NCERT Solutions Class 12 Chemistry Chapter 8 - Aldehydes, Ketones & Carboxylic Acids
Page No. 231 - Intext Questions
Q8.1: Write the structures of the following compounds.
(i) α-Methoxypropionaldehyde
(ii) 3-Hydroxybutanal
(iii) 2-Hydroxycyclopentane carbaldehyde
(iv) 4-Oxopentanal
(v) Di-sec-butyl ketone
(vi) 4-Fluoroacetophenone
Ans:
(i) 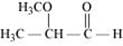
(ii) 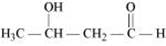
(iii) 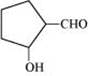
(iv) 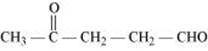
(v) 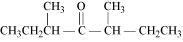
(vi) 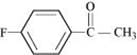
Page No. 234 - Intext Questions
Q8.2: Write the structures of products of the following reactions;
(i) 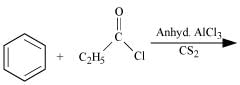
(ii) 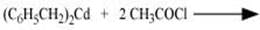
(iii) 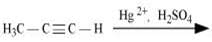
(iv) 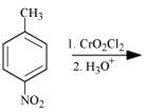
Ans:
(i)
(ii)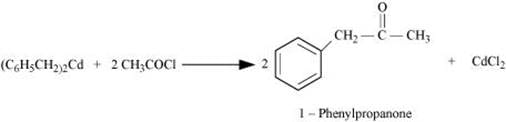
(iii)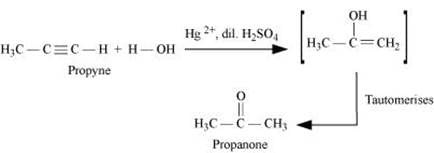
(iv)
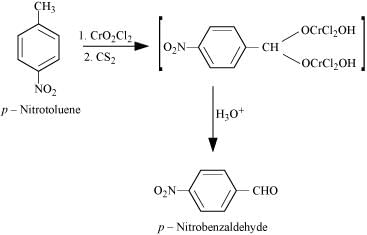
Page No. 236 - Intext Questions
Q8.3: Arrange the following compounds in increasing order of their boiling points.
CH3CHO, CH3CH2OH, CH3OCH3, CH3CH2CH3
Ans: The molecular masses of the given compounds are in the range 44 to 46. CH3CH2OH undergoes extensive intermolecular H-bonding, resulting in the association of molecules. Therefore, it has the highest boiling point. CH3CHO is more polar than CH3OCH3 and so CH3CHO has stronger intermolecular dipole − dipole attraction than CH3OCH3⋅ CH3CH2CH3 has only weak van der Waals force. Thus, the arrangement of the given compounds in the increasing order of their boiling points is given by:
CH3CH2CH3 < CH3OCH3 < CH3CHO < CH3CH2OH
Page No. 243 - Intext Questions
Q8.4: Arrange the following compounds in increasing order of their reactivity in nucleophilic addition reactions.
(i) Ethanal, Propanal, Propanone, Butanone.
(ii) Benzaldehyde, p-Tolualdehyde, p-Nitrobenzaldehyde, Acetophenone.
Hint: Consider the steric effect and electronic effect.
Ans: (i)
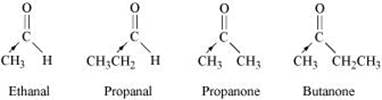
The I effect of the alkyl group increases in the order:
Ethanal < Propanal < Propanone < Butanone
The electron density at the carbonyl carbon increases with the increase in the I effect. As a result, the chances of attack by a nucleophile decrease. Hence, the increasing order of the reactivities of the given carbonyl compounds in nucleophilic addition reactions is:
Butanone < Propanone < Propanal < Ethanal
(ii)
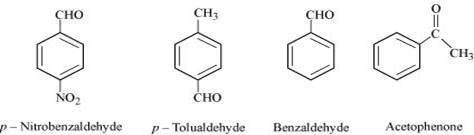
The I effect is more in ketone than in aldehyde. Hence, acetophenone is the least reactive in nucleophilic addition reactions. Among aldehydes, the I effect is the highest in p-tolualdehyde because of the presence of the electron-donating −CH3 group and the lowest in p-nitrobezaldehyde because of the presence of the electron-withdrawing −NO2 group. Hence, the increasing order of the reactivities of the given compounds is:
Acetophenone < p-tolualdehyde < Benzaldehyde < p-Nitrobenzaldehyde
Q8.5: Predict the products of the following reactions:
(i) 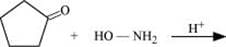
(ii) 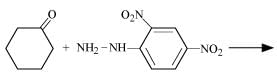
(iii) 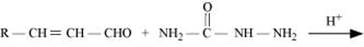
(iv) 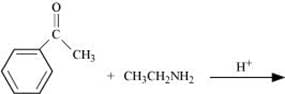
Ans: (i)
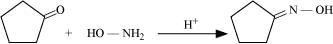
(ii)
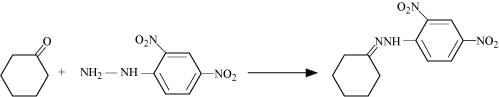
(iii)
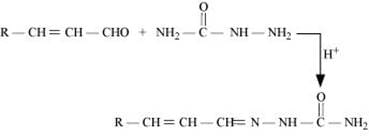
(iv)
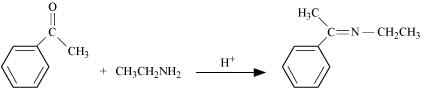
Page No. 245 - Intext Questions
Q8.6: Give the IUPAC names of the following compounds:
(i) PhCH2CH2COOH
(ii) (CH3)2C=CHCOOH
(iii) 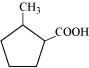
(iv) 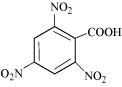
Ans: (i) 3-Phenylpropanoic acid
(ii) 3-Methylbut-2-enoic acid
(iii) 2-Methylcyclopentanecarboxylic acid
(iv) 2,4,6-Trinitrobenzoic acid
Page No. 248 - Intext Questions
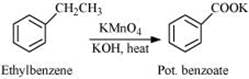
Q 8.7: Show how each of the following compounds can be converted to benzoic acid.
