Class 10 Exam > Class 10 Questions > Fe2O3+ 2 Al → Al2O3+ 2 FeThe above react... Start Learning for Free
Fe2 O3 + 2 Al → Al2O3 + 2 Fe
The above reaction is an example of a
The above reaction is an example of a
- a)Combination reaction
- b)Double displacement reaction
- c)Displacement reaction
- d)Decomposition reaction
Correct answer is option 'C'. Can you explain this answer?
Most Upvoted Answer
Fe2O3+ 2 Al → Al2O3+ 2 FeThe above reaction is an example of aa)C...
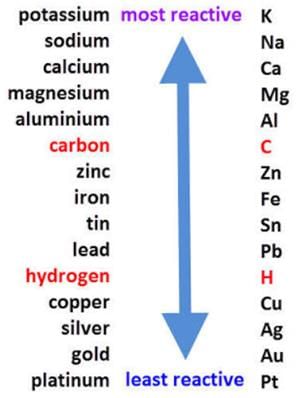
It is an example of displacement reaction. Aluminium being more reactive than iron displaces iron and results in the formation of aluminium oxide.

Free Test
FREE
| Start Free Test |
Community Answer
Fe2O3+ 2 Al → Al2O3+ 2 FeThe above reaction is an example of aa)C...
Understanding the Reaction
The reaction given is:
Fe2O3 + 2 Al → Al2O3 + 2 Fe
This is a significant chemical reaction known as a thermite reaction, which typically involves the reduction of metal oxides by aluminum.
What is a Displacement Reaction?
- A displacement reaction occurs when one element displaces another in a compound.
- In this case, aluminum (Al) displaces iron (Fe) from iron(III) oxide (Fe2O3).
Analysis of the Reaction
- Reactants: Iron(III) oxide (Fe2O3) and aluminum (Al).
- Products: Aluminum oxide (Al2O3) and iron (Fe).
Why is it a Displacement Reaction?
- Aluminum as a Reducing Agent: Aluminum is more reactive than iron, allowing it to replace iron in the compound.
- Change in Oxidation States: Iron in Fe2O3 is reduced (gains electrons), while aluminum is oxidized (loses electrons).
Types of Reactions Explained
- Combination Reaction: Two or more substances combine to form one product.
- Double Displacement Reaction: Exchange of ions between two compounds, typically in a solution.
- Decomposition Reaction: A single compound breaks down into two or more simpler substances.
Conclusion
Given that aluminum displaces iron from iron(III) oxide, this reaction is classified as a displacement reaction. Hence, the correct answer is option 'C'. This showcases the reactivity series of metals and highlights aluminum's ability to reduce metal oxides effectively.
The reaction given is:
Fe2O3 + 2 Al → Al2O3 + 2 Fe
This is a significant chemical reaction known as a thermite reaction, which typically involves the reduction of metal oxides by aluminum.
What is a Displacement Reaction?
- A displacement reaction occurs when one element displaces another in a compound.
- In this case, aluminum (Al) displaces iron (Fe) from iron(III) oxide (Fe2O3).
Analysis of the Reaction
- Reactants: Iron(III) oxide (Fe2O3) and aluminum (Al).
- Products: Aluminum oxide (Al2O3) and iron (Fe).
Why is it a Displacement Reaction?
- Aluminum as a Reducing Agent: Aluminum is more reactive than iron, allowing it to replace iron in the compound.
- Change in Oxidation States: Iron in Fe2O3 is reduced (gains electrons), while aluminum is oxidized (loses electrons).
Types of Reactions Explained
- Combination Reaction: Two or more substances combine to form one product.
- Double Displacement Reaction: Exchange of ions between two compounds, typically in a solution.
- Decomposition Reaction: A single compound breaks down into two or more simpler substances.
