All Exams >
ACT >
Science for ACT >
All Questions
All questions of Alcohols, Phenols and Ethers for ACT Exam
Which of the following statement is correct?- a)The -OH group in phenol is meta directing.
- b)The electron releasing groups increase the acidic character of phenols.
- c)Phenols are less acidic than aromatic alcohols.
- d)Boiling point of phenol is higher than that of toluene of comparable molecular mass.
Correct answer is option 'D'. Can you explain this answer?
Which of the following statement is correct?
a)
The -OH group in phenol is meta directing.
b)
The electron releasing groups increase the acidic character of phenols.
c)
Phenols are less acidic than aromatic alcohols.
d)
Boiling point of phenol is higher than that of toluene of comparable molecular mass.
|
|
Vivek Rana answered |
The correct answer is Option D.
Phenols have higher boiling point than toluene due to the presence of intermolecular hydrogen bonding in phenols. The formation of hydrogen bonds increases the intermolecular force of attraction between the phenol molecules and thereby increases its boiling point.
Identify the alcohol or phenol from the following which is most soluble in water.
- a)CH3– OH
- b)CH(CH3)2 CH2O-H
- c)CH3(CH2)2CH2OH
- d)C6H5OH
Correct answer is option 'A'. Can you explain this answer?
Identify the alcohol or phenol from the following which is most soluble in water.
a)
CH3– OH
b)
CH(CH3)2 CH2O-H
c)
CH3(CH2)2CH2OH
d)
C6H5OH

|
Aryan answered |
As...the ...linear chain in alcohol increase ...the solubility decrease .....due to ....staeric + vanderwall force inreacse ...
When CH2=CHCOOH is reduced with LiAlH4, the compound obtained will be [aieee-2003]- a)CH3CH2CH2OH
- b)CH3CH2CHO
- c)CH3CH2COOH
- d)CH2=CH-CH2OH
Correct answer is option 'D'. Can you explain this answer?
When CH2=CHCOOH is reduced with LiAlH4, the compound obtained will be [aieee-2003]
a)
CH3CH2CH2OH
b)
CH3CH2CHO
c)
CH3CH2COOH
d)
CH2=CH-CH2OH
|
|
Lavanya Menon answered |
The correct answer is option D
LiAlH4 can reduce the carboxylic acid group without affecting the double bond because alkene is electron-rich species.

LiAlH4 can reduce the carboxylic acid group without affecting the double bond because alkene is electron-rich species.

Nitration of anisole gives majorly:- a)Nitroanisole
- b)para-Nitroanisole
- c)ortho-Nitroanisole
- d)meta-Nitroanisole
Correct answer is option 'B'. Can you explain this answer?
Nitration of anisole gives majorly:
a)
Nitroanisole
b)
para-Nitroanisole
c)
ortho-Nitroanisole
d)
meta-Nitroanisole

|
Prasenjit Malik answered |
When anisole is nitrated with a mixture of conc HNO3 and H2SO4 it gives a mixture of ortho-Nitroanisole and para-Nitroanisole (major) products.