(i) Ethylbenzene
(ii) Acetophenone
(iii) Bromobenzene
(iv) Phenylethene (Styrene)
Ans: (i)
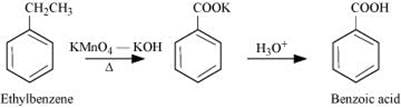
(ii)
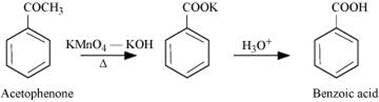
(iii)
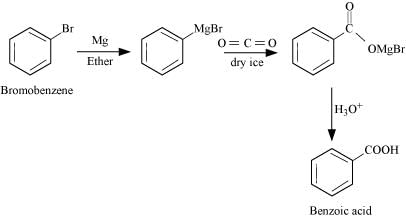
(iv)
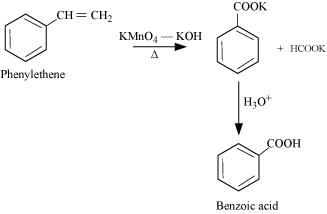
Page No. 254 - Intext Questions
Q8.8: Which acid of each pair shown here would you expect to be stronger?
(i) CH3CO2H or CH2FCO2H
(ii)CH2FCO2H or CH2ClCO2H
(iii) CH2FCH2CH2CO2H or CH3CHFCH2CO2H
(iv)
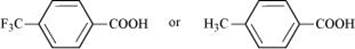
Ans: (i)
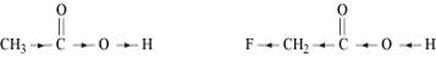
The I effect of −CH3 group increases the electron density on the O−H bond. Therefore, the release of protons becomes difficult. On the other hand, the −I effect of F decreases the electron density on the O−H bond. Therefore, protons can be released easily. Hence, CH2FCO2H is a stronger acid than CH3CO2H.
(ii)

F has a stronger −I effect than Cl. Therefore, CH2FCO2H can release protons more easily than CH2ClCO2H. Hence, CH2FCO2H is a stronger acid than CH2ClCO2H.
(iii)
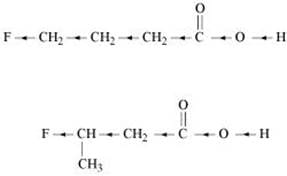
The inductive effect decreases with an increase in distance. Hence, the I effect of F in CH3CHFCH2CO2H is more than it is in CH2FCH2CH2CO2H. Hence, CH3CHFCH2CO2H is a stronger acid than CH2FCH2CH2CO2H.
(iv)
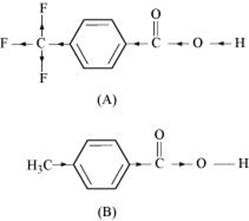
Due to the −I effect of F, it is easier to release a proton in the case of compound (A). However, in the case of compound (B), the release of the proton is difficult due to the I effect of −CH3 group. Hence, (A) is a stronger acid than (B).
Exercises
Q8.1: What is meant by the following terms? Give an example of the reaction in each case.
(i) Cyanohydrin
(ii) Acetal
(iii) Semicarbazone
(iv) Aldol
(v) Hemiacetal
(vi) Oxime
(vii) Ketal
(vii) Imine
(ix) 2,4-DNP-derivative
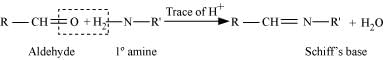
(x) Schiff’s base
Ans: (i) Cyanohydrin:
Cyanohydrins are organic compounds having the formula RR′C(OH)CN, where R and R′ can be alkyl or aryl groups.
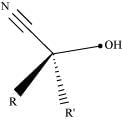
Aldehydes and ketones react with hydrogen cyanide (HCN) in the presence of excess sodium cyanide (NaCN) as a catalyst to field cyanohydrin. These reactions are known as cyanohydrin reactions.

Cyanohydrins are useful synthetic intermediates.
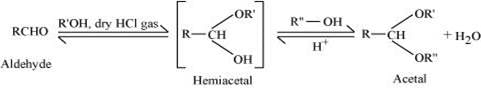
(ii) Acetal:
Acetals are gem−dialkoxy alkanes in which two alkoxy groups are present on the terminal carbon atom. One bond is connected to an alkyl group while the other is connected to a hydrogen atom.
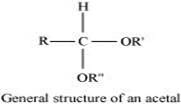
When aldehydes are treated with two equivalents of monohydric alcohol in the presence of dry HCl gas, hemiacetals are produced that further react with one more molecule of alcohol to yield acetal.
(iii) Semicarbarbazone:
Semicarbazones are derivatives of aldehydes and ketones produced by the condensation reaction between a ketone or aldehyde and semicarbazide.
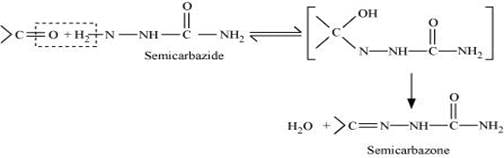
Semicarbazones are useful for the identification and characterization of aldehydes and ketones.
(iv) Aldol:
A β-hydroxy aldehyde or ketone is known as an aldol. It is produced by the condensation reaction of two molecules of the same or one molecule each of two different aldehydes or ketones in the presence of a base.
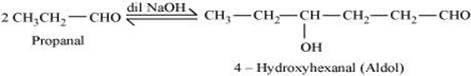
(v) Hemiacetal:
Hemiacetals are α−alkoxyalcohols
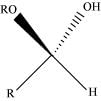
The general structure of a hemiacetal
Aldehyde reacts with one molecule of a monohydric alcohol in the presence of dry HCl gas.
(vi) Oxime:
Oximes are a class of organic compounds having the general formula RR′CNOH, where R is an organic side chain and R′ is either hydrogen or an organic side chain. If R′ is H, then it is known as aldoxime and if R′ is an organic side chain, it is known as ketoxime.
