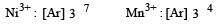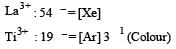All Exams >
NEET >
30-Day Revision Course for NEET >
All Questions
All questions of d- and f-Block Elements for NEET Exam
Transition metals with highest melting point is- a)Cr
- b)W
- c)Hg
- d)Sc
Correct answer is option 'B'. Can you explain this answer?
Transition metals with highest melting point is
a)
Cr
b)
W
c)
Hg
d)
Sc

|
Pragati Choudhury answered |
W belongs to 5d series and also it have lot of unpaired electrons thus it forms strong metallic bonding.
Ag+ ion is isoelectronic with- a)Zn2+
- b)Cd2+
- c)Pd2+
- d)Cu2+
Correct answer is option 'B'. Can you explain this answer?
Ag+ ion is isoelectronic with
a)
Zn2+
b)
Cd2+
c)
Pd2+
d)
Cu2+

|
Malavika Shah answered |
Ag+ is isoelectronic with Cd2+
Lucas reagent is
- a)ZnCl2 + HCl
- b)MnO2 + H2O
- c)H2SO4 + HCl
- d)NO + H2O
Correct answer is option 'A'. Can you explain this answer?
Lucas reagent is
a)
ZnCl2 + HCl
b)
MnO2 + H2O
c)
H2SO4 + HCl
d)
NO + H2O

|
Arshiya Choudhury answered |
Lucas Test is done to distinguish primary secondary and tertiary alcohols. Lucas reagent is ZnCl2 + HCl
Which of the following shows maximum number of oxidation states? [2002]- a)Cr
- b)Fe
- c)Mn
- d)V
Correct answer is option 'C'. Can you explain this answer?
Which of the following shows maximum number of oxidation states? [2002]
a)
Cr
b)
Fe
c)
Mn
d)
V

|
Arpita Tiwari answered |
Mn : [Ar] 3d5 4s2
Shows +2, +3, +4, +5, +6 & +7 oxidation states
Shows +2, +3, +4, +5, +6 & +7 oxidation states
For the four successive transition elements (Cr, Mn, Fe and Co), the stability of +2 oxidation state will be there in which of the following order?- a)Mn > Fe > Cr > Co [2011]
- b)Fe > Mn > Co > Cr
- c)Co > Mn > Fe > Cr
- d)Cr > Mn > Co > Fe
Correct answer is option 'A'. Can you explain this answer?
For the four successive transition elements (Cr, Mn, Fe and Co), the stability of +2 oxidation state will be there in which of the following order?
a)
Mn > Fe > Cr > Co [2011]
b)
Fe > Mn > Co > Cr
c)
Co > Mn > Fe > Cr
d)
Cr > Mn > Co > Fe
|
|
Kalyan Chavan answered |
B)Fe
c)Co
d)Cr
The correct answer is: d) Cr
The stability of the +2 oxidation state decreases as we move from Cr to Co. This is because the effective nuclear charge experienced by the valence electrons increases as we move from left to right in a period, making it more difficult for the element to lose electrons and form a positive ion. Additionally, the electron configuration of Cr2+ (3d4) is more stable than that of Mn2+ (3d5), Fe2+ (3d6), and Co2+ (3d7) due to half-filled and fully-filled subshells being more stable. Therefore, the stability of the +2 oxidation state follows the order Cr > Mn > Fe > Co.
c)Co
d)Cr
The correct answer is: d) Cr
The stability of the +2 oxidation state decreases as we move from Cr to Co. This is because the effective nuclear charge experienced by the valence electrons increases as we move from left to right in a period, making it more difficult for the element to lose electrons and form a positive ion. Additionally, the electron configuration of Cr2+ (3d4) is more stable than that of Mn2+ (3d5), Fe2+ (3d6), and Co2+ (3d7) due to half-filled and fully-filled subshells being more stable. Therefore, the stability of the +2 oxidation state follows the order Cr > Mn > Fe > Co.
The catalytic activity of transition metals and their compounds is ascribed mainly to :[2012 M]- a)their magnetic behaviour
- b)their unfilled d-orbitals
- c)their ability to adopt variable oxidation state
- d)their chemical reactivity
Correct answer is option 'C'. Can you explain this answer?
The catalytic activity of transition metals and their compounds is ascribed mainly to :[2012 M]
a)
their magnetic behaviour
b)
their unfilled d-orbitals
c)
their ability to adopt variable oxidation state
d)
their chemical reactivity

|
Maheshwar Saini answered |
The transition metals and their compounds are used as catalysts. Because of the variable oxidation states, due to this, they easily absorb and re-emit wide range of energy to provide the necessary activation energy.
Mohr’s salt is a- a)Acidic salt
- b)Double salt
- c)Basic Acidic salt
- d)Normal salt
Correct answer is option 'B'. Can you explain this answer?
Mohr’s salt is a
a)
Acidic salt
b)
Double salt
c)
Basic Acidic salt
d)
Normal salt

|
Amar Pillai answered |
Mohr salt is an example of double salt. Mohr Salt is
FeSO4 . (NH4)2 SO4.6H2O
Which of the following pairs has the same size?a)Fe2+ , Ni 2+b)Zr 4+ , Ti4+ c)Zr 4+ , Hf 4+d)Zn 2+ , Hf 4+Correct answer is option 'C'. Can you explain this answer?

|
Prasenjit Pillai answered |
Due to lanthanide contraction, the size of Zr and Hf (atom and ions) become nearly similar
Which of the following ion has smallest radii?- a)V2+
- b)Ni2+
- c)Mn2+
- d)Ti2+
Correct answer is option 'B'. Can you explain this answer?
Which of the following ion has smallest radii?
a)
V2+
b)
Ni2+
c)
Mn2+
d)
Ti2+

|
Preethi Bose answered |
In period when we move from left to right in a period atomic radii 1st decreases till nickel then starts increasing so Ni2+ has smallest radii.
A reduction in the atomic size with increase in atomic number is characteristic of the elements of- a)high atomic masses
- b)d – block
- c)f – block
- d)radioactive series
Correct answer is option 'C'. Can you explain this answer?
A reduction in the atomic size with increase in atomic number is characteristic of the elements of
a)
high atomic masses
b)
d – block
c)
f – block
d)
radioactive series