Conclusion
Given that aluminum displaces iron from iron(III) oxide, this reaction is classified as a displacement reaction. Hence, the correct answer is option 'C'. This showcases the reactivity series of metals and highlights aluminum's ability to reduce metal oxides effectively.
 | Explore Courses for Class 10 exam |  |
Question Description
Fe2O3+ 2 Al → Al2O3+ 2 FeThe above reaction is an example of aa)Combination reactionb)Double displacement reactionc)Displacement reactiond)Decomposition reactionCorrect answer is option 'C'. Can you explain this answer? for Class 10 2026 is part of Class 10 preparation. The Question and answers have been prepared according to the Class 10 exam syllabus. Information about Fe2O3+ 2 Al → Al2O3+ 2 FeThe above reaction is an example of aa)Combination reactionb)Double displacement reactionc)Displacement reactiond)Decomposition reactionCorrect answer is option 'C'. Can you explain this answer? covers all topics & solutions for Class 10 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for Fe2O3+ 2 Al → Al2O3+ 2 FeThe above reaction is an example of aa)Combination reactionb)Double displacement reactionc)Displacement reactiond)Decomposition reactionCorrect answer is option 'C'. Can you explain this answer?.
Fe2O3+ 2 Al → Al2O3+ 2 FeThe above reaction is an example of aa)Combination reactionb)Double displacement reactionc)Displacement reactiond)Decomposition reactionCorrect answer is option 'C'. Can you explain this answer? for Class 10 2026 is part of Class 10 preparation. The Question and answers have been prepared according to the Class 10 exam syllabus. Information about Fe2O3+ 2 Al → Al2O3+ 2 FeThe above reaction is an example of aa)Combination reactionb)Double displacement reactionc)Displacement reactiond)Decomposition reactionCorrect answer is option 'C'. Can you explain this answer? covers all topics & solutions for Class 10 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for Fe2O3+ 2 Al → Al2O3+ 2 FeThe above reaction is an example of aa)Combination reactionb)Double displacement reactionc)Displacement reactiond)Decomposition reactionCorrect answer is option 'C'. Can you explain this answer?.
Solutions for Fe2O3+ 2 Al → Al2O3+ 2 FeThe above reaction is an example of aa)Combination reactionb)Double displacement reactionc)Displacement reactiond)Decomposition reactionCorrect answer is option 'C'. Can you explain this answer? in English & in Hindi are available as part of our courses for Class 10. Download more important topics, notes, lectures and mock test series for Class 10 Exam by signing up for free.
Here you can find the meaning of Fe2O3+ 2 Al → Al2O3+ 2 FeThe above reaction is an example of aa)Combination reactionb)Double displacement reactionc)Displacement reactiond)Decomposition reactionCorrect answer is option 'C'. Can you explain this answer? defined & explained in the simplest way possible. Besides giving the explanation of Fe2O3+ 2 Al → Al2O3+ 2 FeThe above reaction is an example of aa)Combination reactionb)Double displacement reactionc)Displacement reactiond)Decomposition reactionCorrect answer is option 'C'. Can you explain this answer?, a detailed solution for Fe2O3+ 2 Al → Al2O3+ 2 FeThe above reaction is an example of aa)Combination reactionb)Double displacement reactionc)Displacement reactiond)Decomposition reactionCorrect answer is option 'C'. Can you explain this answer? has been provided alongside types of Fe2O3+ 2 Al → Al2O3+ 2 FeThe above reaction is an example of aa)Combination reactionb)Double displacement reactionc)Displacement reactiond)Decomposition reactionCorrect answer is option 'C'. Can you explain this answer? theory, EduRev gives you an ample number of questions to practice Fe2O3+ 2 Al → Al2O3+ 2 FeThe above reaction is an example of aa)Combination reactionb)Double displacement reactionc)Displacement reactiond)Decomposition reactionCorrect answer is option 'C'. Can you explain this answer? tests, examples and also practice Class 10 tests.
 | Explore Courses for Class 10 exam |  |
Top Courses for Class 10
Explore CoursesSignup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.