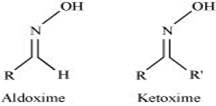
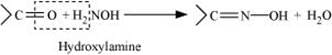
On treatment with hydroxylamine in a weakly acidic medium, aldehydes or ketones form oximes.
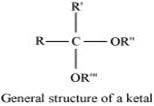
(vii) Ketal:
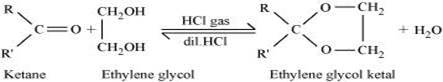
Ketals are gem−dialkoxyalkanes in which two alkoxy groups are present on the same carbon atom within the chain. The other two bonds of the carbon atom are connected to two alkyl groups.
Ketones react with ethylene glycol in the presence of dry HCl gas to give a cyclic product known as ethylene glycol ketals.
(viii) Imine:
Imines are chemical compounds containing a carbon nitrogen double bond.
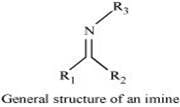
Imines are produced when aldehydes and ketones react with ammonia and its derivatives.
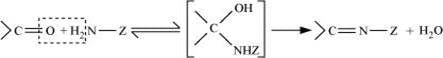
(ix) 2, 4−DNP−derivative:
2, 4−dinitrophenylhydragones are 2, 4−DNP−derivatives, which are produced when aldehydes or ketones react with 2, 4−dinitrophenylhydrazine in a weakly acidic medium.
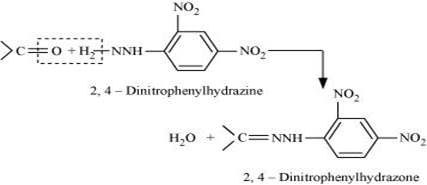
To identify and characterize aldehydes and ketones, 2, 4−DNP derivatives are used.
(x) Schiff’s base:
Schiff’s base (or azomethine) is a chemical compound containing a carbon-nitrogen double bond with the nitrogen atom connected to an aryl or alkyl group but not hydrogen. They have the general formula R1R2C = NR3. Hence, it is an imine.
It is named after a scientist, Hugo Schiff.
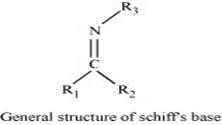
Aldehydes and ketones on treatment with primary aliphatic or aromatic amines in the presence of trace of an acid yields a Schiff’s base.
Q8.2: Name the following compounds according to IUPAC system of nomenclature:
(i) CH3CH(CH3)CH2CH2CHO
(ii) CH3CH2COCH(C2H5)CH2CH2Cl
(iii) CH3CH=CHCHO
(iv) CH3COCH2COCH3
(v) CH3CH(CH3)CH2C(CH3)2COCH3
(vi) (CH3)3CCH2COOH
(vii) OHCC6H4CHO-p
Ans: (i) 4-methylpentanal
(ii) 6-Chloro-4-ethylhexan-3-one
(iii) But-2-en-1-al
(iv) Pentane-2,4-dione
(v) 3,3,5-Trimethylhexan-2-one
(vi) 3,3-Dimethylbutanoic acid
(vii) Benzene-1,4-dicarbaldehyde
Q8.2: Draw the structures of the following compounds.
(i) 3-Methylbutanal
(ii) p-Nitropropiophenone
(iii) p-Methylbenzaldehyde
(iv) 4-Methylpent-3-en-2-one
(v) 4-Chloropentan-2-one
(vi) 3-Bromo-4-phenylpentanoic acid
(vii) p,p’-Dihydroxybenzophenone
(viii) Hex-2-en-4-ynoic acid
Ans:
(i) 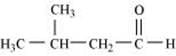
(ii)
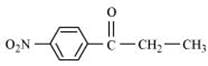
(iii)
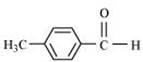
(iv) 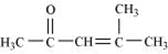
(v) 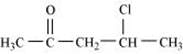 (vi)
(vi) 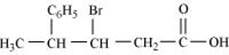
(vii)
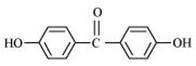 (viii)
(viii) 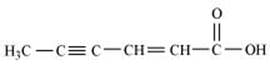
Q8.4: Write the IUPAC names of the following ketones and aldehydes. Wherever possible, give also common names.
(i) CH3CO(CH2)4CH3
(ii) CH3CH2CHBrCH2CH(CH3)CHO
(iii) CH3(CH2)5CHO
(iv) Ph-CH=CH-CHO
(v) 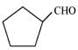 (vi) PhCOPh
(vi) PhCOPh
Ans: (i) CH3CO(CH2)4CH3
IUPAC name: Heptan-2-one
Common name: Methyl n-propyl ketone
(ii) CH3CH2CHBrCH2CH(CH3)CHO
IUPAC name: 4-Bromo-2-methylhaxanal
Common name: (γ-Bromo-α-methyl-caproaldehyde)
(iii) CH3(CH2)5CHO
IUPAC name: Heptanal
(iv) Ph-CH=CH-CHO
IUPAC name: 3-phenylprop-2-enal
Common name: β-Pheynolacrolein
(v)
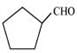
IUPAC name: Cyclopentanecarbaldehyde
(vi)PhCOPh
IUPAC name: Diphenylmethanone
Common name: Benzophenone
Q8.5: Draw structures of the following derivatives.
(i) The 2,4-dinitrophenylhydrazone of benzaldehyde
(ii) Cyclopropanone oxime
(iii) Acetaldehydedimethylacetal
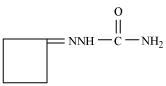
(iv) The semicarbazone of cyclobutanone
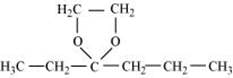
(v) The ethylene ketal of hexan-3-one
(vi) The methyl hemiacetal of formaldehyde
Ans:
(i) 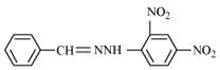 (ii)
(ii) 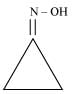
(iii)
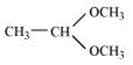
(iv)
(v)
(vi)
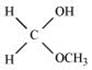
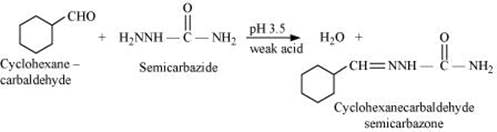
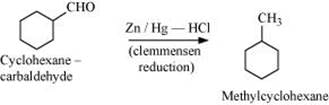
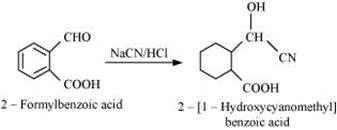
Q8.6: Predict the products formed when cyclohexanecarbaldehyde reacts with following reagents.