|
Nidhi Nambiar answered |
In f block elements with increase in atomic number atomic radii decreases smoothly.
The correctorder of decreasing second ionisation enthalpy of Ti (22), V(23), Cr(24) and Mn (25) is : [2008]- a)Cr > Mn > V > Ti
- b)V > Mn > Cr > Ti
- c)Mn > Cr > Ti > V
- d)Ti > V > Cr > Mn
Correct answer is option 'A'. Can you explain this answer?
The correctorder of decreasing second ionisation enthalpy of Ti (22), V(23), Cr(24) and Mn (25) is : [2008]
a)
Cr > Mn > V > Ti
b)
V > Mn > Cr > Ti
c)
Mn > Cr > Ti > V
d)
Ti > V > Cr > Mn

|
Nilotpal Gupta answered |
Ti ; Z (22) is 1s22s22p63s23p64s23d2
V ; Z (23) is 1s22s22p63s23p64s23d3
Cr ; Z (24) is 1s22s22p63s23p63d54s1
Mn ; Z (25) is 1s22s22p63s23d54s2
V ; Z (23) is 1s22s22p63s23p64s23d3
Cr ; Z (24) is 1s22s22p63s23p63d54s1
Mn ; Z (25) is 1s22s22p63s23d54s2
The second electron in all the cases (except Cr) is taken out from 4s-orbital and for Cr it is an electron from completely half filled 3d-orbital. The force required for removal of second electron will be more for Mn (except for Cr) having more positive charge.
Based on this we find the correct order is Mn > V > Ti. Cr > Mn > V > Ti. So correct answeris (a).
Based on this we find the correct order is Mn > V > Ti. Cr > Mn > V > Ti. So correct answeris (a).
Which is called chromic acid?- a)Cr2O3
- b)Cr3O4
- c)CrO
- d)H2CrO4
Correct answer is option 'D'. Can you explain this answer?
Which is called chromic acid?
a)
Cr2O3
b)
Cr3O4
c)
CrO
d)
H2CrO4
|
|
Shraddha Dey answered |
Chromic Acid is a naturally occurring oxide with a formula H2CrO4.
In which of the following pairs are both the ions coloured in aqueous solutions ? [2006] (At. no. : Sc = 21, Ti = 22, Ni = 28, Cu = 29, Co = 27)- a)Sc3+, Ti3+
- b)Sc3+, Co2+
- c)Ni2+, Cu+
- d)Ni2+, Ti3+
Correct answer is option 'D'. Can you explain this answer?
In which of the following pairs are both the ions coloured in aqueous solutions ? [2006] (At. no. : Sc = 21, Ti = 22, Ni = 28, Cu = 29, Co = 27)
a)
Sc3+, Ti3+
b)
Sc3+, Co2+
c)
Ni2+, Cu+
d)
Ni2+, Ti3+

|
Dipanjan Mehta answered |
Sc3+ : 1s2, 2s2p6, 3s2p6d0, 4s0; no unpaired electron.
Cu+ : 1s2, 2s2p6, 3s2p6d10, 4s0; no unpaired electron.
Ni2+: 1s2, 2s2p6, 3s2p6d8, 4s0; unpaired electron present.
Ti3+ : 1s2, 2s2p6, 2s2p6d1, 4s0; unpaired electron present Co2+ : 1s2, 2s2p6, 3s2p6d7, 4s0; unpaired electron present So from the given option the only correct combination is Ni2+ and Ti3+
Cu+ : 1s2, 2s2p6, 3s2p6d10, 4s0; no unpaired electron.
Ni2+: 1s2, 2s2p6, 3s2p6d8, 4s0; unpaired electron present.
Ti3+ : 1s2, 2s2p6, 2s2p6d1, 4s0; unpaired electron present Co2+ : 1s2, 2s2p6, 3s2p6d7, 4s0; unpaired electron present So from the given option the only correct combination is Ni2+ and Ti3+
Ferrous sulphate on heating gives- a)SO2 and SO3
- b)SO2 and O2
- c)SO2
- d)SO3
Correct answer is option 'A'. Can you explain this answer?
Ferrous sulphate on heating gives
a)
SO2 and SO3
b)
SO2 and O2
c)
SO2
d)
SO3

|
M. Vishnu answered |
On heating, ferrous sulphate crystals lose water and anhydrous ferrous sulphate (FeSO4) is formed. So their colour changes from light green to white. On furtherheating, anhydrous ferrous sulphate decomposes to form ferric oxide (Fe2O3), sulphur dioxide (SO2) and sulphur trioxide (SO3).
Lanthanoids are [2004]- a)14 elements in the sixth period (atomic no. = 90 to 103) that are filling 4f sublevel
- b)14 elements in the seventh period (atomic no. = 90 to 103) that are filling 5f sublevel
- c)14 elements in the sixth period (atomic no. = 58 to 71) that are filling 4f sublevel
- d)14 elements in the seventh period (atomic no. = 58 to 71) that are filling 4f sublevel
Correct answer is option 'C'. Can you explain this answer?
Lanthanoids are [2004]
a)
14 elements in the sixth period (atomic no. = 90 to 103) that are filling 4f sublevel
b)
14 elements in the seventh period (atomic no. = 90 to 103) that are filling 5f sublevel
c)
14 elements in the sixth period (atomic no. = 58 to 71) that are filling 4f sublevel
d)
14 elements in the seventh period (atomic no. = 58 to 71) that are filling 4f sublevel