Can you explain the answer of this question below:The acidity of phenols is due to
- A:
Oxidation process
- B:
Resonance stabilization of its ions.
- C:
Hybridisation
- D:
Presence of O-H group
The answer is b.
The acidity of phenols is due to
Oxidation process
Resonance stabilization of its ions.
Hybridisation
Presence of O-H group
|
|
Rahul Bansal answered |
The acidity of phenols is due to its ability to lose hydrogen ion to form phenoxide ions. In a phenol molecule, the sp2hybridised carbon atom of benzene ring attached directly to the hydroxyl group acts as an electron withdrawing group. This sp2 hybridized carbon atom of benzene ring attached directly to the hydroxyl group has higher electronegativity in comparison to hydroxyl group. Due to the higher electronegativity of this carbon atom in comparison to the hydroxyl group attached, electron density decreases on oxygen atom. The decrease in electron density increases the polarity of O-H bond and results in the increase in ionization of phenols. Thus, the phenoxide ion is formed. The phenoxide ion formed is stabilized by the delocalization of negative charge due to the resonance in benzene ring. Phenoxide ion has greater stability than phenols, as in case of phenol charge separation takes place during resonance.The resonance structures of phenoxide ions explain the delocalization of negative charge. In case of substituted phenols, acidity of phenols increases in the presence of electron withdrawing group. This is due to the stability of the phenoxide ion generated. The acidity of phenols further increases if these groups are attached at ortho and para positions. This is due to the fact that the negative charge in phenoxide ion is mainly delocalized at ortho and para positions of the attached benzene ring. On the other hand, the acidity of phenols decreases in presence of electron donating groups as they prohibit the formation of phenoxide ion.
In the following sequence of reactions, CH3CH2OH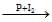 A
A 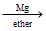 B
B  C
C 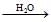 D, then compound `D' is - [AIEEE 2007]
D, then compound `D' is - [AIEEE 2007]- a)Butanal
- b)N-butyl alcohol
- c)N-propyl alcohol
- d)Propanal
Correct answer is option 'C'. Can you explain this answer?
In the following sequence of reactions, CH3CH2OH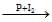 A
A 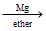 B
B  C
C 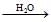 D, then compound `D' is - [AIEEE 2007]
D, then compound `D' is - [AIEEE 2007]
a)
Butanal
b)
N-butyl alcohol
c)
N-propyl alcohol
d)
Propanal
|
|
Nikita Singh answered |
Correct Answer is option C
When ethanol is treated with a mixture of phosphorus and iodine, ethyl iodide CH3CH2I is obtained.
Compound A on reaction with Mg metal in ether forms ethyl magnesium iodide CH3CH2 MgI.
The compound B reacts with formaldehyde to form compound C; CH3CH2−CH2−OMgX which on hydrolysis gives n propyl alcohol CH3CH2CH2−OH.
The compound D is n-propyl alcohol.
Hence, option C is correct.
When ethanol is treated with a mixture of phosphorus and iodine, ethyl iodide CH3CH2I is obtained.
Compound A on reaction with Mg metal in ether forms ethyl magnesium iodide CH3CH2 MgI.
The compound B reacts with formaldehyde to form compound C; CH3CH2−CH2−OMgX which on hydrolysis gives n propyl alcohol CH3CH2CH2−OH.
The compound D is n-propyl alcohol.
Hence, option C is correct.
The main product of the following reaction is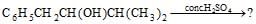 [AIEEE 2010]
[AIEEE 2010]- a)
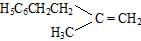
- b)
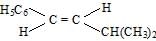
- c)
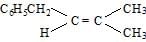
- d)
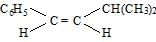
Correct answer is option 'B'. Can you explain this answer?
The main product of the following reaction is
a)
b)
c)
d)
|
|
Preeti Khanna answered |
Whenever dehydration can form two different alkenes major product is formed according to sides of rule that is more substituted alkene or alkene having lesson number of hydrogen atoms on the to double bonded carbon atoms is the major product.
Such reactions which can produce two or more structural isomers but one of them in Greater amount than the others are called ratio selective encasa reaction is hundred percent regioselective it is termed as reduce specific
In addition to being reduced selective alcohol dehydration are stereoselective reaction in which a single starting material can yield two or more stereoisomeric products but give one of them in Greater amount than any other