(i) PhMgBr and then H3O
(ii) Tollens’ reagent
(iii) Semicarbazide and weak acid
(iv) Excess ethanol and acid
(v) Zinc amalgam and dilute hydrochloric acid
Ans: (i)
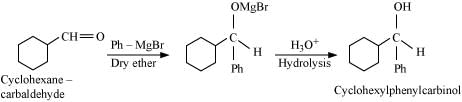
(ii)
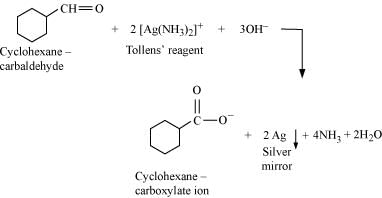
(iii)
(iv)
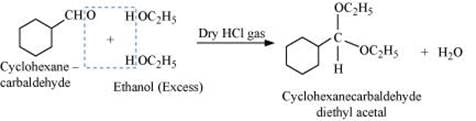
(v)
Q8.7: Which of the following compounds would undergo aldol condensation, which the Cannizzaro reaction, and which neither? Write the structures of the expected products of aldol condensation and Cannizzaro reaction.
(i) Methanal
(ii) 2-Methylpentanal
(iii) Benzaldehyde
(iv) Benzophenone
(v) Cyclohexanone
(vi) 1-Phenylpropanone
(vii) Phenylacetaldehyde
(viii) Butan-1-ol
(ix) 2, 2-Dimethylbutanal
Ans: Aldehydes and ketones having at least one α-hydrogen undergo aldol condensation. The compounds (ii) 2−methylpentanal, (v) cyclohexanone, (vi) 1-phenylpropanone, and (vii) phenylacetaldehyde contain one or more α-hydrogen atoms. Therefore, these undergo aldol condensation.
Aldehydes having no α-hydrogen atoms undergo Cannizzaro reactions. The compounds (i) Methanal, (iii) Benzaldehyde, and (ix) 2, 2-dimethylbutanal do not have any α-hydrogen. Therefore, these undergo cannizzaro reactions.
Compound (iv) Benzophenone is a ketone having no α-hydrogen atom and compound (viii) Butan-1-ol is an alcohol. Hence, these compounds do not undergo either aldol condensation or cannizzaro reactions.
Aldol condensation
(ii)
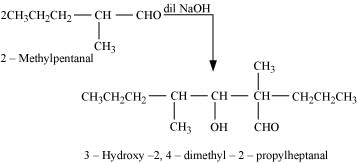
(v)
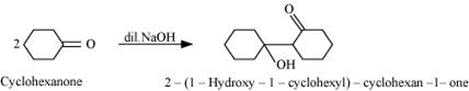
(vi)
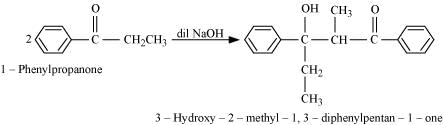
(vii)
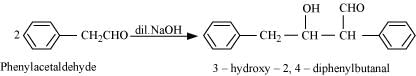
Cannizzaro reaction
(i)
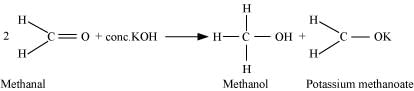
(iii)
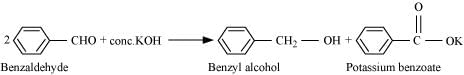
(ix)
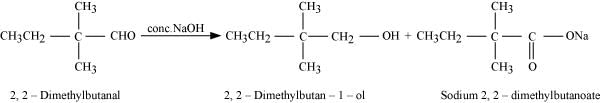
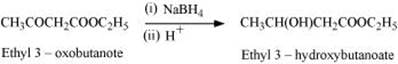
Q8.8: How will you convert ethanal into the following compounds?
(i) Butane-1, 3-diol
(ii) But-2-enal
(iii) But-2-enoic acid
Ans: (i) On treatment with dilute alkali, ethanal produces 3-hydroxybutanal gives butane-1, 3-diol on reduction.
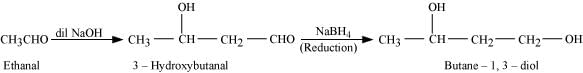
(ii) On treatment with dilute alkali, ethanal gives 3-hydroxybutanal which on heating produces but-2-enal.
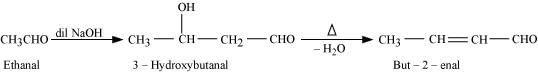
(iii) When treated with Tollen’s reagent, But-2-enal produced in the above reaction produces but-2-enoic acid .
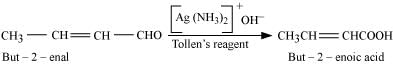
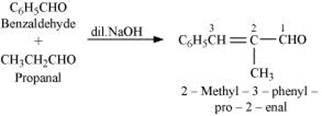
Q8.9: Write structural formulas and names of four possible aldol condensation products from propanal and butanal. In each case, indicate which aldehyde acts as a nucleophile and which as an electrophile.
Ans: (i) Taking two molecules of propanal, one which acts as a nucleophile and the other as an electrophile.
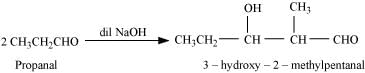
(ii) Taking two molecules of butanal, one which acts as a nucleophile and the other as an electrophile.
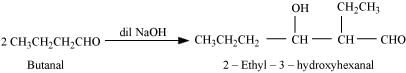
(iii) Taking one molecule each of propanal and butanal in which propanal acts as a nucleophile and butanal acts as an electrophile.
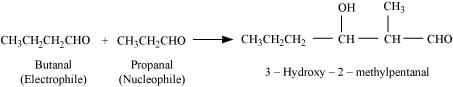
(iv) Taking one molecule each of propanal and butanal in which propanal acts as an electrophile and butanal acts as a nucleophile.