|
Athira Datta answered |
The correct answer is option C: 14 elements in the sixth period (atomic no. = 58 to 71) that are filling the 4f sublevel.
Explanation:
- The periodic table is organized into periods and groups. The periods are horizontal rows, and the groups are vertical columns.
- The lanthanoids, also known as the lanthanides, are a group of elements that belong to the f-block of the periodic table.
- The lanthanoid series consists of 14 elements that fill the 4f sublevel. These elements have atomic numbers ranging from 58 to 71.
- The 4f sublevel is part of the fourth energy level (n=4) and is characterized by its unique electron configuration.
- In the lanthanoid series, the 4f sublevel is gradually filled up as you move across the period. Each element in the series adds one more electron to the 4f sublevel until it is completely filled.
- The lanthanoids are located in the sixth period of the periodic table because they correspond to the filling of the 4f sublevel.
- The lanthanoids are known for their similar chemical properties, which arise from the filling of the 4f sublevel. They are generally soft, malleable metals with high melting points and good electrical conductivity.
- Some common examples of lanthanoids include cerium (Ce), praseodymium (Pr), and europium (Eu).
- It's important to note that the atomic numbers of the lanthanoids range from 58 to 71, not from 90 to 103 as mentioned in options A and B. The atomic numbers from 90 to 103 correspond to a different series of elements called the actinoids, which fill the 5f sublevel.
- Therefore, the correct answer is option C: 14 elements in the sixth period (atomic no. = 58 to 71) that are filling the 4f sublevel.
Explanation:
- The periodic table is organized into periods and groups. The periods are horizontal rows, and the groups are vertical columns.
- The lanthanoids, also known as the lanthanides, are a group of elements that belong to the f-block of the periodic table.
- The lanthanoid series consists of 14 elements that fill the 4f sublevel. These elements have atomic numbers ranging from 58 to 71.
- The 4f sublevel is part of the fourth energy level (n=4) and is characterized by its unique electron configuration.
- In the lanthanoid series, the 4f sublevel is gradually filled up as you move across the period. Each element in the series adds one more electron to the 4f sublevel until it is completely filled.
- The lanthanoids are located in the sixth period of the periodic table because they correspond to the filling of the 4f sublevel.
- The lanthanoids are known for their similar chemical properties, which arise from the filling of the 4f sublevel. They are generally soft, malleable metals with high melting points and good electrical conductivity.
- Some common examples of lanthanoids include cerium (Ce), praseodymium (Pr), and europium (Eu).
- It's important to note that the atomic numbers of the lanthanoids range from 58 to 71, not from 90 to 103 as mentioned in options A and B. The atomic numbers from 90 to 103 correspond to a different series of elements called the actinoids, which fill the 5f sublevel.
- Therefore, the correct answer is option C: 14 elements in the sixth period (atomic no. = 58 to 71) that are filling the 4f sublevel.
Four successive members of the first row transition elements are listed below with their atomic numbers. Which one of them is expected to have the highest third ionization enthalpy? [2 00 5]- a)Vanadium (Z = 23)
- b)Chromium (Z = 24)
- c)Man gan ese (Z = 25)
- d)Ir on (Z = 26)
Correct answer is option 'C'. Can you explain this answer?
Four successive members of the first row transition elements are listed below with their atomic numbers. Which one of them is expected to have the highest third ionization enthalpy? [2 00 5]
a)
Vanadium (Z = 23)
b)
Chromium (Z = 24)
c)
Man gan ese (Z = 25)
d)
Ir on (Z = 26)

|
Ashwini Khanna answered |
For third ion ization enthalpy last configuration of
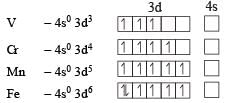
For third Ionization enthalpy Mn has stable configuration due to half filled d-orbital.
Copper sulphate dissolves in excess of KCN to give[2006]- a)[Cu(CN)4]3–
- b)[Cu(CN)4]2–
- c)Cu(CN)2
- d)CuCN
Correct answer is option 'A'. Can you explain this answer?
Copper sulphate dissolves in excess of KCN to give[2006]
a)
[Cu(CN)4]3–
b)
[Cu(CN)4]2–
c)
Cu(CN)2
d)
CuCN

|
Rajeev Sharma answered |
Copper sulphate on treatment with excess of KCN forms complex K3[Cu(CN)4] or
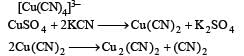