What is the product formed when excess of ethanol reacts with concentrated sulphuric acid at 383 K and then the temperature of reaction mixture is increased to 443 K?- a)Ethene
- b)Ethyl hydrogen sulphate
- c)Ethyl chloride
- d)Ethoxy ethane
Correct answer is 'A'. Can you explain this answer?
What is the product formed when excess of ethanol reacts with concentrated sulphuric acid at 383 K and then the temperature of reaction mixture is increased to 443 K?
a)
Ethene
b)
Ethyl hydrogen sulphate
c)
Ethyl chloride
d)
Ethoxy ethane
|
|
Rajeev Saxena answered |
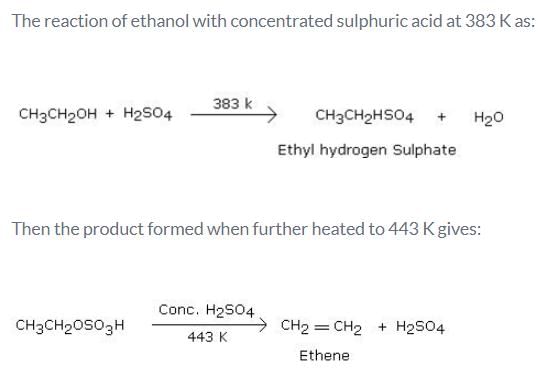
Identify the alcohol or phenol having a stronger acidic nature from the following.- a)CH3CH2OH
- b)C6H5OH
- c)CH3CHOHCH2CH3
- d)CH3CH2CH2CH2OH
Correct answer is option 'B'. Can you explain this answer?
Identify the alcohol or phenol having a stronger acidic nature from the following.
a)
CH3CH2OH
b)
C6H5OH
c)
CH3CHOHCH2CH3
d)
CH3CH2CH2CH2OH
|
|
Niti Mishra answered |
Alcohol and Phenol Acidity
Alcohols and phenols both contain a hydroxyl (-OH) group, which can act as an acid by donating a proton (H+). However, the acidity of alcohols and phenols varies depending on the structure of the molecule.
Factors Affecting Acidity
The acidity of alcohols and phenols depends on the following factors:
- Size of the alkyl group attached to the hydroxyl group
- Resonance stabilization of the conjugate base
- Inductive effect of adjacent functional groups
Strongest Acidic Nature
Out of the given options, C6H5OH (phenol) has the strongest acidic nature. This is because of the following reasons:
- Size of the alkyl group attached to the hydroxyl group: Phenol has a smaller alkyl group (C6H5) attached to the hydroxyl group than the other options, which have larger alkyl groups (CH3CH2, CH3CHOHCH2, and CH3CH2CH2CH2).
- Resonance stabilization of the conjugate base: Phenol's conjugate base (C6H5O-) is stabilized by resonance delocalization of the negative charge on the benzene ring. This makes the conjugate base more stable and the acid more acidic.
- Inductive effect of adjacent functional groups: The adjacent benzene ring in phenol also has an electron-withdrawing effect on the hydroxyl group, making it more acidic.
Therefore, phenol has the strongest acidic nature out of the given options.
Alcohols and phenols both contain a hydroxyl (-OH) group, which can act as an acid by donating a proton (H+). However, the acidity of alcohols and phenols varies depending on the structure of the molecule.
Factors Affecting Acidity
The acidity of alcohols and phenols depends on the following factors:
- Size of the alkyl group attached to the hydroxyl group
- Resonance stabilization of the conjugate base
- Inductive effect of adjacent functional groups
Strongest Acidic Nature
Out of the given options, C6H5OH (phenol) has the strongest acidic nature. This is because of the following reasons:
- Size of the alkyl group attached to the hydroxyl group: Phenol has a smaller alkyl group (C6H5) attached to the hydroxyl group than the other options, which have larger alkyl groups (CH3CH2, CH3CHOHCH2, and CH3CH2CH2CH2).
- Resonance stabilization of the conjugate base: Phenol's conjugate base (C6H5O-) is stabilized by resonance delocalization of the negative charge on the benzene ring. This makes the conjugate base more stable and the acid more acidic.
- Inductive effect of adjacent functional groups: The adjacent benzene ring in phenol also has an electron-withdrawing effect on the hydroxyl group, making it more acidic.
Therefore, phenol has the strongest acidic nature out of the given options.
Ethers may be used as solvents because they react only with which of the following reactants?- a)Acids
- b)Bases
- c)Oxidising agent
- d)Reducing agents
Correct answer is option 'A'. Can you explain this answer?
Ethers may be used as solvents because they react only with which of the following reactants?
a)
Acids
b)
Bases
c)
Oxidising agent
d)
Reducing agents
|
|
Geetika Shah answered |
Ethers resist the attack of nucleophiles and bases. However, they are very good solvents in many organic reactions due to their ability to solvate cations by donating the electron pair from oxygen atom. Ethers are generally less reactive and react only with acids.
One or More than One Options Correct TypeDirection (Q. Nos. 9-14) This section contains 6 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correctConsider the following reaction,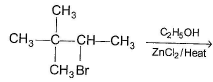 Q. The expected substitution product(s) is/are
Q. The expected substitution product(s) is/are- a)
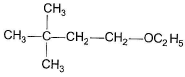
- b)
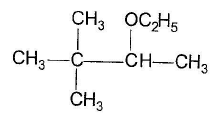
- c)
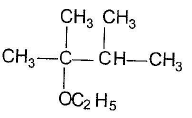
- d)
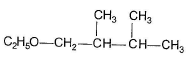
Correct answer is option 'B,C'. Can you explain this answer?
One or More than One Options Correct Type
Direction (Q. Nos. 9-14) This section contains 6 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct
Consider the following reaction,
Q.
The expected substitution product(s) is/are
a)
b)

