbon monoxide to give nickel tetracarbonyl which is volatile at 50-80 °C; on heating to higher temperatures it decomposes to give pure nickel and CO.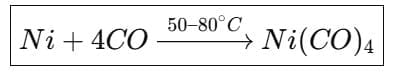
Photography
(iii) In photography the silver halide image on the photographic plate/film is fixed by dissolving unexposed silver bromide as a soluble complex with sodium thiosulphate (hypo), forming a soluble silver-thiosulphate complex.
AgBr + 2 Na2S2O3Na3[Ag(S2O3)2] + NaBr
The soluble complex removes unreacted silver bromide leaving the reduced (metallic) silver image intact.
Electroplating
(iv) Metal complexes release metal ions slowly and give uniform metal deposition. Cyano complexes of silver, gold, copper and other metals are commonly used in electrodeposition baths to produce smooth, adherent metal coatings.
Biological Processes
(v) Metal complexes are central to many biological functions.
- Haemoglobin is an iron(II) complex that carries oxygen in blood.
- Vitamin B12 is a cobalt complex essential for biological processes such as nucleic acid metabolism.
- Chlorophyll is a magnesium complex that plays a central catalytic role in photosynthesis.
Try yourself: Which of the following biologically important coordination compounds has a magnesium central atom?
Organometallic Compounds
Organometallic compounds are defined as compounds in which there is at least one direct bond between a metal atom (or metal centre) and a carbon atom of an organic group (commonly an alkyl, aryl, vinyl or allyl group). Compounds of boron, silicon, phosphorus and other elements with direct M-C bonds are also classified under organometallic chemistry. Many organometallics are highly useful reagents and catalysts in organic synthesis and industrial processes.
Classification of Organometallic Compounds
Organometallic compounds are commonly classified as follows.
- Sigma (σ)-bonded organometallics: The metal-carbon bond is a direct σ bond between the metal and the carbon atom. Examples include Grignard reagents R-Mg-X (where R is alkyl/aryl and X is halogen) and dialkylmetal compounds such as (C2H5)2Zn, (CH3)4Sn, Pb(CH3)4, Al2(CH3)6. Many of these are electron-deficient and may form bridged (dimeric) structures similar to diborane.
- Pi (π)-bonded organometallics: The ligand donates π-electrons (from a double bond, triple bond or an aromatic system) into the metal and often accepts backbonding from the metal. Examples include Zeise's salt and ferrocene. The number of carbon atoms interacting with the metal is indicated by the h (eta) notation: η2, η5, η6, etc., which denote the number of contiguous atoms of the ligand bonded to the metal.
- Sigma and pi bonded organometallics: Metal carbonyls belong here because the metal-carbon bond has both σ-donor and π-backbonding character. These compounds commonly contain metals in a low oxidation state (often zero) and may be mononuclear, bridged or polynuclear.
Al2(CH3)6 is a dimeric example; two methyl groups act as bridges between two aluminium atoms, making the compound electron deficient and structurally similar to diborane (B2H6).
Common π-bound examples: Zeise's salt (K[PtCl3(η2-C2H4)]) and ferrocene (Fe(η5-C5H5)2).
In a metal carbonyl the σ-bond is formed by donation from the lone pair on the carbon of CO into a vacant orbital of the metal.
π back-bonding is caused when a filled metal orbital overlaps with the empty π* orbital of CO, sometimes called back donation. In diagrams the π overlap is shown perpendicular to the nodal plane of the σ bond.
In olefinic (alkene) complexes the ligand donates π electrons to the metal and the metal back-donates into the π* orbital of the olefin; this combined donation/back-donation weakens the C=C bond and is a key concept in many catalytic processes (e.g., hydrogenation, polymerisation).
Applications of Organometallic Compounds
- Tetraethyl lead (TEL) (an organolead compound) was used as an antiknock additive in gasoline (historical application; use has declined due to environmental and health concerns).
- Wilkinson's catalyst [Rh(PPh3)3Cl] is a homogeneous catalyst used for selective hydrogenation of alkenes under mild conditions.
- Mond's process uses the formation and decomposition of volatile Ni(CO)4 for extraction and purification of nickel (described earlier).
- Ziegler-Natta catalysts (e.g., trialkylaluminium + TiCl4) are heterogeneous catalysts employed for polymerisation of ethene to polyethene and for stereospecific polymerisations of other alkenes.
Points to be remembered
- CH3B(OCH3) is an organometallic compound because it contains a B-C bond; in contrast, B(OCH3) without a direct B-C bond is not organometallic.
- The closed ring complexes formed by polydentate ligands are called chelates. Chelation usually increases complex stability (chelate effect).
- Estimation of Ni2+ is done by complexing with dimethylglyoxime (DMG), while estimation of Ca2+ and Mg2+ ions is commonly done by titration against EDTA (complexometric titration).
- Complexes in which ligands can be readily substituted by other ligands are termed labile complexes. For example [Cu(NH3)4]2+ is labile and ammonia ligands can be replaced by cyanide:
[Cu(NH3)4]2+ + 4 CN- → [Cu(CN)4]2- + 4 NH3
The product [Cu(CN)4]2- is more stable than the initial complex, illustrating ligand substitution driven by relative stabilities.
- Octahedral complexes of type MA3B3 can show geometric isomerism as facial (fac) and meridional (mer) isomers. In the fac isomer each trio of identical donor atoms occupies adjacent positions at the corners of one face of the octahedron; in the mer isomer they lie around a meridian.
- Biomolecules: Haemoglobin (Fe complex), chlorophyll (Mg complex) and vitamin B12 (Co complex) are important biological coordination compounds.
- σ-bonded organometallics generally contain a non-transition metal linked to a carbon atom by a σ bond (for example, Grignard reagent R-MgX).
- π-bonded organometallics are formed by donation of π electrons from a multiple bond or aromatic system to the metal (for example, Zeise's salt K[PtCl3(η2-C2H4)], and ferrocene Fe(η5-C5H5)2).
- Grignard reagents are among the most useful organometallics; the polarity of the C-Mg bond (carbon carrying partial negative charge) makes them powerful nucleophiles for synthesis of a wide range of organic compounds.
Try yourself: The coordination complex chloridotris (triphenylphosphine) rhodium(I) is used in the hydrogenation of alkenes. It is also known as __________ catalyst.
FAQs on Bonding in Metal Complexes & Applications of Coordination Compounds
| 1. What is the bonding in metal complexes? |  |
| 2. What are the applications of coordination compounds? |  |
| 3. What are organometallic compounds? |  |
| 4. What is the crystal field theory? |  |
| 5. How does ligand field theory differ from crystal field theory? |  |