Maximum magnetic moment is shown by- a)d6
- b)d8
- c)d5
- d)d7
Correct answer is option 'C'. Can you explain this answer?
Maximum magnetic moment is shown by
a)
d6
b)
d8
c)
d5
d)
d7
|
|
Shraddha Chavan answered |
Magnetic Moment and d-orbitals
To understand why the maximum magnetic moment is shown by d5, we need to understand the concept of magnetic moment and the behavior of d-orbitals in transition metals.
Magnetic Moment
Magnetic moment is a measure of the strength and orientation of a magnet in a magnetic field. In the context of transition metals, it refers to the magnetic properties of unpaired electrons in the d-orbitals. The magnetic moment can be calculated using the formula:
Magnetic moment (μ) = √[n(n+2)] BM
Where n is the number of unpaired electrons and BM is the Bohr magneton, a unit of magnetic moment.
d-orbitals
d-orbitals are the five orbitals (dxy, dxz, dyz, dx²-y², dz²) available for electrons to occupy in the d-subshell. These orbitals have different shapes and orientations, and they can accommodate a maximum of 10 electrons.
Explanation
In the given options, the maximum magnetic moment is shown by d5. Let's analyze each option to understand why d5 has the highest magnetic moment.
- d6: In d6 configuration, there are 4 unpaired electrons. Applying the magnetic moment formula, we get μ = √[4(4+2)] BM = √24 BM.
- d8: In d8 configuration, there are 2 unpaired electrons. Applying the magnetic moment formula, we get μ = √[2(2+2)] BM = √8 BM.
- d5: In d5 configuration, there are 5 unpaired electrons. Applying the magnetic moment formula, we get μ = √[5(5+2)] BM = √35 BM.
- d7: In d7 configuration, there are 3 unpaired electrons. Applying the magnetic moment formula, we get μ = √[3(3+2)] BM = √15 BM.
Comparing the magnetic moments calculated for each option, we can see that d5 has the highest magnetic moment of √35 BM. Therefore, the correct answer is option 'C'.
It is important to note that the magnetic moment depends on the number of unpaired electrons. The more unpaired electrons present in the d-orbitals, the higher the magnetic moment. This is because unpaired electrons have individual magnetic moments that align with an external magnetic field, resulting in a stronger overall magnetic moment.
Which of the following pairs of ions have same paramagnetic moment?- a)Cu2+, Ti3+
- b)Ti3+, Ni2+
- c)Ti4+, Cu2+
- d)Mn2+, Cu2+
Correct answer is option 'A'. Can you explain this answer?
Which of the following pairs of ions have same paramagnetic moment?
a)
Cu2+, Ti3+
b)
Ti3+, Ni2+
c)
Ti4+, Cu2+
d)
Mn2+, Cu2+
|
|
Ameya Pillai answered |
Explanation:
Paramagnetic Moment:
Paramagnetic substances are those substances that are weakly attracted by an external magnetic field. The paramagnetic moment is a measure of the strength of this attraction. It is given by the formula:
Paramagnetic Moment (μ) = √(n(n+2)) BM
Where:
- n is the number of unpaired electrons in the atom or ion
- BM stands for Bohr Magneton, a unit of magnetic moment.
Identifying the Number of Unpaired Electrons:
To determine which pairs of ions have the same paramagnetic moment, we need to first identify the number of unpaired electrons in each ion. Unpaired electrons are those that do not have a partner or are not paired with another electron in the same orbital.
Calculating the Number of Unpaired Electrons:
To calculate the number of unpaired electrons, we need to know the electron configuration of each ion. Let's calculate the number of unpaired electrons for each pair of ions:
a) Cu2+ (Copper ion with +2 charge)
- The electron configuration of Cu2+ is: 1s2 2s2 2p6 3s2 3p6 3d9
- In Cu2+, there are 9 electrons in the 3d orbital, which means there is 1 unpaired electron.
b) Ti3+ (Titanium ion with +3 charge)
- The electron configuration of Ti3+ is: 1s2 2s2 2p6 3s2 3p6 3d1
- In Ti3+, there is 1 electron in the 3d orbital, which means there is 1 unpaired electron.
c) Ti4+ (Titanium ion with +4 charge)
- The electron configuration of Ti4+ is: 1s2 2s2 2p6 3s2 3p6
- In Ti4+, all the electrons are paired, which means there are no unpaired electrons.
d) Mn2+ (Manganese ion with +2 charge)
- The electron configuration of Mn2+ is: 1s2 2s2 2p6 3s2 3p6 3d5
- In Mn2+, there are 5 electrons in the 3d orbital, which means there are 5 unpaired electrons.
Comparing the Number of Unpaired Electrons:
From the calculations above, we can see that:
- Cu2+ and Ti3+ both have 1 unpaired electron.
- Ti4+ and Mn2+ have 0 unpaired electrons.
Conclusion:
Based on the number of unpaired electrons, we can conclude that the pair of ions with the same paramagnetic moment is:
a) Cu2+ (1 unpaired electron)
b) Ti3+ (1 unpaired electron)
Paramagnetic Moment:
Paramagnetic substances are those substances that are weakly attracted by an external magnetic field. The paramagnetic moment is a measure of the strength of this attraction. It is given by the formula:
Paramagnetic Moment (μ) = √(n(n+2)) BM
Where:
- n is the number of unpaired electrons in the atom or ion
- BM stands for Bohr Magneton, a unit of magnetic moment.
Identifying the Number of Unpaired Electrons:
To determine which pairs of ions have the same paramagnetic moment, we need to first identify the number of unpaired electrons in each ion. Unpaired electrons are those that do not have a partner or are not paired with another electron in the same orbital.
Calculating the Number of Unpaired Electrons:
To calculate the number of unpaired electrons, we need to know the electron configuration of each ion. Let's calculate the number of unpaired electrons for each pair of ions:
a) Cu2+ (Copper ion with +2 charge)
- The electron configuration of Cu2+ is: 1s2 2s2 2p6 3s2 3p6 3d9
- In Cu2+, there are 9 electrons in the 3d orbital, which means there is 1 unpaired electron.
b) Ti3+ (Titanium ion with +3 charge)
- The electron configuration of Ti3+ is: 1s2 2s2 2p6 3s2 3p6 3d1
- In Ti3+, there is 1 electron in the 3d orbital, which means there is 1 unpaired electron.
c) Ti4+ (Titanium ion with +4 charge)
- The electron configuration of Ti4+ is: 1s2 2s2 2p6 3s2 3p6
- In Ti4+, all the electrons are paired, which means there are no unpaired electrons.
d) Mn2+ (Manganese ion with +2 charge)
- The electron configuration of Mn2+ is: 1s2 2s2 2p6 3s2 3p6 3d5
- In Mn2+, there are 5 electrons in the 3d orbital, which means there are 5 unpaired electrons.
Comparing the Number of Unpaired Electrons:
From the calculations above, we can see that:
- Cu2+ and Ti3+ both have 1 unpaired electron.
- Ti4+ and Mn2+ have 0 unpaired electrons.
Conclusion:
Based on the number of unpaired electrons, we can conclude that the pair of ions with the same paramagnetic moment is:
a) Cu2+ (1 unpaired electron)
b) Ti3+ (1 unpaired electron)
Which one of the following combines with Fe2+ ion to form a brown complex?- a)N2O
- b)N2O3
- c)N2O5
- d)NO
Correct answer is option 'D'. Can you explain this answer?
Which one of the following combines with Fe2+ ion to form a brown complex?
a)
N2O
b)
N2O3
c)
N2O5
d)
NO

|
Sankar Chakraborty answered |
[Fe(H2O)5NO]2+is brown color complex.
Which among the following transition metal has lowest melting point?- a)Titanium
- b)Cobalt
- c)Mercury
- d)Scandium
Correct answer is option 'C'. Can you explain this answer?
Which among the following transition metal has lowest melting point?
a)
Titanium
b)
Cobalt
c)
Mercury
d)
Scandium
|
|
Srestha Patel answered |
Understanding Melting Points of d-Block Elements
The melting points of d-block elements vary significantly due to their unique electron configurations and bonding characteristics. Among the options provided, we will analyze why mercury has the lowest melting point.
Factors Influencing Melting Points
- Electron Configuration: Transition metals have varying numbers of d-electrons, influencing their bonding and stability.
- Metallic Bonding: Stronger metallic bonds generally lead to higher melting points. The presence of d-electrons contributes to the strength of these bonds.
- Atomic Structure: The arrangement of atoms and the presence of any additional forces (like van der Waals forces) can also affect melting points.
Analysis of Each Element
- Titanium (Ti):
- Has a melting point of approximately 1668°C.
- Strong metallic bonds due to a larger number of d-electrons.
- Cobalt (Co):
- Melting point around 1495°C.
- Exhibits significant metallic bonding strength.
- Scandium (Sc):
- Melting point is about 1541°C.
- More stable due to its electron configuration but still has a higher melting point than mercury.
- Mercury (Hg):
- Notably has a melting point of -38.83°C.
- Exists as a liquid at room temperature, indicating weak metallic bonding and minimal d-electron participation in bonding.
Conclusion
Mercury's unique properties, including its liquid state at room temperature and weak metallic bonds, result in its significantly lower melting point compared to titanium, cobalt, and scandium. Thus, the correct answer is option 'C', mercury, which has the lowest melting point among the listed d-block elements.
The melting points of d-block elements vary significantly due to their unique electron configurations and bonding characteristics. Among the options provided, we will analyze why mercury has the lowest melting point.
Factors Influencing Melting Points
- Electron Configuration: Transition metals have varying numbers of d-electrons, influencing their bonding and stability.
- Metallic Bonding: Stronger metallic bonds generally lead to higher melting points. The presence of d-electrons contributes to the strength of these bonds.
- Atomic Structure: The arrangement of atoms and the presence of any additional forces (like van der Waals forces) can also affect melting points.
Analysis of Each Element
- Titanium (Ti):
- Has a melting point of approximately 1668°C.
- Strong metallic bonds due to a larger number of d-electrons.
- Cobalt (Co):
- Melting point around 1495°C.
- Exhibits significant metallic bonding strength.
- Scandium (Sc):
- Melting point is about 1541°C.
- More stable due to its electron configuration but still has a higher melting point than mercury.
- Mercury (Hg):
- Notably has a melting point of -38.83°C.
- Exists as a liquid at room temperature, indicating weak metallic bonding and minimal d-electron participation in bonding.
Conclusion
Mercury's unique properties, including its liquid state at room temperature and weak metallic bonds, result in its significantly lower melting point compared to titanium, cobalt, and scandium. Thus, the correct answer is option 'C', mercury, which has the lowest melting point among the listed d-block elements.
The aqueous solution containing which one of the following ions will be colourless? (Atomic number: Sc = 21, Fe = 26, Ti = 22, Mn = 25)[2005]- a)Sc3+
- b)Fe2+
- c)Ti3+
- d)Mn 2+
Correct answer is option 'A'. Can you explain this answer?
The aqueous solution containing which one of the following ions will be colourless? (Atomic number: Sc = 21, Fe = 26, Ti = 22, Mn = 25)[2005]
a)
Sc3+
b)
Fe2+
c)
Ti3+
d)
Mn 2+