c)

d)
|
|
Om Desai answered |
Reaction proceeds by SN1 mechanism.
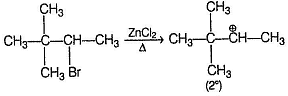
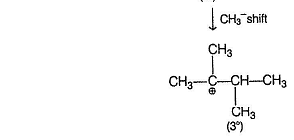
Above carbocations give the desired products.
Above carbocations give the desired products.
Williamsons synthesis is an example of :- a)Electrophilic substitution
- b)Electrophilic addition
- c)Nucleophilic substitution reaction
- d)Nucleophilic addition
Correct answer is option 'C'. Can you explain this answer?
Williamsons synthesis is an example of :
a)
Electrophilic substitution
b)
Electrophilic addition
c)
Nucleophilic substitution reaction
d)
Nucleophilic addition
|
|
Shreya Gupta answered |
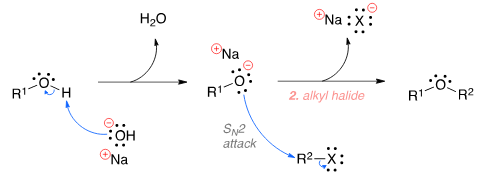
The Williamson ether synthesis is an organic reaction used to convert an alcohol and an alkyl halide to an ether using a base such as NaOH. The mechanism begins with the base abstracting the proton from the alcohol to form an alkoxide intermediate. The alkoxide then attacks the alkyl halide in a nucleophilic substi-tution reaction (SN2), which results in the formation of the final ether product and a metal halide by-product.

An ether is more volatile than an alcohol having the same molecular formula. This is due to- a)dipolar character of ethers
- b)alcohols having resonance structures
- c)inter-molecular hydrogen bonding in ethers
- d)inter-molecular hydrogen bonding in alcohols
Correct answer is option 'D'. Can you explain this answer?
An ether is more volatile than an alcohol having the same molecular formula. This is due to
a)
dipolar character of ethers
b)
alcohols having resonance structures
c)
inter-molecular hydrogen bonding in ethers
d)
inter-molecular hydrogen bonding in alcohols
|
|
Ashish Kumar Sahoo answered |
Inter molecular h bonding in alcohol makes its bond more stronger hence less volatile
Consider the following reaction :
C2H5OH + H2SO4 → Product Among the following which one cannot be formed as a product under any conditions ?
[AIEEE 2011]
- a) Ethylene
- b)Acetylene
- c)Dimethyl ether
- d)Ethyl-hydrogen sulphate
Correct answer is option 'B'. Can you explain this answer?
Consider the following reaction :
C2H5OH + H2SO4 → Product Among the following which one cannot be formed as a product under any conditions ?
[AIEEE 2011]
a)
Ethylene
b)
Acetylene
c)
Dimethyl ether
d)
Ethyl-hydrogen sulphate
|
|
Lavanya Menon answered |
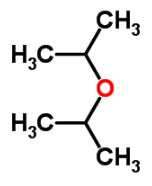
What is the IUPAC name of di-isopropyl ethera)Salicyaldehydeb)Ethyl acetatec)Iso propoxy propaned)Ethyl methyl etherCorrect answer is option 'C'. Can you explain this answer?
|
|
Shreya Gupta answered |
2-Iso propoxy propane
- Molecular Formula: C6H14O
- Average mass: 102.174797 Da
- Monoisotopic mass: 102.104462 Da