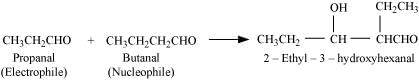
Q8.10: An organic compound with the molecular formula C9H10O forms 2, 4-DNP derivative, reduces Tollens’ reagent and undergoes Cannizzaro reaction. On vigorous oxidation, it gives 1, 2-benzenedicarboxylic acid. Identify the compound.
Ans: It is given that the compound (with molecular formula C9H10O) forms 2, 4-DNP derivative and reduces Tollen’s reagent. Therefore, the given compound must be an aldehyde.
Again, the compound undergoes cannizzaro reaction and on oxidation gives 1, 2-benzenedicarboxylic acid. Therefore, the −CHO group is directly attached to a benzene ring and this benzaldehyde is ortho-substituted. Hence, the compound is 2-ethylbenzaldehyde.
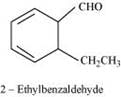
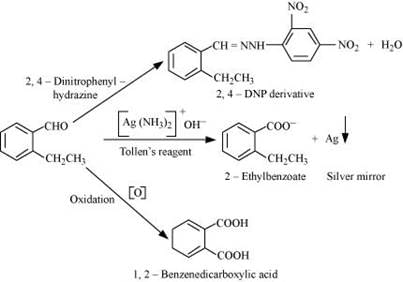
The given reactions can be explained by the following equations.
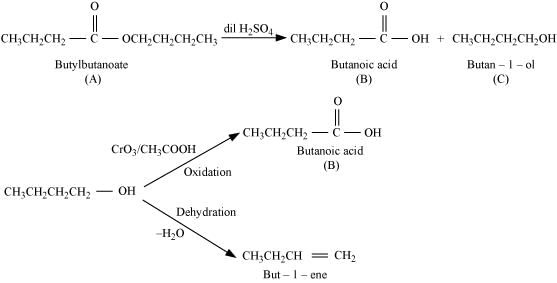
Q8.11: An organic compound (A) (molecular formula C8H16O2) was hydrolyzed with dilute sulphuric acid to give a carboxylic acid (B) and alcohol (C). Oxidation of (C) with chromic acid produced (B). (C) on dehydration gives but-1-ene.Write equations for the reactions involved.
Ans: An organic compound A with molecular formula C8H16O2 gives a carboxylic acid (B) and an alcohol (C) on hydrolysis with dilute sulphuric acid. Thus, compound A must be an ester. Further, alcohol C gives acid B on oxidation with chromic acid. Thus, B and C must contain equal number of carbon atoms.
Since compound A contains a total of 8 carbon atoms, each of B and C contain 4 carbon atoms.
Again, on dehydration, alcohol C gives but-1-ene. Therefore, C is of straight chain and hence, it is butan-1-ol.
On oxidation, Butan-1-ol gives butanoic acid. Hence, acid B is butanoic acid.
Hence, the ester with molecular formula C8H16O2 is butylbutanoate.
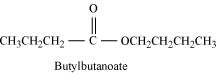
All the given reactions can be explained by the following equations.
Q8.12: Arrange the following compounds in increasing order of their property as indicated:
(i) Acetaldehyde, Acetone, Di-tert-butyl ketone, Methyl tert-butyl ketone (reactivity towards HCN)
(ii) CH3CH2CH(Br)COOH, CH3CH(Br)CH2COOH, (CH3)2CHCOOH, CH3CH2CH2COOH (acid strength)
(iii) Benzoic acid, 4-Nitrobenzoic acid, 3,4-Dinitrobenzoic acid, 4-Methoxybenzoic acid (acid strength)
ANSWER : (i) When HCN reacts with a compound, the attacking species is a nucleophile, CN−. Therefore, as the negative charge on the compound increases, its reactivity with HCN decreases. In the given compounds, the I effect increases as shown below. It can be observed that steric hindrance also increases in the same
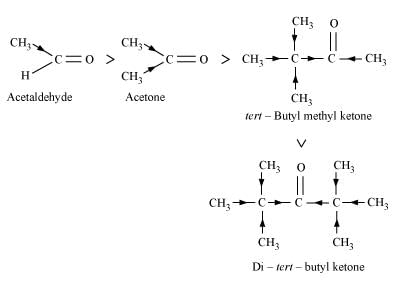
Hence,the given compounds can be arranged according to their increasing reactivities toward HCN as:
Di-tert-butyl ketone < Methyl tert-butyl ketone < Acetone < Acetaldehyde
(ii) After losing a proton, carboxylic acids gain a negative charge as shown:
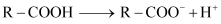
Now, any group that will help stabilise the negative charge will increase the stability of the carboxyl ion and as a result, will increase the strength of the acid. Thus, groups having I effect will decrease the strength of the acids and groups having −I effect will increase the strength of the acids. In the given compounds, −CH3 group has I effect and Br− group has −I effect. Thus, acids containing Br− are stronger.
Now, the I effect of isopropyl group is more than that of n-propyl group. Hence, (CH3)2CHCOOH is a weaker acid than CH3CH2CH2COOH.
Also, the −I effect grows weaker as distance increases. Hence, CH3CH(Br)CH2COOH is a weaker acid than CH3CH2CH(Br)COOH.
Hence, the strengths of the given acids increase as:
(CH3)2CHCOOH < CH3CH2CH2COOH < CH3CH(Br)CH2COOH < CH3CH2CH(Br)COOH
(iii) As we have seen in the previous case, electron-donating groups decrease the strengths of acids, while electron-withdrawing groups increase the strengths of acids. As methoxy group is an electron-donating group, 4-methoxybenzoic acid is a weaker acid than benzoic acid. Nitro group is an electron-withdrawing group and will increase the strengths of acids. As 3,4-dinitrobenzoic acid contains two nitro groups, it is a slightly stronger acid than 4-nitrobenzoic acid. Hence, the strengths of the given acids increase as:
4-Methoxybenzoic acid < Benzoic acid < 4-Nitrobenzoic acid < 3,4-Dinitrobenzoic acid
Q8.13: Give simple chemical tests to distinguish between the following pairs of compounds.