|
Maheshwar Saini answered |
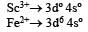

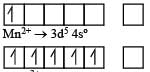
In Sc3+ there as no unpaired electron So the aqueous solution of Sc3+ will be colourless.
The main reason for larger number of oxidation states exhibited by the actinoids than the corresponding lanthanoids, is [2005, 2006]- a)more energy difference between 5f and 6d orbitals than between 4f and 5d orbitals.
- b)lesser energy difference between 5f and 6d orbitals than between 4f and 5d orbitals.
- c)larger atomic size of actinoids than the lanthanoids.
- d)greater reactive nature of the actinoids than the lanthanoids.
Correct answer is option 'B'. Can you explain this answer?
The main reason for larger number of oxidation states exhibited by the actinoids than the corresponding lanthanoids, is [2005, 2006]
a)
more energy difference between 5f and 6d orbitals than between 4f and 5d orbitals.
b)
lesser energy difference between 5f and 6d orbitals than between 4f and 5d orbitals.
c)
larger atomic size of actinoids than the lanthanoids.
d)
greater reactive nature of the actinoids than the lanthanoids.

|
Nilotpal Gupta answered |
The cause of showing different oxidation is due to the fact that there is only a small difference between the energies of electron in the ns orbitals and (n – 1)d orbitals with the result both ns as well (n – 1)d electrons can be used for compound formation. Lesser energy difference between 5f and 6d orbitals than between 4f and 5d orbitals result in larger no. of oxidation state.
Which of the following methods is based on distribution law?- a)Parke’s process
- b)Poling process
- c)Mond’s process
- d)Cupellation process
Correct answer is option 'A'. Can you explain this answer?
Which of the following methods is based on distribution law?
a)
Parke’s process
b)
Poling process
c)
Mond’s process
d)
Cupellation process

|
Saptarshi Ghoshal answered |
Parkes process is a process for removing silver from Pb. It is an example of Liquid liquid extractions
Which of the following oxidation states are the most characteristic for lead and tin respectively?- a)+ 2, + 4
- b)+ 4, + 4 [2007]
- c)+ 2, + 2
- d)+ 4, + 2
Correct answer is option 'A'. Can you explain this answer?
Which of the following oxidation states are the most characteristic for lead and tin respectively?
a)
+ 2, + 4
b)
+ 4, + 4 [2007]
c)
+ 2, + 2
d)
+ 4, + 2

|
Prasenjit Malik answered |
The oxidation states of lead and tin can vary depending on the compound they are present in. However, there are certain oxidation states that are more characteristic for these elements.
Lead:
- The most characteristic oxidation state for lead is +2. This means that lead can lose two electrons to form a 2+ cation. This oxidation state is commonly observed in lead compounds such as lead(II) oxide (PbO), lead(II) chloride (PbCl2), and lead(II) nitrate (Pb(NO3)2).
Tin:
- The most characteristic oxidation state for tin is +4. This means that tin can lose four electrons to form a 4+ cation. This oxidation state is commonly observed in tin compounds such as tin(IV) oxide (SnO2), tin(IV) chloride (SnCl4), and tin(IV) sulfate (Sn(SO4)2).
Explanation:
- The oxidation state of an element refers to the charge that element would have if all its bonds were ionic. It is a measure of the electron gain or loss of an atom in a compound.
- In the case of lead, it has a tendency to lose two electrons to achieve a stable configuration. This results in its most characteristic oxidation state of +2.
- Similarly, tin has a tendency to lose four electrons to achieve a stable configuration. This results in its most characteristic oxidation state of +4.
- It is important to note that lead and tin can also exhibit other oxidation states depending on the compound they are present in. For example, lead can also exist in the +4 oxidation state in compounds such as lead(IV) oxide (PbO2), while tin can also exist in the +2 oxidation state in compounds such as tin(II) chloride (SnCl2).
- However, the most characteristic oxidation states for lead and tin respectively are +2 and +4.
- Therefore, the correct answer is option A, which states that the most characteristic oxidation states for lead and tin are 2 and 4 respectively.
Lead:
- The most characteristic oxidation state for lead is +2. This means that lead can lose two electrons to form a 2+ cation. This oxidation state is commonly observed in lead compounds such as lead(II) oxide (PbO), lead(II) chloride (PbCl2), and lead(II) nitrate (Pb(NO3)2).
Tin:
- The most characteristic oxidation state for tin is +4. This means that tin can lose four electrons to form a 4+ cation. This oxidation state is commonly observed in tin compounds such as tin(IV) oxide (SnO2), tin(IV) chloride (SnCl4), and tin(IV) sulfate (Sn(SO4)2).
Explanation:
- The oxidation state of an element refers to the charge that element would have if all its bonds were ionic. It is a measure of the electron gain or loss of an atom in a compound.
- In the case of lead, it has a tendency to lose two electrons to achieve a stable configuration. This results in its most characteristic oxidation state of +2.
- Similarly, tin has a tendency to lose four electrons to achieve a stable configuration. This results in its most characteristic oxidation state of +4.
- It is important to note that lead and tin can also exhibit other oxidation states depending on the compound they are present in. For example, lead can also exist in the +4 oxidation state in compounds such as lead(IV) oxide (PbO2), while tin can also exist in the +2 oxidation state in compounds such as tin(II) chloride (SnCl2).
- However, the most characteristic oxidation states for lead and tin respectively are +2 and +4.
- Therefore, the correct answer is option A, which states that the most characteristic oxidation states for lead and tin are 2 and 4 respectively.
Which one of the elements with the following outer orbital configurations may exhibit the largest number of oxidation states? [2009]- a)3d54s1
- b)3d54s2
- c)3d24s2
- d)3d34s2
Correct answer is option 'B'. Can you explain this answer?
Which one of the elements with the following outer orbital configurations may exhibit the largest number of oxidation states? [2009]
a)
3d54s1
b)
3d54s2
c)
3d24s2
d)
3d34s2