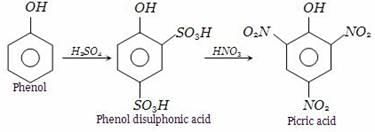
We can obtain picric acid from phenol by:- a)Sulphonation of phenol
- b)By Reimer Tiemann reaction
- c)Nitration of phenol
- d)Halogenation of phenol
Correct answer is option 'C'. Can you explain this answer?
We can obtain picric acid from phenol by:
a)
Sulphonation of phenol
b)
By Reimer Tiemann reaction
c)
Nitration of phenol
d)
Halogenation of phenol
|
|
Shreya Gupta answered |
Phenol heated with sulphuric acid gives phenol disulphonic acid, which further on reaction with nitric acid forms picric acid (2,4,6-trinitrophenol).
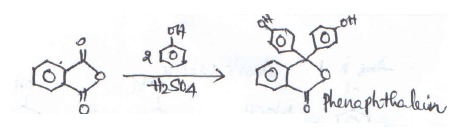
Phenol on treatment with Phthalic anhydride gives:- a)Phenolphthalein
- b)Salicylaldehyde
- c)Phthalic acid
- d)Salicylic acid
Correct answer is option 'A'. Can you explain this answer?
Phenol on treatment with Phthalic anhydride gives:
a)
Phenolphthalein
b)
Salicylaldehyde
c)
Phthalic acid
d)
Salicylic acid
|
|
Rohit Shah answered |
Formation of Phenolphthalein: When phenol is treated with a pthalic anhydride in the presence of concentrated H2SO4, it gives phenolphthalein, an indicator.
Phenols do not respond to which of these tests?- a)Schiff’s reagent test
- b)FeCl3 test
- c)Br2 water test
- d)Litmus test
Correct answer is option 'A'. Can you explain this answer?
Phenols do not respond to which of these tests?
a)
Schiff’s reagent test
b)
FeCl3 test
c)
Br2 water test
d)
Litmus test
|
|
Rajeev Saxena answered |
Phenols respond to all the above mentioned tests except Schiff’s reagent test, which is shown by aldehydes.
How many alcohols with molecular formula C4H10O are chiral in nature?a)2b)1c)4d)3Correct answer is option 'B'. Can you explain this answer?
|
|
Mansi Nair answered |

Here, again carbon is not chiral in nature.
So, only one alcohol is chiral in nature
So, only one alcohol is chiral in nature
An organic compound X is oxidised by using acidified K2Cr2O7. The product obtained reacts with Phenyl hydrazine but does not answer silver mirror test. The possible structure of X is- a)CH3CH2OH
- b)CH3CHO
- c)(CH3)2CHOH
- d)None of the these
Correct answer is option 'C'. Can you explain this answer?
An organic compound X is oxidised by using acidified K2Cr2O7. The product obtained reacts with Phenyl hydrazine but does not answer silver mirror test. The possible structure of X is
a)
CH3CH2OH
b)
CH3CHO
c)
(CH3)2CHOH
d)
None of the these
|
|
Baby Ghosh answered |
Yup...it will be propan-2-ol..by oxidation it..it will convert into acetone and it reacts with phenyl hydrazine,it produces acetone phenyl hydrazone and there is no aldehyde grp in acetone. so it doesn't react in silver mirror test means with tollen's reagent..
IUPAC name of m-cresol is ___________.- a)3-methylphenol
- b)3-chlorophenol
- c) 3-methoxyphenol
- d)benzene-1,3-diol
Correct answer is option 'A'. Can you explain this answer?
IUPAC name of m-cresol is ___________.
a)
3-methylphenol
b)
3-chlorophenol
c)
3-methoxyphenol
d)
benzene-1,3-diol
|
|
Preeti Iyer answered |
Meta-Cresol, also 3-methylphenol, is an organic compound with the formula CH3C6H4(OH). It is a colourless, viscous liquid that is used as an intermediate in the production of other chemicals. It is a derivative of phenol and is an isomer of p-cresol and o-cresol.
To prepare tert-butyl ethyl ether, the reagents required are: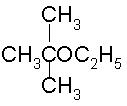
- a)Sodium ethoxide and tert-butyl bromide
- b)Sodium tert butoxide and ethyl bromide
- c)Dimethyl ketone, ethylbromide and sodium
- d)Sodium propoxide and propyl bromide
Correct answer is option 'B'. Can you explain this answer?
To prepare tert-butyl ethyl ether, the reagents required are:
a)
Sodium ethoxide and tert-butyl bromide
b)
Sodium tert butoxide and ethyl bromide
c)
Dimethyl ketone, ethylbromide and sodium
d)
Sodium propoxide and propyl bromide