(i) Propanal and Propanone
(ii) Acetophenone and Benzophenone
(iii) Phenol and Benzoic acid
(iv) Benzoic acid and Ethyl benzoate
(v) Pentan-2-one and Pentan-3-one
(vi) Benzaldehyde and Acetophenone
(vii) Ethanal and Propanal
Ans: (i) Propanal and propanone can be distinguished by the following tests.
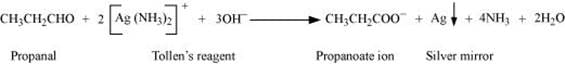
(a) Tollen’s test
Propanal is an aldehyde. Thus, it reduces Tollen’s reagent. But, propanone being a ketone does not reduce Tollen’s reagent.
(b) Fehling’s test
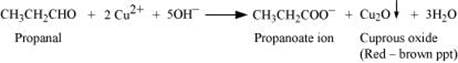
Aldehydes respond to Fehling’s test, but ketones do not.
Propanal being an aldehyde reduces Fehling’s solution to a red-brown precipitate of Cu2O, but propanone being a ketone does not.
(c) Iodoform test:
Aldehydes and ketones having at least one methyl group linked to the carbonyl carbon atom respond to iodoform test. They are oxidized by sodium hypoiodite (NaOI) to give iodoforms. Propanone being a methyl ketone responds to this test, but propanal does not.
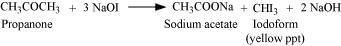
(ii) Acetophenone and Benzophenone can be distinguished using the iodoform test.
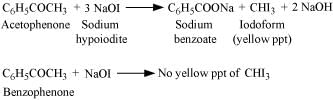
Iodoform test:
Methyl ketones are oxidized by sodium hypoiodite to give yellow ppt. of iodoform. Acetophenone being a methyl ketone responds to this test, but benzophenone does not.
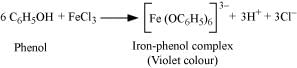
(iii) Phenol and benzoic acid can be distinguished by the ferric chloride test.
Ferric chloride test:
Phenol reacts with neutral FeCl3 to form an iron-phenol complex giving violet colouration.
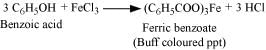
But benzoic acid reacts with neutral FeCl3 to give a buff coloured ppt. of ferric benzoate.
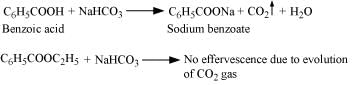
(iv) Benzoic acid and Ethyl benzoate can be distinguished by sodium bicarbonate test.
Sodium bicarbonate test:
Acids react with NaHCO3 to produce brisk effervescence due to the evolution of CO2 gas.
Benzoic acid being an acid responds to this test, but ethylbenzoate does not.
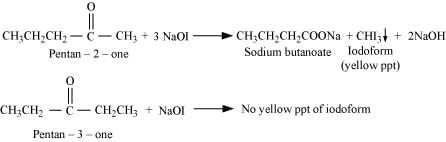
(v) Pentan-2-one and pentan-3-one can be distinguished by iodoform test.
Iodoform test:
Pentan-2-one is a methyl ketone. Thus, it responds to this test. But pentan-3-one not being a methyl ketone does not respond to this test.
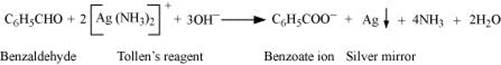
(vi) Benzaldehyde and acetophenone can be distinguished by the following tests.
(a) Tollen’s Test
Aldehydes respond to Tollen’s test. Benzaldehyde being an aldehyde reduces Tollen’s reagent to give a red-brown precipitate of Cu2O, but acetophenone being a ketone does not.
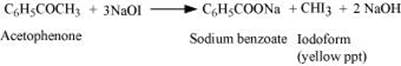
(b) Iodoform test
Acetophenone being a methyl ketone undergoes oxidation by sodium hypoiodite (NaOI) to give a yellow ppt. of iodoform. But benzaldehyde does not respond to this test.
(vii) Ethanal and propanal can be distinguished by iodoform test.
Iodoform test
Aldehydes and ketones having at least one methyl group linked to the carbonyl carbon atom responds to the iodoform test. Ethanal having one methyl group linked to the carbonyl carbon atom responds to this test. But propanal does not have a methyl group linked to the carbonyl carbon atom and thus, it does not respond to this state.
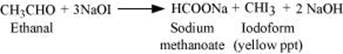
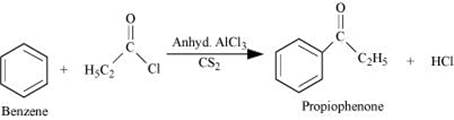
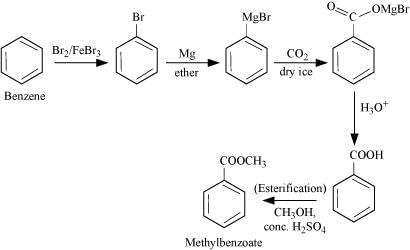
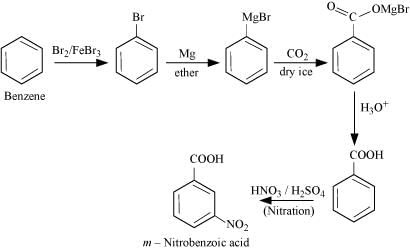
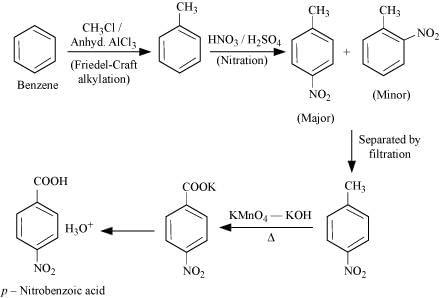
Q8.14: How will you prepare the following compounds from benzene? You may use any inorganic reagent and any organic reagent having not more than one carbon atom
(i) Methyl benzoate
(ii) m-Nitrobenzoic acid
(iii) p-Nitrobenzoic acid
(iv) Phenylacetic acid
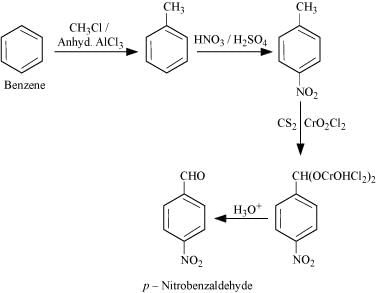
(v) p-Nitrobenzaldehyde.