|
Devansh Mehra answered |
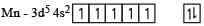
The no. of various oxidation states possible are + 2, + 3, + 4, (+ 5), + 6, + 7.
Identify the incorrect statement among the following: [2007]- a)Lanthanoid contraction is the accumulation of successive shrinkages.
- b)As a result of lanthanoid contraction, the properties of 4d series of the transition elements have no similarities with the 5d series of elements.
- c)Shielding power of 4f electron s is qui te weak.
- d)There is a decrease in the radii of the atoms or ions as one proceeds from La to Lu.
Correct answer is option 'B'. Can you explain this answer?
Identify the incorrect statement among the following: [2007]
a)
Lanthanoid contraction is the accumulation of successive shrinkages.
b)
As a result of lanthanoid contraction, the properties of 4d series of the transition elements have no similarities with the 5d series of elements.
c)
Shielding power of 4f electron s is qui te weak.
d)
There is a decrease in the radii of the atoms or ions as one proceeds from La to Lu.

|
Rohan Unni answered |
There is a steady decrease in the radii as the atomic number of the lanthanide elements increases. For every additional proton added in nucleus the corresponding electron goes to 4f subshell.
The shape of f -orbitals is very much diffused and they have poor shielding effect. The effective nuclear charge increases which causes the contraction in the size of electron charge cloud. This contraction in size is quite regular and known as Lanthanoid contraction.
Since the change in the ionic radii in the lanthanide series is very small, thus their chemical properties are similar.
The shape of f -orbitals is very much diffused and they have poor shielding effect. The effective nuclear charge increases which causes the contraction in the size of electron charge cloud. This contraction in size is quite regular and known as Lanthanoid contraction.
Since the change in the ionic radii in the lanthanide series is very small, thus their chemical properties are similar.
Qut of TiF62–, CoF63–, Cu2Cl2 and NiCl24– (Z of Ti = 22, Co = 27, Cu = 29, Ni = 28), the colourless species are: [2009]- a)Cu2Cl2 and NiCl24–
- b)TiF62–,and Cu2Cl2
- c)CoF63– ,and NiCl24–
- d)TiF62–,and CoF63–
Correct answer is option 'B'. Can you explain this answer?
Qut of TiF62–, CoF63–, Cu2Cl2 and NiCl24– (Z of Ti = 22, Co = 27, Cu = 29, Ni = 28), the colourless species are: [2009]
a)
Cu2Cl2 and NiCl24–
b)
TiF62–,and Cu2Cl2
c)
CoF63– ,and NiCl24–
d)
TiF62–,and CoF63–

|
Pankaj Banerjee answered |
The colour exhibited by transition metal ions is due to the presence of unpaired electrons in -orbitals which permits the - excitation of electrons.
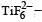 Ti is in + 4 O.S. ; 3d0 = colourless
Ti is in + 4 O.S. ; 3d0 = colourless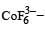 Co is in + 3 O.S ; 3d5 = coloured
Co is in + 3 O.S ; 3d5 = colouredIn Cu2Cl2– Cu is in +1 O.S. ; 3d10 – colourless
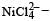 Ni is in + 2 O.S ; 3d8 – coloured
Ni is in + 2 O.S ; 3d8 – colouredAcidified K2Cr2O7 solution turns green when Na2SO3 is added to it. This is due to the formation of : [2011]- a)Cr2(SO4)3
- b)CrO42–
- c)Cr2(SO3)3
- d)CrSO4
Correct answer is option 'A'. Can you explain this answer?
Acidified K2Cr2O7 solution turns green when Na2SO3 is added to it. This is due to the formation of : [2011]
a)
Cr2(SO4)3
b)
CrO42–
c)
Cr2(SO3)3
d)
CrSO4

|
Akshat Chavan answered |
The green colour appears due to the formation of Cr+++ion

Which of the statements is not true? [2012]- a)On passing H2S through acidified K2Cr2O7 solution, a milky colour is observed.
- b)Na2Cr2O7 is preferred over K2Cr2O7 in volumetric analysis.
- c)K2Cr2O7 solution in acidic medium is orange.
- d)K2 Cr2O7 solution becomes yellow on increasing the pH beyond 7.
Correct answer is option 'B'. Can you explain this answer?
Which of the statements is not true? [2012]
a)
On passing H2S through acidified K2Cr2O7 solution, a milky colour is observed.
b)
Na2Cr2O7 is preferred over K2Cr2O7 in volumetric analysis.
c)
K2Cr2O7 solution in acidic medium is orange.
d)
K2 Cr2O7 solution becomes yellow on increasing the pH beyond 7.