|
Ayush Joshi answered |
Because 3degree haloalkanes like tert-butyl bromide (option a) give alkenes and not ethers when treated with a strong base like sodium ethoxide. So the exact answer is (b).
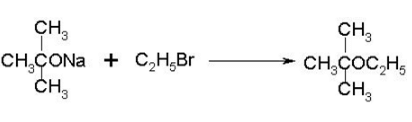
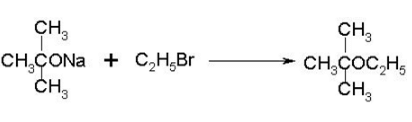
Isopropyl alcohol on oxidation forms:- a)Acetone
- b)Ether
- c)Acetaldehyde
- d)Methane
Correct answer is option 'A'. Can you explain this answer?
Isopropyl alcohol on oxidation forms:
a)
Acetone
b)
Ether
c)
Acetaldehyde
d)
Methane
|
|
Rahul Bansal answered |
The oxidation of isopropyl alcohol by potassium dichromate (K 2Cr 2O 7) gives acetone, the simplest ketone: Unlike aldehydes, ketones are relatively resistant to further oxidation, so no special precautions are required to isolate them as they form.
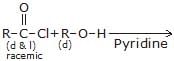 Product of above reaction is -
Product of above reaction is -- a)Enantiomer
- b)Racemic
- c)Diastereomers
- d)Meso
Correct answer is option 'C'. Can you explain this answer?
Product of above reaction is -
a)
Enantiomer
b)
Racemic
c)
Diastereomers
d)
Meso
|
|
Krishna Iyer answered |

HCl is formed in above reaction and react with pyridine and gives
Which catalyst is used in Fischer-Speier esterification?
- a)Concentrated H2SO4
- b)Dry HCl gas
- c)Concentrated HNO3
- d)Pyridine
Correct answer is option 'A'. Can you explain this answer?
Which catalyst is used in Fischer-Speier esterification?
a)
Concentrated H2SO4
b)
Dry HCl gas
c)
Concentrated HNO3
d)
Pyridine
|
|
Pooja Mehta answered |
Esterification is a relatively slow process at room temperature and does not proceed to completion. Concentrated sulfuric acid is used as a catalyst, and has a dual role: Speeds up the reaction. Acts as a dehydrating agent, forcing the equilibrium to the right and resulting in a greater yield of ester.
Which of the following compounds will react with sodium hydroxide solution in water?- a)C6H5OH
- b)C6H5CH2OH
- c)(CH3)3COH
- d)C2H5OH
Correct answer is option 'A'. Can you explain this answer?
Which of the following compounds will react with sodium hydroxide solution in water?
a)
C6H5OH
b)
C6H5CH2OH
c)
(CH3)3COH
d)
C2H5OH
|
|
Vijay Bansal answered |
When phenol reacts with sodium hydroxide solution it gives a colourless solution containing sodium phenoxide.
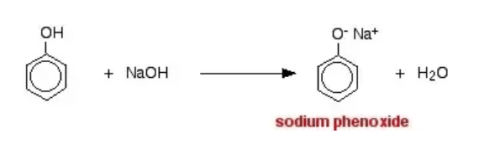
In this reaction, the hydrogen ion has been removed by the strongly basic hydroxide ion in the sodium hydroxide solution.
Which of the following compound reduces by NaBH4 ?- a)

- b)CH3–NO2
- c)
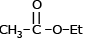
- d)
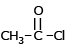
Correct answer is option 'D'. Can you explain this answer?
Which of the following compound reduces by NaBH4 ?
a)
b)
CH3–NO2
c)
d)
|
|
Suresh Iyer answered |
NaBH4 are weak reducing agent so only acid chloride are reduced in alcohal.
Phenol can be distinguished from ethanol by the reactions with _________.
- a)Br2/water
- b)Na
- c)Neutral FeCl3
- d)A and C
Correct answer is option 'D'. Can you explain this answer?
Phenol can be distinguished from ethanol by the reactions with _________.
a)
Br2/water
b)
Na
c)
Neutral FeCl3
d)
A and C