Ans: (i)
(ii)
(iii)
(iv)
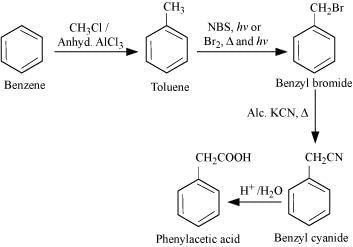
(v)
Q8.15: How will you bring about the following conversions in not more than two steps?
(i) Propanone to Propene
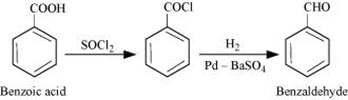
(ii) Benzoic acid to Benzaldehyde
(iii) Ethanol to 3-Hydroxybutanal
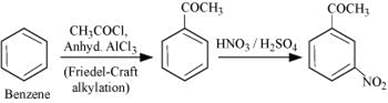
(iv) Benzene to m-Nitroacetophenone
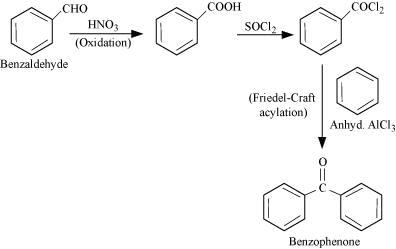
(v) Benzaldehyde to Benzophenone
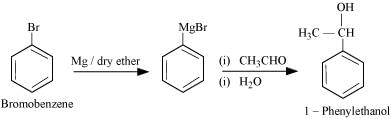
(vi) Bromobenzene to 1-Phenylethanol
(vii) Benzaldehyde to 3-Phenylpropan-1-ol
(viii) Benazaldehyde to α-Hydroxyphenylacetic acid
(ix) Benzoic acid to m- Nitrobenzyl alcohol
Ans: (i)
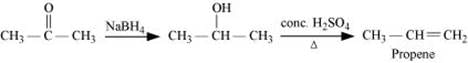
(ii)
(iii)
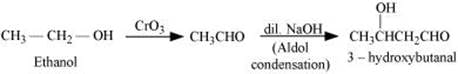
(iv)
(v)
(vi)
(vii)
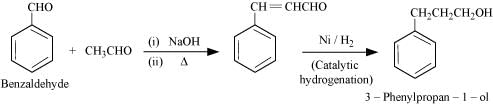
(viii)
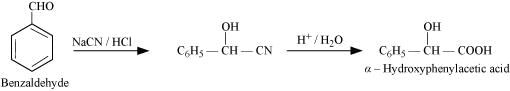
(ix)
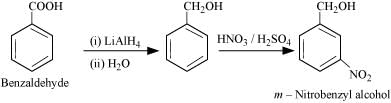
Q8.16: Describe the following:
(i) Acetylation
(ii) Cannizzaro reaction
(iii) Cross aldol condensation
(iv) Decarboxylation
Ans: (i) Acetylation
The introduction of an acetyl functional group into an organic compound is known as acetylation. It is usually carried out in the presence of a base such as pyridine, dirnethylaniline, etc. This process involves the substitution of an acetyl group for an active hydrogen atom. Acetyl chloride and acetic anhydride are commonly used as acetylating agents.
For example, acetylation of ethanol produces ethyl acetate.

(ii) Cannizzaro reaction:
The self oxidation-reduction (disproportionation) reaction of aldehydes having no α-hydrogens on treatment with concentrated alkalis is known as the Cannizzaro reaction. In this reaction, two molecules of aldehydes participate where one is reduced to alcohol and the other is oxidized to carboxylic acid.
For example, when ethanol is treated with concentrated potassium hydroxide, ethanol and potassium ethanoate are produced.
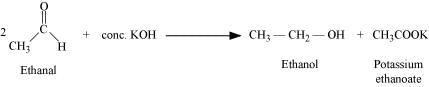
(iii) Cross-aldol condensation:
When aldol condensation is carried out between two different aldehydes, or two different ketones, or an aldehyde and a ketone, then the reaction is called a cross-aldol condensation. If both the reactants contain α-hydrogens, four compounds are obtained as products.
For example, ethanal and propanal react to give four products.
(iv) Decarboxylation:
Decarboxylation refers to the reaction in which carboxylic acids lose carbon dioxide to form hydrocarbons when their sodium salts are heated with soda-lime.
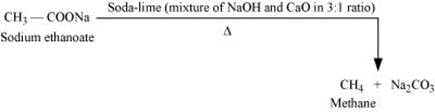
Decarboxylation also takes place when aqueous solutions of alkali metal salts of carboxylic acids are electrolyzed. This electrolytic process is known as Kolbe’s electrolysis.
Q8.17: Complete each synthesis by giving missing starting material, reagent or products
(i) 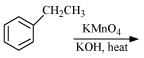
(ii) 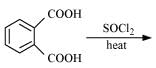
(iii) 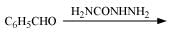
(iv) 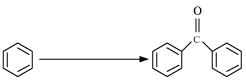
(v) 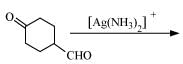
(vi) 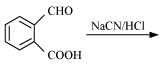
(vii) 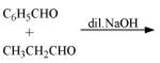
(viii) 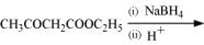
(ix) 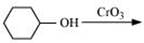
(x) 
(xi) 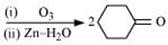
Ans: (i)
(ii)
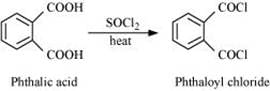
(iii)
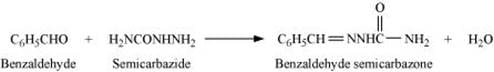
(iv)
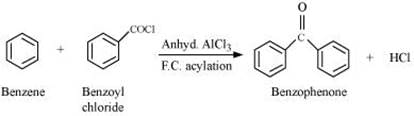
(v)
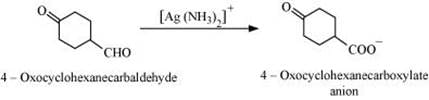
(vi)
(vii)
(viii)
(ix)
(x)
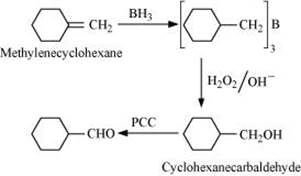
(xi)
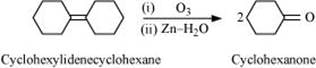
Q8.18: Give possible explanation for each of the following:
(i) Cyclohexanone forms cyanohydrin in good yield but 2, 2, 6 trimethylcyclohexanone does not.