|
Ruchi Chakraborty answered |
Na2Cr2O7 is hygroscopic.
Which of the following lanth anoidions is diamagnetic ? (At nos. Ce = 58, Sm = 62, Eu = 63, Yb = 70) [NEET 2013]- a)Sm2+
- b)Eu2+
- c)Yb2+
- d)Ce2+
Correct answer is option 'C'. Can you explain this answer?
Which of the following lanth anoidions is diamagnetic ? (At nos. Ce = 58, Sm = 62, Eu = 63, Yb = 70) [NEET 2013]
a)
Sm2+
b)
Eu2+
c)
Yb2+
d)
Ce2+
|
|
Rhea Iyer answered |
Diamagnetic materials are those that have all their electrons paired up in their atomic or molecular orbitals, resulting in no net magnetic moment. On the other hand, paramagnetic materials have unpaired electrons and exhibit a net magnetic moment.
In the given options, we need to identify the lanthanoid anion that is diamagnetic.
Let's analyze each option one by one:
a) Sm2: Samarium (Sm) has an atomic number of 62. In its neutral state, it has the electron configuration [Xe] 4f6 6s2. Sm2 would mean that two electrons are removed from the 6s orbital. The resulting electron configuration would be [Xe] 4f6. Since there are unpaired electrons present, Sm2 is paramagnetic and not diamagnetic.
b) Eu2: Europium (Eu) has an atomic number of 63. In its neutral state, it has the electron configuration [Xe] 4f7 6s2. Eu2 would mean that two electrons are removed from the 6s orbital. The resulting electron configuration would be [Xe] 4f7. Since there are unpaired electrons present, Eu2 is paramagnetic and not diamagnetic.
c) Yb2: Ytterbium (Yb) has an atomic number of 70. In its neutral state, it has the electron configuration [Xe] 4f14 6s2. Yb2 would mean that two electrons are removed from the 6s orbital. The resulting electron configuration would be [Xe] 4f14. Since all the orbitals are completely filled and there are no unpaired electrons, Yb2 is diamagnetic.
d) Ce2: Cerium (Ce) has an atomic number of 58. In its neutral state, it has the electron configuration [Xe] 4f1 5d1 6s2. Ce2 would mean that two electrons are removed from the 6s orbital. The resulting electron configuration would be [Xe] 4f1 5d1. Since there is an unpaired electron present in the 4f orbital, Ce2 is paramagnetic and not diamagnetic.
Therefore, the correct answer is option C, Yb2, as it is the only lanthanoid anion that has a completely filled f-orbital and no unpaired electrons, making it diamagnetic.
In the given options, we need to identify the lanthanoid anion that is diamagnetic.
Let's analyze each option one by one:
a) Sm2: Samarium (Sm) has an atomic number of 62. In its neutral state, it has the electron configuration [Xe] 4f6 6s2. Sm2 would mean that two electrons are removed from the 6s orbital. The resulting electron configuration would be [Xe] 4f6. Since there are unpaired electrons present, Sm2 is paramagnetic and not diamagnetic.
b) Eu2: Europium (Eu) has an atomic number of 63. In its neutral state, it has the electron configuration [Xe] 4f7 6s2. Eu2 would mean that two electrons are removed from the 6s orbital. The resulting electron configuration would be [Xe] 4f7. Since there are unpaired electrons present, Eu2 is paramagnetic and not diamagnetic.
c) Yb2: Ytterbium (Yb) has an atomic number of 70. In its neutral state, it has the electron configuration [Xe] 4f14 6s2. Yb2 would mean that two electrons are removed from the 6s orbital. The resulting electron configuration would be [Xe] 4f14. Since all the orbitals are completely filled and there are no unpaired electrons, Yb2 is diamagnetic.
d) Ce2: Cerium (Ce) has an atomic number of 58. In its neutral state, it has the electron configuration [Xe] 4f1 5d1 6s2. Ce2 would mean that two electrons are removed from the 6s orbital. The resulting electron configuration would be [Xe] 4f1 5d1. Since there is an unpaired electron present in the 4f orbital, Ce2 is paramagnetic and not diamagnetic.
Therefore, the correct answer is option C, Yb2, as it is the only lanthanoid anion that has a completely filled f-orbital and no unpaired electrons, making it diamagnetic.
Which one of the following ions is the most stable in aqueous solution? [2007] (At.No. Ti = 22, V = 23, Cr = 24, Mn = 25)- a)V3+
- b)Ti3+
- c)Mn 3+
- d)Cr3+
Correct answer is option 'D'. Can you explain this answer?
Which one of the following ions is the most stable in aqueous solution? [2007] (At.No. Ti = 22, V = 23, Cr = 24, Mn = 25)
a)
V3+
b)
Ti3+
c)
Mn 3+
d)
Cr3+

|
Ishaan Menon answered |
For chromium ion + 3oxidation state is most stable.
The basic character of the transition metal monoxides follows the order [2003] (Atomic Nos.,Ti = 22, V = 23, Cr = 24, Fe = 26)- a)TiO > VO > CrO > FeO
- b)VO > CrO > TiO > FeO
- c)CrO > VO > FeO > TiO
- d)TiO > FeO > VO > CrO
Correct answer is option 'A'. Can you explain this answer?
The basic character of the transition metal monoxides follows the order [2003] (Atomic Nos.,Ti = 22, V = 23, Cr = 24, Fe = 26)
a)
TiO > VO > CrO > FeO
b)
VO > CrO > TiO > FeO
c)
CrO > VO > FeO > TiO
d)
TiO > FeO > VO > CrO

|
Gauri Khanna answered |
The basic character of the transition metal monoxides follows the order:
TiO < v2o="">< cro=""><>
This means that TiO is the least basic, while FeO is the most basic among the given transition metal monoxides.
TiO < v2o="">< cro=""><>
This means that TiO is the least basic, while FeO is the most basic among the given transition metal monoxides.
Maximum oxidation state is shown by- a)Os
- b)Co
- c)Cr
- d)Mn
Correct answer is option 'A'. Can you explain this answer?
Maximum oxidation state is shown by
a)
Os
b)
Co
c)
Cr
d)
Mn

|
Shreya Gupta answered |
Os shows maximum oxidation state of +8.
Which of the following statement is not correct? [2001]- a)La (OH)3 is less basic than Li(OH)3
- b)La is actually an element of transition series rather lanthanides
- c)Atomic radius of Zr and Hf are same because of lanthanide contraction
- d)In lanthanide series ionic radius of Ln+3 ions decreases
Correct answer is option 'A'. Can you explain this answer?
Which of the following statement is not correct? [2001]
a)
La (OH)3 is less basic than Li(OH)3
b)
La is actually an element of transition series rather lanthanides
c)
Atomic radius of Zr and Hf are same because of lanthanide contraction
d)
In lanthanide series ionic radius of Ln+3 ions decreases

|
Pallavi Desai answered |
La (OH)3 is more basic than Li (OH)3. In lanthanides the basic character of hydroxides decreases as the ionic radius decreases.
The outer electronic configuration of Gd (At. No. 64) is: [NEET Kar. 2013]- a) 4f3 5d5 6s2
- b)4f8 5d0 6s2
- c) 4f7 5d1 6s2
- d)4f4 5d4 6s2
Correct answer is option 'C'. Can you explain this answer?
The outer electronic configuration of Gd (At. No. 64) is: [NEET Kar. 2013]
a)
4f3 5d5 6s2
b)
4f8 5d0 6s2
c)
4f7 5d1 6s2
d)
4f4 5d4 6s2

|
Tanisha Shah answered |
Answer of this question can be easily obtain from d and f block chapter
it is an element of lanthanoid series
Due to stability of half filled orbital answer is c
it is an element of lanthanoid series
Due to stability of half filled orbital answer is c
Four successive members of the first series of the transition metals are listed below. For which one of them the standard potential (E0M2+ /M) value has a positive sign? [2012 M]- a)Co (Z = 27)
- b)Ni (Z = 28)
- c)Cu (Z = 29)
- d)Fe (Z = 26)
Correct answer is option 'C'. Can you explain this answer?
Four successive members of the first series of the transition metals are listed below. For which one of them the standard potential (E0M2+ /M) value has a positive sign? [2012 M]
a)
Co (Z = 27)
b)
Ni (Z = 28)
c)
Cu (Z = 29)
d)
Fe (Z = 26)