|
Nisha Banerjee answered |
- Phenol can be distinguished from ethanol using Br2/water and Neutral FeCl3.
- Br2/water: Phenol reacts with bromine water to give a white precipitate of 2,4,6-tribromophenol, while ethanol does not react.
- Neutral FeCl3: Phenol forms a violet complex with neutral ferric chloride, whereas ethanol shows no color change.
- Therefore, options A and C are correct for distinguishing phenol from ethanol.
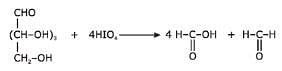
One Integer Value Correct TypeDirection (Q. Nos. 19-22) This section contains 4 questions. When worked out will result in an integer from 0 to 9 (both inclusive).In the following reaction,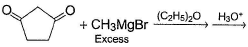 Q. How many different diols are formed as a result of nucleophilic addition reaction?
Q. How many different diols are formed as a result of nucleophilic addition reaction?
Correct answer is '3'. Can you explain this answer?
One Integer Value Correct Type
Direction (Q. Nos. 19-22) This section contains 4 questions. When worked out will result in an integer from 0 to 9 (both inclusive).
In the following reaction,
Q.
How many different diols are formed as a result of nucleophilic addition reaction?
|
|
Ritu Singh answered |
An alcohol has molecular formula C6H12O X and it gives immediate turbidity with cold, concentrated HCI even in the absence of ZnCI2. X can also be obtained by treatment of an ether with excess of CH3MgBr followed by acid hydrolysis. Hence, the correct statement regarding X is- a)It is 3-methyl-3-pentanol
- b)It is 2-methyl-3-pentanol
- c)It is 2-methyl-2-pentanol
- d)Either (b) or (c)
Correct answer is option 'C'. Can you explain this answer?
An alcohol has molecular formula C6H12O X and it gives immediate turbidity with cold, concentrated HCI even in the absence of ZnCI2. X can also be obtained by treatment of an ether with excess of CH3MgBr followed by acid hydrolysis. Hence, the correct statement regarding X is
a)
It is 3-methyl-3-pentanol
b)
It is 2-methyl-3-pentanol
c)
It is 2-methyl-2-pentanol
d)
Either (b) or (c)

|
Arpita Nair answered |
Which of the following compound reduces by DIBAL-H ?- a)CH3–
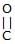 –OH
–OH - b)
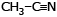
- c)CH3–
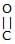 –O–Et
–O–Et - d)All
Correct answer is option 'D'. Can you explain this answer?
Which of the following compound reduces by DIBAL-H ?
a)
CH3–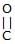 –OH
–OH
b)
c)
CH3–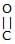 –O–Et
–O–Et
d)
All

|
Kiran Sanodiya answered |
Yes it can reduce ester ,acid n cyanide to corresponding aldehydes.
Which of the following is obtained as a major product in Friedel-Craft’s alkylation of phenol ?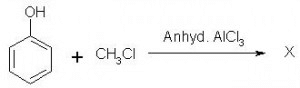
- a)
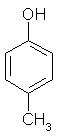
- b)
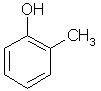
- c)
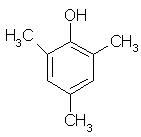
- d)
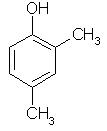
Correct answer is option 'A'. Can you explain this answer?
Which of the following is obtained as a major product in Friedel-Craft’s alkylation of phenol ?
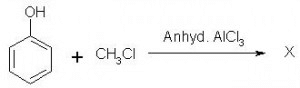
a)
b)
c)

d)

|
Kiran Sanodiya answered |
P-methyl phenol is the major product.
A is correct.
A is correct.
Chapter doubts & questions for Alcohols, Phenols and Ethers - Science for ACT 2025 is part of ACT exam preparation. The chapters have been prepared according to the ACT exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for ACT 2025 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of Alcohols, Phenols and Ethers - Science for ACT in English & Hindi are available as part of ACT exam.
Download more important topics, notes, lectures and mock test series for ACT Exam by signing up for free.
Science for ACT
486 videos|517 docs|337 tests
|
Signup to see your scores go up within 7 days!
Study with 1000+ FREE Docs, Videos & Tests
10M+ students study on EduRev