(ii) There are two −NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones.
(iii) During the preparation of esters from a carboxylic acid and an alcohol in the presence of an acid catalyst, the water or the ester should be removed as soon as it is formed.
Ans: (i) Cyclohexanone form cyanohydrin according to the following equation.
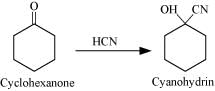
In this case, the nucleophile CN− can easily attack without any steric hindrance. However, in the case of 2, 2, 6 trimethylcydohexanone, methyl groups at α-positions offer steric hindrances and as a result, CN− cannot attack effectively.
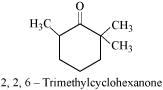
For this reason, it does not form a cyanohydrin.
(ii) Semicarbazide undergoes resonance involving only one of the two −NH2 groups, which is attached directly to the carbonyl-carbon atom.
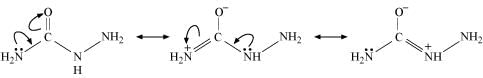
Therefore, the electron density on −NH2 group involved in the resonance also decreases. As a result, it cannot act as a nucleophile. Since the other −NH2 group is not involved in resonance; it can act as a nucleophile and can attack carbonyl-carbon atoms of aldehydes and ketones to produce semicarbazones.
(iii) Ester along with water is formed reversibly from a carboxylic acid and an alcohol in presence of an acid.
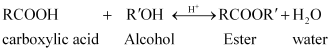
If either water or ester is not removed as soon as it is formed, then it reacts to give back the reactants as the reaction is reversible. Therefore, to shift the equilibrium in the forward direction i.e., to produce more ester, either of the two should be removed.
Q8.19: An organic compound contains 69.77% carbon, 11.63% hydrogen and rest oxygen. The molecular mass of the compound is 86. It does not reduce Tollens’ reagent but forms an addition compound with sodium hydrogensulphite and give positive iodoform test. On vigorous oxidation it gives ethanoic and propanoic acid. Write the possible structure of the compound.
Ans: % of carbon = 69.77 %
% of hydrogen = 11.63 %
% of oxygen = {100 − (69.77 11.63)}%
= 18.6 %
Thus, the ratio of the number of carbon, hydrogen, and oxygen atoms in the organic compound can be given as:
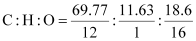
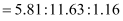
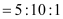
Therefore, the empirical formula of the compound is C5H10O. Now, the empirical formula mass of the compound can be given as:
5 × 12 10 ×1 1 × 16
= 86
Molecular mass of the compound = 86
Therefore, the molecular formula of the compound is given by C5H10O.
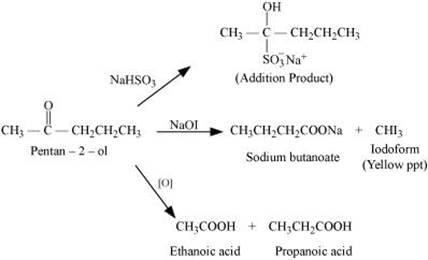
Since the given compound does not reduce Tollen’s reagent, it is not an aldehyde. Again, the compound forms sodium hydrogen sulphate addition products and gives a positive iodoform test. Since the compound is not an aldehyde, it must be a methyl ketone.
The given compound also gives a mixture of ethanoic acid and propanoic acid.
Hence, the given compound is pentan−2−ol.
The given reactions can be explained by the following equations:
Q8.20: Although phenoxide ion has more number of resonating structures than carboxylate ion, carboxylic acid is a stronger acid than phenol. Why?
Ans: Resonance structures of phenoxide ion are:
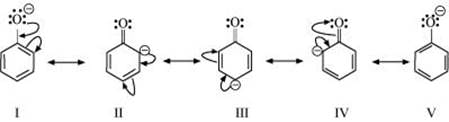
It can be observed from the resonance structures of phenoxide ion that in II, III and IV, less electronegative carbon atoms carry a negative charge. Therefore, these three structures contribute negligibly towards the resonance stability of the phenoxide ion. Hence, these structures can be eliminated. Only structures I and V carry a negative charge on the more electronegative oxygen atom.
Resonance structures of carboxylate ion are:
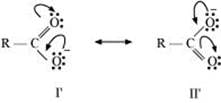
In the case of carboxylate ion, resonating structures I′ and II′ contain a charge carried by a more electronegative oxygen atom.
Further, in resonating structures I′ and II′, the negative charge is delocalized over two oxygen atoms. But in resonating structures I and V of the phexoxide ion, the negative charge is localized on the same oxygen atom. Therefore, the resonating structures of carboxylate ion contribute more towards its stability than those of phenoxide ion. As a result, carboxylate ion is more resonance-stabilized than phenoxide ion. Hence, carboxylic acid is a stronger acid than phenol.
|
75 videos|278 docs|78 tests
|
FAQs on NCERT Solutions Class 12 Chemistry Chapter 8 - Aldehydes, Ketones & Carboxylic Acids
| 1. What are some common examples of aldehydes? |  |
| 2. How are ketones different from aldehydes? |  |
| 3. What is the functional group present in carboxylic acids? |  |
| 4. How do aldehydes and ketones react with nucleophiles? |  |
| 5. Can carboxylic acids undergo decarboxylation reactions? |  |

