|
Arpita Tiwari answered |
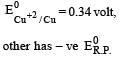
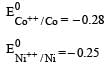
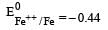
Which one of the following characteristics of the transition metals is associated with their catalytic activity? [2003]- a)Variable oxidation states
- b)High enthalpy of atomization
- c)Parmagnetic behaviour
- d)Colour of hydrated ions
Correct answer is option 'A'. Can you explain this answer?
Which one of the following characteristics of the transition metals is associated with their catalytic activity? [2003]
a)
Variable oxidation states
b)
High enthalpy of atomization
c)
Parmagnetic behaviour
d)
Colour of hydrated ions

|
Ayush Chavan answered |
Transition metals have the ability to utilize (n –1)d -orbitals. Thus variable oxidation states enables the transition element to associate with the reactants in different forms.
General electronic configuration of lanthanides is[2002]- a)(n – 2) f1 –14 (n –1) s2p6d0 – 1 ns2
- b)(n – 2) f10 –14 (n –1) d0 – 1 ns2
- c)(n – 2) f0 –14 (n –1) d10 ns2
- d)(n – 2) d0 –1 (n –1) f1 – 14 ns2
Correct answer is option 'A'. Can you explain this answer?
General electronic configuration of lanthanides is[2002]
a)
(n – 2) f1 –14 (n –1) s2p6d0 – 1 ns2
b)
(n – 2) f10 –14 (n –1) d0 – 1 ns2
c)
(n – 2) f0 –14 (n –1) d10 ns2
d)
(n – 2) d0 –1 (n –1) f1 – 14 ns2

|
Sarthak Saini answered |
The Lanthanides are transition metals from Atomic numbers 58 (Ce) to 71(Lu).
Hence the electron configulation becomes : (n –2) f 1– 14 (n – 1) s2p6 d0 – 1 ns2.
Hence the electron configulation becomes : (n –2) f 1– 14 (n – 1) s2p6 d0 – 1 ns2.
Which one of the following does not correctly represent the correct order of the property indicated against it? [2012 M]- a)Ti < V < Cr < Mn : increasing number of oxidation states
- b)Ti3+ < V3+ < Cr 3+ < Mn 3+ : increasing magnetic moment
- c)Ti < V < Cr < Mn : increasing melting points
- d)Ti < V < Mn < Cr : increasing 2nd ionization enthalpy
Correct answer is option 'C'. Can you explain this answer?
Which one of the following does not correctly represent the correct order of the property indicated against it? [2012 M]
a)
Ti < V < Cr < Mn : increasing number of oxidation states
b)
Ti3+ < V3+ < Cr 3+ < Mn 3+ : increasing magnetic moment
c)
Ti < V < Cr < Mn : increasing melting points
d)
Ti < V < Mn < Cr : increasing 2nd ionization enthalpy

|
Ashwini Khanna answered |
The melting points of the transition element first rise to a maximum and then fall as the atomic number increases manganese have abnormally low melting points.
Which is least soluble in water?- a)Ag2S
- b)AgBr
- c)AgCl
- d)AgI
Correct answer is option 'A'. Can you explain this answer?
Which is least soluble in water?
a)
Ag2S
b)
AgBr
c)
AgCl
d)
AgI
|
|
Alok Mehta answered |
Water is polar. Using the axiom, “like dissolves like”, hexane would water- insoluble. CH3OH, CH3CO2H and CH3NH2 are polar and can participate in hydrogen- bonding interactions with water, which would make these compounds quite water- soluble.Silver sulfide (Ag 2S) is the sulfide of silver. It is useful as a photosensitizer in photography. .... The crystallography of silver sulfide, Ag2S. Zeitschrift fur ... Ag2S. least soluble in water
KMnO4 can be prepared from K2MnO4 as per the reaction: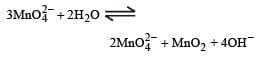 The reaction can go to completion by removing OH– ions by adding. [NEET 2013]
The reaction can go to completion by removing OH– ions by adding. [NEET 2013]- a)KOH
- b)CO2
- c)SO2
- d)HCl
Correct answer is option 'B'. Can you explain this answer?
KMnO4 can be prepared from K2MnO4 as per the reaction:
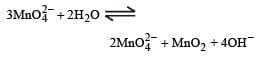
The reaction can go to completion by removing OH– ions by adding. [NEET 2013]
a)
KOH
b)
CO2
c)
SO2
d)
HCl

|
Ayush Chavan answered |
HCl and SO2 are reducing agents and can reduce MnO4–. CO2 which is neither oxidising and nor reducing will provide only acidic medium. It can shift reaction in forward direction and reaction can go to completion.
Which is the most stable oxidation state of iron?- a)+2
- b)+3
- c)0
- d)+1
Correct answer is option 'B'. Can you explain this answer?
Which is the most stable oxidation state of iron?
a)
+2
b)
+3
c)
0
d)
+1

|
Charvi Ahuja answered |
Fe has d5 configuration in +3 oxidation state.
Among the following series of transition metal ions, the one where all metal ions have 3d2 electronic configuration is(At. nos. Ti = 22; V = 23; Cr = 24; Mn = 25) [2004]- a)

- b)
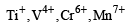
- c)
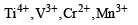
- d)
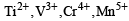
Correct answer is option 'D'. Can you explain this answer?
Among the following series of transition metal ions, the one where all metal ions have 3d2 electronic configuration is(At. nos. Ti = 22; V = 23; Cr = 24; Mn = 25) [2004]
a)

b)
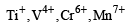
c)
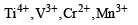
d)
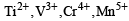

|
Rashi Agarwal answered |
It will be option D write the general configuration first and then take out electrons as required
Chapter doubts & questions for d- and f-Block Elements - 30-Day Revision Course for NEET 2026 is part of NEET exam preparation. The chapters have been prepared according to the NEET exam syllabus. The Chapter doubts & questions, notes, tests & MCQs are made for NEET 2026 Exam. Find important definitions, questions, notes, meanings, examples, exercises, MCQs and online tests here.
Chapter doubts & questions of d- and f-Block Elements - 30-Day Revision Course for NEET in English & Hindi are available as part of NEET exam.
Download more important topics, notes, lectures and mock test series for NEET Exam by signing up for free.
;
Signup to see your scores
go up
within 7 days!
within 7 days!
Takes less than 10 seconds to signup